Cell death in grape berries: varietal differences linked to xylem pressure and berry weight loss
Joanne Tilbrook A B and Stephen D. Tyerman A B CA Cooperative Research Centre for Viticulture, PO Box 154, Glen Osmond, SA 5064, Australia.
B School of Agriculture, Food and Wine, University of Adelaide, Glen Osmond, SA 5064, Australia.
C Corresponding author. Email: steve.tyerman@adelaide.edu.au
Functional Plant Biology 35(3) 173-184 https://doi.org/10.1071/FP07278
Submitted: 22 November 2007 Accepted: 13 March 2008 Published: 23 April 2008
Abstract
Some varieties of Vitis vinifera L. can undergo berry weight loss during later stages of ripening. This defines a third phase of development in addition to berry formation and berry expansion. Berry weight loss is due to net water loss, but the component water flows through different pathways have remained obscure. Because of the very negative osmotic potential of the cell sap, the maintenance of semipermeable membranes in the berry is required for the berry to counter xylem and apoplast tensions that may be transferred from the vine. The transfer of tension is determined by the hydraulic connection through the xylem from the berry to the vine, which changes during development. Here we assess the membrane integrity of three varieties of V. vinifera berries (cvv. Shiraz, Chardonnay and Thompson seedless) throughout development using the vitality stains, fluorescein diacetate and propidium iodide, on fresh longitudinal sections of whole berries. We also measured the xylem pressure using a pressure probe connected to the pedicel of detached berries. The wine grapes, Chardonnay and Shiraz, maintained fully vital cells after veraison and during berry expansion, but began to show cell death in the mesocarp and endocarp at or near the time that the berries attain maximum weight. This corresponded to a change in rate of accumulation of solutes in the berry and the beginning of weight loss in Shiraz, but not in Chardonnay. Continuous decline in mesocarp and endocarp cell vitality occurred for both varieties until normal harvest dates. Shiraz grapes classified as high quality and sourced from a different vineyard also showed the same death response at the same time after anthesis, but they displayed a more consistent pattern of pericarp cell death. The table grape, Thompson seedless, showed near to 100% vitality for all cells throughout development and well past normal harvest date, except for berries with noticeable berry collapse that were treated with giberellic acid. The high cell vitality in Thompson seedless berries corresponded to negative xylem pressures that contrasted to the slightly positive pressures for Shiraz and Chardonnay. We hypothesise that two variety dependent strategies exist for grapevine berries late in development: (1) programmed cell death in the pericarp and loss of osmotically competent membranes that requires concomitant reduction in the hydraulic conductance via the xylem to the vine; (2) continued cell vitality and osmotically competent membranes that can allow high hydraulic conductance to the vine.
Additional keywords: berry shrivel, berry weight loss, cell death, grape berry development.
Introduction
There has been much interest recently in the water budget of the developing grape berry (Bondada et al. 2005; Keller et al. 2006; Rogiers et al. 2006; Thomas et al. 2006; Tilbrook and Tyerman 2006; Zhang et al. 2006b). This is for two reasons. First, the berry is a good model for other fleshy fruits and can provide some generalisations of how water movement into and out of the fruit is regulated through changes in solute partitioning, phloem and xylem transport, and transpiration. Second, the water content of the harvested berries and the way in which water is retained in the berry has a large impact on quality; whether this be via concentration of soluble solids and flavour molecules in wine grapes, or through crispness (turgidity) of the fruit in the case of table grapes. Yield is also determined largely by water content since water makes up the major component of berry mass. Loss of water from the berry can be substantial in some varieties and this can reduce yield by over 25%, for example in Shiraz (McCarthy 1999; McCarthy and Coombe 1999).
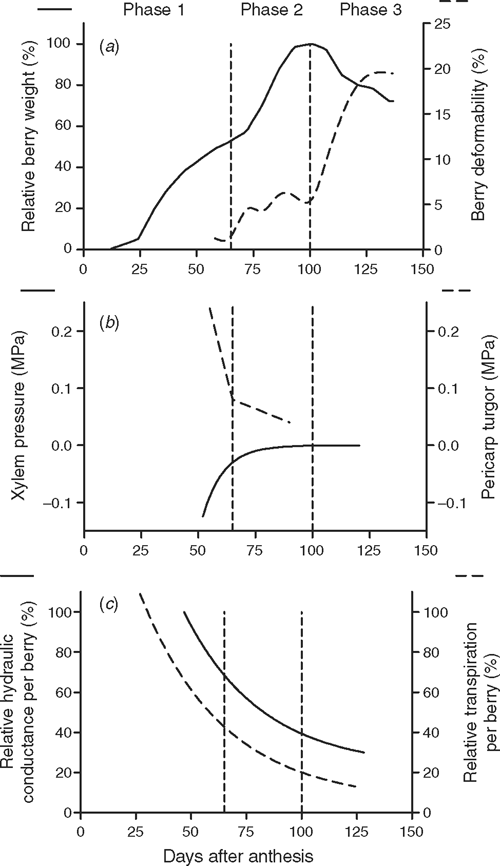
It is generally agreed that before veraison during the first phase of berry development (berry formation, phase 1, Fig. 1), the berry transpires and water inflow occurs via the phloem and the xylem (Lang and Thorpe 1989; Greenspan et al. 1994; Dreier et al. 2000; Rogiers et al. 2004, 2006). After a lag in expansion and just after the berry softens, sugar and water accumulation rapidly increase via phloem import, here referred to as phase 2 (Fig. 1). After the berry reaches maximum weight a third phase is apparent in some varieties, when berry weight loss begins (Sadras and McCarthy 2007). This can occur before grape flavour development for winemaking is evident (Coombe and McCarthy 1997), and sugar concentration can be further increased by a combination of volume decrease of the berry and further sugar import, although further sugar import to the berry seems to be plastic between seasons (Sadras and McCarthy 2007). Fig. 1 shows these phases of berry development in context with other changes in xylem function and berry water relations taken from the literature and our own data. It should be noted that phase 1 incorporates Stages I and II by Coombe (1992).
|
During phase 2, the berry appears to become less hydraulically connected to the vine (Greenspan et al. 1994, 1996; Tyerman et al. 2004; Tilbrook and Tyerman 2006). From earlier dye uptake studies and calcium uptake into the berry (as a xylem tracer) (Findlay et al. 1987; Creasy et al. 1993; Rogiers et al. 2001), the hydraulic isolation was proposed to be due to discontinuity of xylem vessels in the berry. However, our previous quantitative measurements of the xylem pathway to the berry showed that the pathway remained functional, though hydraulic conductance was reduced in magnitude depending on variety (Fig. 1; Tyerman et al. 2004). Bondada et al. (2005) qualitatively confirmed that xylem hydraulic conductance continued in post veraison berries by demonstrating that dye uptake could still occur provided the appropriate driving force on water flow to the berry could be sustained. Tyerman et al. (2004) also showed that the xylem pressure changed from being negative to slightly positive during veraison, supporting the view that the nature of the driving force for water movement to the berry in the xylem changes during phase 2 (Fig. 1). These discoveries have focussed attention on the solute partitioning between apoplast and symplast in the berry because this will determine how the very negative osmotic potential of the berry juice, largely mesocarp cell sap, is translated into the driving force for water movement, both through the phloem and the xylem.
The demonstration by (Zhang et al. 2006b) that phloem unloading switches from symplastic to apolastic during veraison, suggests that osmotic potential of the apoplast decreases. This could drive greater flow through the phloem (Patrick 1997) and may explain the transition to slightly positive xylem pressure measured by Tyerman et al. (2004). An important issue remains, however, and this is whether or not the osmotic competence of the cell membranes, particularly of the large mesocarp cells, changes in the post veraison berry. This is important because normal cell membranes will show semipermeability, and for large solutes like sugars, an osmotic potential difference across the membrane is reflected by an equivalent hydrostatic pressure difference. The large negative osmotic potential of the mesocarp cells could balance only negative apoplast pressures and xylem tensions if the membranes remain semipermeable.
There is indirect evidence that cell membranes become leaky in the mesocarp of post veraison Riesling (Lang and Düring 1991; Dreier et al. 1998) and cell compartmentation can break down in Thompson seedless (Dreier et al. 1998). However, turgor at low pressures (0.05 MPa), and, therefore, membrane semipermeability and cell vitality, was maintained in cells to a depth of 1.5 mm below the cuticle until 100 days after anthesis (DAA) in several wine grape varieties, including Chardonnay (Thomas et al. 2006). The reduction in turgor pressure at veraison (transition from phase 1 to phase 2) of the mesocarp closely corresponds to increased deformability of the berry (Fig. 1) and suggests that the apoplast osmotic potential declines.
The turgor pressure observations, like most, do not continue into the final 2–3 weeks before harvest, because the fruit becomes very deformable (Fig. 1), and berry contents can be extremely difficult to work with. It is during phase 3, post 90–100 DAA in varieties like Shiraz, that considerable loss of weight can occur to the point where the berries may shrivel (Fig. 1). This loss of weight has been proposed to be due to a combination of reduced phloem inflow and continued transpiration (McCarthy and Coombe 1999; Rogiers et al. 2004; Tyerman et al. 2004; Keller et al. 2006) though backflow to the vine via the xylem may also contribute (McCarthy and Coombe 1999; Rogiers et al. 2004; Tyerman et al. 2004; Keller et al. 2006). Backflow was directly demonstrated by dye loading at the stylar end of post veraison berries and observing the dye in the xylem of the vine (Keller et al. 2006). Though the apoplasmic water of the berry is available for movement back to the vine, Keller et al. (2006) conclude that the berry cell membranes remain semipermeable, thereby making it difficult for the leaves to extract water from the berry cells because of the large negative osmotic potential of the cell sap. Thus, if backflow alone were to account for a loss of up to 30% of maximum weight, as is often observed in Shiraz, there would have to be a loss of membrane semipermeability for a large proportion of the cells in the berry.
The aim of this work was to test the hypothesis that cells across the pericarp of berries maintain membrane competence and vitality until harvest maturity is achieved. It formed part of a project to ascertain the cause of weight loss in Shiraz berries. Shiraz, Chardonnay and Thompsons seedless fruit were compared, because Chardonnay does not normally show weight loss and Thompson seedless was found by us to have very different xylem pressures compared with the other varieties. We used two vital stains, fluorescein diacetate (FDA) and propidium iodide (PI). These dyes are a well documented and reliable method of determining cell vitality in cell suspensions and flow cytometry (Jones and Senft 1985). FDA is a non-polar, non-fluorescent molecule that crosses cell membranes. Once in the cytoplasm, esterases cleave the acetate groups from the molecule and it becomes highly polar and fluorescent green. If a cell does not have an active metabolism, it will not fluoresce. PI is a membrane impermeant dye that enters cells only when cell membranes are disrupted or not intact. It binds non-covalently, intercalating in a stoichiometric manner with single and double stranded DNA and RNA. When intercalated it fluoresces red (Cosa et al. 2001; Bernas et al. 2004; Kral et al. 2005). The loss of cell membrane integrity in plants is considered to mark the end of homeostasis and indicates cell death (Noodén 2004). Using these vital stains we examined the vitality of cells in medial longitudinal sections of fresh berries through development. The data were analysed to provide a new insight into cell vitality and cell membrane competence through berry development that we relate to measurements of xylem pressure using the pressure probe attached to individual berry pedicels.
Materials and methods
Fruit material
Experimental fruit (Vitis vinifera L.) used in the time course experiments was from the Coombe vineyard (Shiraz BVRC12 and Chardonnay I10V1, 12 years old) and Alverstoke vineyard (Thompson seedless M12, not treated with giberellic acid, 4 years old) on the Waite Campus of the University of Adelaide. Bunches of fruit were labelled individually when an estimated 50% of the flower caps from that bunch had dehisced. The day this occurred was designated as ‘anthesis’. Anthesis was complete over 1–2 days. Premium quality Shiraz (unknown clone, ~30 years old) was obtained on the day of harvest courtesy of Hardys, McLaren Vale from a McLaren Flat site. Thompson seedless fruit treated with giberellic acid and showing signs of berry collapse was from CSIRO Merbein, courtesy of Mike Treeby and Tori Nguyen. All data from vineyard fruit was obtained during the 2005–06 season. The pre-veraison Shiraz BVRC12 berries in the fluorescent dye control experiment were from glasshouse grown vines at the Plant Research Centre in the Waite Campus precinct.
Berry weight, °Brix and osmolality
Berry weight and °Brix data reflect whole vineyard berry development. Weights are the means of 50 berry samples. For Shiraz, samples of five berries from proximal, mid and base of two random bunches on separate vines from each of five replicate panels in separate rows that formed part of a randomised block rootstock trial were collected. For Chardonnay, five berry samples from proximal, mid and base of 10 random bunches on separate vines in a row of 24 adjacent vines. These data were not collected for Thompson seedless as insufficient experimental fruit was locally available. Berries collected for weight samples were crushed, juice collected and briefly centrifuged to settle any solids. °Brix of the juice was measured using a temperature compensated digital refractometer (Model PR101; ATAGO, Tokyo, Japan). A water vapour pressure osmometer (Model 5500; Wescor, Logan, UT, USA) was used to measure juice osmolality, which was converted to osmotic potential (Ψπ = −RTC, where r = 8.3143 J/mol.K and T = absolute temperature in K). Solutes per berry (g) was approximated by the product of berry weight and °Brix/100 (McCarthy and Coombe 1999).
Vital and non-vital staining of pericarp tissue
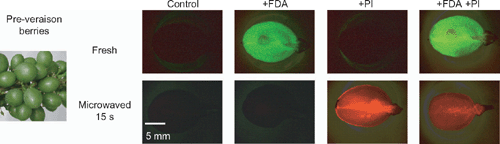
Bunches (all clones of Shiraz and Chardonnay) or clusters (Thompson seedless) were cut from within the vine canopies, placed in plastic bags on ice and taken to the laboratory (~500 m). Berries were sectioned longitudinally between the seeds (where present). One-half of each berry was pooled and crushed for juice to measure °Brix and to calculate osmotic pressure (as above). The FDA section of the method was developed from that kindly shared by Professor Ken Shackel, UC Davis. From a 4.8 mm FDA (Sigma-Aldrich, St Louis, MO, USA) in acetone stock solution, an aqueous 4.8 μm FDA solution (Oparka and Read 1994) was prepared to a similar (to within 10%) osmotic pressure as the berry juice with sucrose, then applied to excess on the cut surface of the half berries. After 15 min incubation, sectioned berries were viewed with a Leica M-Z FL111 (Leica, Wetzlar, Germany) dissecting microscope at minimum magnification under ultraviolet light with a green fluorescent protein filter in place. Images were promptly obtained with a Leica DC 300F camera and Image Pro Plus 5.1 (MediaCybernetics, Bethsda, MD, USA) with consistent settings and exposure times.
The FDA solution was blotted from the cut berry surfaces, then a counterstain of aqueous 190 μm PI solution (Sigma-Aldrich) (Oparka and Read 1994) freshly prepared from an aqueous stock solution and made to a similar (to within 10%) juice osmotic pressure with sucrose. It was applied to excess, incubated for 10 min, viewed and imaged as before. To confirm that the method was a reliable indicator of cell membrane competence and cell vitality, fresh and microwaved pre-veraison berries were prepared and imaged according to the described method (Fig. 2). The pre-veraison fruit for the set of control sections was prepared as above for the fresh sections or microwaved for 15 s at 650 W before the dyes were applied. Exposure times were consistent for the set. The fresh, sectioned berries showed a vivid fluorescent green response to FDA indicating that the cells across the pericarp had competent membranes and living cytoplasm. Berries that had been killed by microwaving had no fluorescent response to FDA (Fig. 2). The results of the PI application to the sections were a direct contrast. No red fluorescence was visible in the fresh sections (with the exception of a sliver of seed coat), however, it was intensely visible in the pericarp cells of the microwaved, cell membrane disrupted samples. PI is known to stain cell walls, but this was not visible, nor was auto-fluorescence or artefacts at the cut surface of the pericarp using this method at low magnification.
|
Although we refer here to the pericarp (comprising exocarp mesocarp and endocarp), the microscopy technique used in this study prevented cellular details in the exocarp region to be delineated. The vitality observations are weighted more to the large volume of the mesocarp and endocarp. However, vitality or death of cells of the exocarp can be seen as thin green outlines or red regions in the sections.
Analysis of vitality
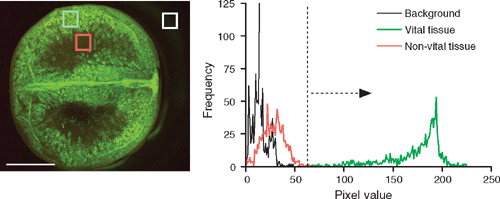
Pixel analysis of Shiraz and Chardonnay sections stained only with FDA was performed by Global Laboratory Image/2 version 2.5 (Data Translation, Marlboro, MA, USA). Comparative analysis of pixel blocks or line transects within images showed that pixels with arbitrary values designated as >75 were vital and reflected living cytoplasm in cells (Fig. 3). Two methods were compared: (1) the berry cuticle was outlined and pixels within the outline (i.e. the cut surface) analysed. ‘Vital’ pixels were expressed as a percentage of the total number of pixels; (2) line transects were taken across the distal, mid and mesial (DMM) of berry longitudinal sections and the percentage of total pixels with values >75 were averaged between the three sections. A separate transect was taken through obviously vital regions of the pericarp where cell cytoplasm surrounding large vacuoles could be delineated. The percentage of total pixels with values >75 was recorded and the ratio of the averaged DMM over the pericarp transect value was converted to a percentage. This is referred to as ‘relative cell vitality’. The second method yielded qualitatively similar results to the first method, but was deemed more suitable because it could account for the expansion of living pericarp cells, which resulted in a reduced count of vital pixels due to ‘dilution’ by the large central vacuoles. The second method also accounted for slight variations in exposure between preparations since it was self referencing within a berry. To quantify the structural variability in location of living and dead cells between high quality and mid-quality fruit, DMM transects were drawn and pixel values graphed for each transect.
|
Xylem equilibrium pressures
Xylem equilibrium pressures of berries attached to the pressure probe via the pedicel were measured as detailed by Tyerman et al. (2004). Briefly, whole shoots with bunches attached were cut from the field grown vines described above and were transported immediately to the laboratory with the cut end immersed in water. Once in the laboratory an individual berry and pedicel was cut from the bunch while under water. To seal the pedicel with the pressure probe a piece of tubing 15–20 mm long (Tefzel-Schlauch (Sinsheim, Germany) tubing internal diameter (ID) 1.0 mm outside diameter (OD) 1.6 mm or ID 1.6 mm OD 3.2 mm depending on pedicel diameter) was flared at one end in order to snugly fit the pedicel. The pedicel was sealed into the flared end with a cyanoacrylate glue (Super Glue TM or Loctite TM 401 or 406 adhesive with tubing treated with 770 primer). The water used to fill the tubing and probe was millipore purified water de-aerated by vacuum treatment and filtered to 0.2 μm. As the seal was tightened around the tubing the pressure was elevated in the system and was allowed to relax to a steady-state level.
Results
Pericarp cell death is evident in berries of Chardonnay and Shiraz late in development, but not Thompson seedless
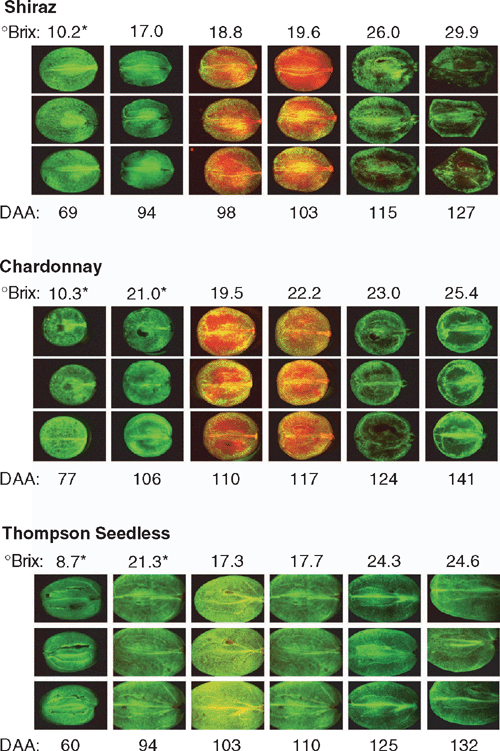
At 73 days after anthesis (DAA), Shiraz and Chardonnay berries moved from the lag phase of berry development (late part of phase 1) into the rapid phase of weight and sugar accumulation (phase 2). The staining of sections of both varieties with FDA indicated that cells across the pericarp maintained vitality through veraison and continued as the fruit approached maximum weight. In Shiraz, the berries were still enlarging at 98 DAA when counterstaining with PI showed an intense red fluorescent response across the pericarp, which was repeated approaching maximum weight at 103 DAA (Fig. 4a). In Chardonnay the berries reached maximum weight at 108 DAA and exhibited a PI response at 110 DAA that was sustained at 117 DAA (Fig. 4b). Yellow fluorescence was visible where tissue showed a mixed response – living and dead cells in proximity. Although the period of sudden PI response did not occur at the same number of days after anthesis for the wine grape varieties, it was contemporaneous in terms of the calendar date. No weather, water or other stresses were noted over this period. We also observed similar death events in berries from glasshouse grown vines grown during winter of 2006 and this also did not correspond to any particular climate change. More cells adjacent to the central and peripheral vasculature maintained cell membrane competence compared with the body of the mesocarp.
|
Thompson seedless berries had a slight blush of PI red fluorescence at 103 DAA (Fig. 4c), contemporaneous with the initial wine grape PI response. This was not sustained a week later and does not appear to be linked to sugar content, number of days after anthesis or maximum weight of the berries. In complete contrast to the wine grapes, the Thompson seedless berries continued to have cells with functioning cytoplasm and competent membranes across the entire pericarp up to and beyond normal harvest date. The cell vacuoles enlarge and appear dark, particularly at maximum distance from the central and peripheral vascular bundles, but vital cytoplasm was clearly visible within each cell.
Cell vitality relative to sugar and water relations of the berry
Relative cell vitality as a function of days after anthesis is plotted in Fig. 5a for each of the grapevine varieties. This data is shown in the context of juice osmotic potential for all the varieties (Fig. 5b), and the weight and sugar accumulation in the berries of Chardonnay and Shiraz (Fig. 5c, d).
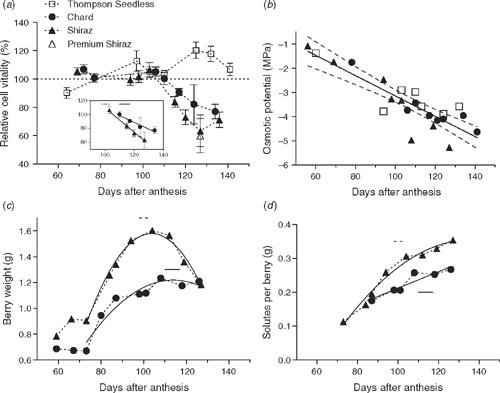
|
At or just after veraison all varieties showed 100% relative cell vitality. This was maintained throughout the entire measurement period for Thompson seedless and until maximum berry weight for Chardonnay and Shiraz. The measurements above 100% for Thompson seedless are ascribed to the higher density of cells adjacent to central and peripheral vascular bundles of the berry when compared with the mesocarp cells in the calculation of relative cell vitality.
The range of days after anthesis over which the PI fluorescence was observed in Shiraz and Chardonnay, are indicated on Fig. 5 as horizontal bars. These events corresponded with the onset of a linear decline in relative cell vitality measured with FDA in both varieties (inset Fig. 5a). The linear regressions for this phase of declining vitality indicated that Shiraz had a significantly higher rate of decline compared with Chardonnay. The initial death events also corresponded to the period just before maximum berry weight in Shiraz and just after maximum berry weight in Chardonnay (Fig. 5c). The death event and onset of decline in cell vitality also corresponded to a change in slope of accumulation of solutes per berry in Shiraz and a plateau in Chardonnay (Fig. 5d).
Xylem pressure measured using the pressure probe attached to the pedicel of individual berries is shown in Table 1. These measurements were taken in the period when cell vitality was declining. Despite the very negative osmotic potentials of the berry juice of Chardonnay and Shiraz (Fig. 5b) the xylem pressures of detached berries were rather small and positive (note that values given are in KPa). Contrasting behaviour was observed for Thompson seedless berries that had higher osmotic potentials (Fig. 5b) but which developed negative xylem pressures that would cavitate the pressure probe. In this case the measurements given in Table 1 are extrapolations of the exponential approach to equilibrium after the berry was attached to the probe (Tyerman et al. 2004).

|
Aberrant behaviour detected using the vitality assay
Transects across the pericarps of premium Shiraz fruit from 30-year-old vines in McLaren Flat, South Australia, were compared with the mid-quality fruit from 12-year-old Shiraz vines in the Coombe vineyard. The premium fruit showed a consistent and distinct pattern of vital and non-vital cells across the pericarp which can be graphically represented (Fig. 6a). This structural pattern was consistent in the sections examined. At harvest maturity the cells in the premium fruit around the peripheral and central vasculature clearly maintain competent membranes but the mesocarp at maximum distance from the vascular tissue shows no response to FDA. This suggests that there are distinct regions within the pericarp where there are no vital cells at harvest. The mid-quality Shiraz showed more structural variation in the distribution of cells with and without a functioning cytoplasm compared with the premium Shiraz. The apparent loss of membrane competence in cells, or cell death, was spread across the pericarp in a less organised manner (Fig. 6b), although some berry sections did show similarities to the premium fruit pattern of cell death. Field-grown premium and the mid-quality Shiraz were harvested for winemaking on the same day at 29.6 and 30 °Brix, respectively. Although the apparent ‘vitality’ structure of the two sets of berry sections are quite different on the day of harvest (Fig. 6), there was no significant difference in the relative cell vitality measurement (unpaired t-test, n = 6–7, P = 0.49, Fig. 5a). The fruit from both locations appeared similarly shrivelled.
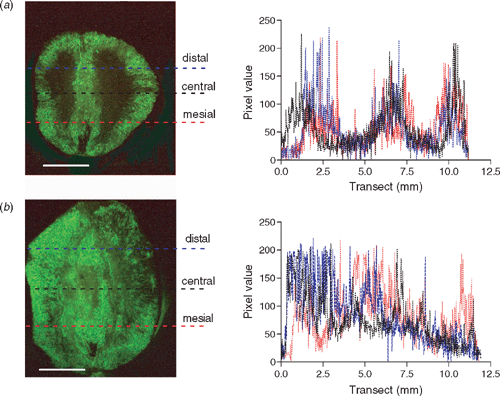
|
The FDA method was applied to Thompson seedless berries (21 °Brix, unknown DAA) that had been treated with giberellic acid, and had signs of early stages of berry collapse (Fig. 7a). Regions of cells with no living cytoplasm were observed in the distal half of the pericarp, with adjacent distal regions appearing to show smaller areas of apparent cell death. Distinct differences were noted when this fruit was compared with fruit that was treated with giberellic acid but had no signs of berry collapse (20 °Brix, Fig. 7b), and untreated fruit (24.3 °Brix, Fig. 7c). The berries showing signs of berry collapse had elongated, semi-rectangular cells that appeared to be very loosely stacked in rows across the pericarp between the central and peripheral vascular bundles, and cell death had already occurred in some regions. The central and peripheral vascular bundles appear disrupted around the regions of cell death. The giberellic acid treated fruit that was sound, and untreated berries (Fig. 7b, c) had dense, compact, irregularly shaped cells across the pericarp, with some compact elongated cells in the mesocarp between the central and peripheral vasculature. The giberellic acid treated fruit was much larger than untreated fruit, as would be expected.
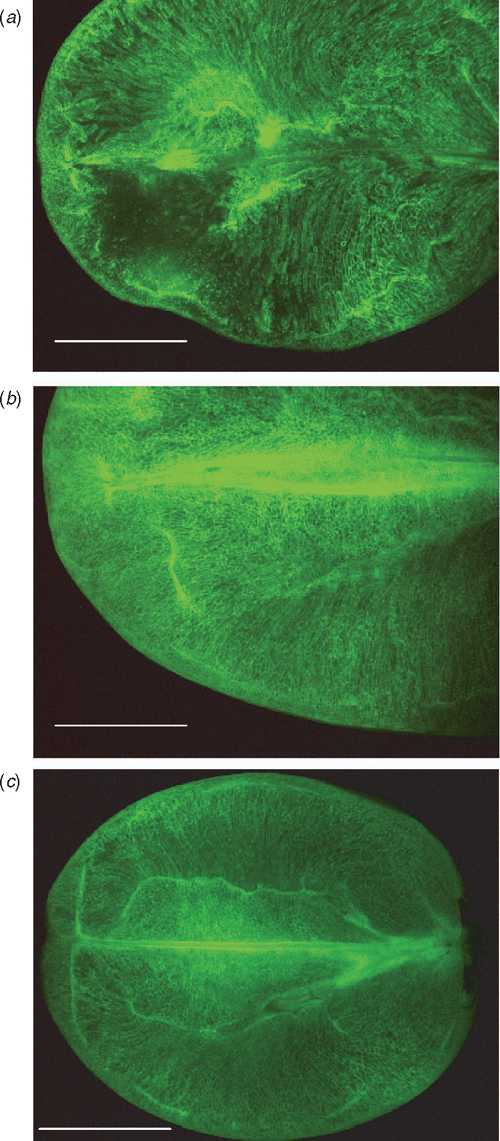
|
Discussion
Veraison is the beginning of the ripening phase (phase 2 in Fig. 1) of grape berry development in V. vinifera. It is indicated by softening of the berry, a rapid accumulation of hexose sugars, and in some varieties, colour development. The fleshy, carbohydrate rich fruit is thought to have selectively evolved as a high value food reward for animals, birds in particular, that consume it and disperse the seeds widely without physical damage (Hardie and O’Brien 1988). At veraison, seeds have reached full size (Hardie et al. 1996) and can germinate with cold treatment (reviewed in Pratt 1971). Unpalatable phenolic compounds in berries reduce from veraison (Adams 2006) as sugars are accumulating. A second phase of softening and further sugar concentration is evident in some wine grape varieties (phase 3 in Fig. 1). The selection by human kind of cultivated varieties of grapevines for wine making or table grapes probably enhanced or reduced some of these characters over time, depending on the desired end use of the fruit. Contrasting behaviour is exhibited between the wine grape and table grape varieties observed in this work. We show here the first direct evidence of loss of membrane competence in the mesocarp in two wine grape varieties that corresponds to the phase 3 stage of development. This occurs at or near maximum berry weight and clearly defines the third phase of berry development independently of whether berry weight loss occurs or not. This third phase of development defined by loss of cell vitality was not evident in the berries of the table grape variety Thompson seedless.
From the fluorescent dye test series it is clear that autofluorescence, damaged cells on the cut surface and other artefacts do not interfere at this ‘macro’ level of microscopy. In the second phase of development in Chardonnay, Shiraz and Thompson seedless berries, the dye studies initially showed vital cells across the pericarp and no visible response to PI. On the same day (14 February 2006), the wine grape sections showed a dramatic, vivid red response to PI, and the Thompson seedless sections had a slight blush of red fluorescence. The response was the same a week later in the wine varieties, but was not repeated in the table grape. No weather, water or other stresses were noted over this period. We believe that the contemporaneous calendar timing of the PI response was not an artefact of any treatment we imposed on the sampling regime for the following reasons: (i) the strong PI response in the wine grapes corresponded to the beginning in the decline of cell vitality using the FDA method that was observed over the following weeks. It also corresponded to other developmental changes including maximum berry weight and slowing of sugar accumulation; (ii) the same degree of cell death measured with FDA was observed in fruit sourced from a different region and vineyard, and measured at the same time after anthesis.
The reason why the PI response was not sustained in the wine varieties when the FDA studies show increasing cell death is not clear. Perhaps the nucleic acids denature to a degree that a structural change interferes with the PI intercalating into a conformation that is fluorescent. Compounds in the cytosol of other plants have been found to reduce PI accessibility of DNA when cells are lysed (Price et al. 2000; Noirot et al. 2003; Loureiro et al. 2006). If intercalation with RNA was responsible for the fluorescence seen, RNA is rapidly degraded during senescence (Jones 2004) and there may be insufficient quantities to fluoresce after the initial response. This needs further work, but it is likely that the strong PI response observed in the two wine grapes is part of the cell death process in the fruit because it mirrors the increasing, subsequent pattern of cell death across the pericarp indicated by the FDA data. Although PI can stain cell walls it was not evident in the control experiments at the magnification used. Total DNA in the pericarp of Shiraz berries has been found to peak at 35 DAA, then be maintained until 100 DAA on a per berry basis (Ojeda et al. 1999). The dataset stops several days after maximum berry weight was achieved at 95 DAA, which is probably the period where loss of membrane competence has started to occur so it cannot be related to data in this paper. Anecdotal evidence of increasing difficulty in extracting DNA and RNA as harvest maturity of berries approaches may be related to cell death and subsequent catabolism of nucleic acids. This is certainly an area of research that warrants further investigation.
Cells surrounding the central and peripheral vasculature of berry sections maintained vitality compared with the rest of the pericarp, although this reduces as harvest approaches. This pattern is highly evident in the transects of the premium Shiraz berries (Fig. 6). Although the relative vitality was not significantly different at harvest, a difference was found in the distribution or pattern of vital and non-vital cells across the sectioned pericarp of premium Shiraz fruit from 30-year-old vines in McLaren Flat when compared with the mid-quality fruit from 12-year-old Shiraz vines in the Coombe vineyard. The premium fruit showed a consistent and distinct pattern of vital and non-vital cells across the pericarp. At harvest maturity the cells in the premium fruit around the peripheral and central vasculature clearly maintain competent membranes (and living cells) but the mesocarp at maximum distance from the vascular tissue shows no response to FDA. This suggests that there are distinct regions within the pericarp where there are no vital cells at harvest. The mid-quality fruit showed variation in pattern and less organised cell death across the pericarp.
Visible disintegration of cell membranes are described as terminal or late events in the cell death process in plants (Noodén 2004). In later stages of berry development, the mesocarp cells of Traminer have lost internal membranes and cell contents including lipids, starches and polyphenols were mixed within (Hardie et al. 1996). Autolysis of vacuoles is likely to be indicative of cell death (Thomas et al. 2003) and no functioning cytosol clearly means a cell is dead. Both mitochondria and plasma membranes appeared intact in mesocarp cells late in the ripening of fruit of the table grape V. vinifera × V. labrusca cv. Kyoho (Zhang et al. 1997). Mitochondrial integrity is often maintained until late in the senescence process (Noodén 2004). The continuing vitality we observed in Thompson seedless indicates that cell death is variety dependent and, therefore, vitality may also be maintained in the Kyoho grape.
In contrast to the two wine grape varieties, membrane competence and vitality of pericarp cells in Thompson seedless berries was maintained throughout development and into the post harvest period. This may explain why Thompson seedless grapes maintain turgidity, crispness and consumer appeal post harvest. The continuing cell vitality data corresponds with pedicel xylem pressure measurements showing that this variety generates negative pressures in the pedicel xylem until harvest. This indicates a continuing, functioning xylem connection to the apoplast, and cells with osmotically competent membranes that sustain a pressure gradient across them as the berry matures. This observation is consistent with anecdotal evidence of growers severing fruiting canes of Thompson seedless from the vine to prevent water uptake and berry splitting if there is a rain event as harvest approaches. The data from Thompson seedless berries in early stages of collapse warrants further investigation using the methods described here, which provide a quick visual way of identifying cell death and patterns of aberrant cell death.
Recent work shows a change of phloem unloading from a symplastic to an apoplastic pathway at or just before veraison (Zhang et al. 2006b). Both Zhang et al. (2006b) and Lang and Düring (1991) presented direct evidence that the apoplastic solute concentration rises from 65 and 60 DAA, respectively, in a table grape and a wine grape variety. Lang and Düring (1991) found that there was no significant difference in the osmotic potential of berry pedicel xylem exudate and berry juice between 60 and 110 DAA. These data clearly separate the increase in apoplastic solute concentration in berries from the later loss of cell membrane competence that is shown in this paper. This means that the ‘leaky membrane’ theory described by Lang and Düring (1991) as an explanation of the rise in apoplast solute concentration at veraison is not supported, but it is apt later in berry development in Chardonnay and Shiraz. The loss of cell vitality in Chardonnay and Shiraz compared with its maintenance in Thompson seedless suggests that their senescence processes are significantly different, so care needs to be taken when applying research results from one variety to another.
The image analysis showed that Shiraz and Chardonnay had a significantly different time course of loss of cell vitality in the pericarp relative to DAA. Regions in the pericarp of Shiraz entered the senescence or cell death phase earlier in berry development compared with Chardonnay, and this difference increases significantly because relative vitality declines more rapidly in Shiraz berries. This difference is exacerbated by later harvesting for winemaking (127 DAA) compared with the Chardonnay fruit that was harvested at 117 DAA.
Our data suggests an hypothesis for the mechanism of weight loss observed in Shiraz berries that occurs generally after 90–100 DAA (McCarthy 1999; Tyerman et al. 2004). Since the membranes of the pericarp cells begin to loose semipermeability at this point, as judged by loss of vitality, the large negative osmotic potential of the berry sap is no longer effective in opposing the xylem tensions developed by the leaves. We have shown previously that Shiraz berries late in development maintain higher hydraulic conductance back to the vine compared with Chardonnay berries (Tyerman et al. 2004), therefore, significant volume may leave the berry back to the vine in Shiraz. Reduced phloem inflow and continued transpiration would all combine to cause a net loss of water from Shiraz berries. Chardonnay berries also maintain some finite conductance back to the vine (Tyerman et al. 2004), but the quantitative conductance is much less than in Shiraz and drops to very low levels by 90 DAA, that is, at about the same time that cell death begins to be evident in Chardonnay. Recent work of Bondada et al. (2005) and Keller et al. (2006) demonstrated qualitatively that backflow can occur, however, our previous work demonstrated large varietal differences in a quantitative measure of the pathway for this flow, the hydraulic conductance.
More generally we propose that there is a balance in development between the programmed cell death of the pericarp cells and the decline in hydraulic conductance from the berry to the vine. If the hydraulic conductance to the vine is reduced sufficiently when the pericarp cell membranes lose semipermeability (cells non-vital), backflow would be reduced. This would be the case for Chardonnay. For Shiraz, there is an apparent miss-timing where the pericarp cells loose vitality and osmotic competence earlier in development, but the hydraulic conductance remains high back to the vine later in development. For Thompson seedless where cell vitality is maintained through development, the xylem tension can be resisted by the osmotic potential of the pericarp cell sap. In this case the hydraulic conductance to the vine could be kept high. This is supported by our measurements of xylem pressure which remains negative in Thompson seedless.
The question remains how the hydraulic conductance from berry to vine via the xylem is regulated. Tyerman et al. (2004) showed that the largest change in hydraulic conductance through development occurred within the distal part of the berry. The dye studies have shown that the xylem remain qualitatively connected (Bondada et al. 2005; Keller et al. 2006) late in development, but it is possible that the hydraulic conductance of vessels and tracheids is still reduced. Another possibility is that the cells surrounding the xylem vessels function as an effective barrier from the apoplast of the berry to the xylem lumens. Variable hydraulic conductivity of these cells could be via regulated activity of aquaporins. The overall semipermeability of this layer of cells, characterized by the reflection coefficient, would depend on the degree of development of apoplast barriers analogous to the composite transport model applied to roots (Tyerman et al. 2004). Our work has demonstrated that despite extensive cell death in the mesocarp and endocarp, the vascular tissue remains conspicuously vital, indicating that regulation at the membrane level in these cells is possible.
The processes of ripening and senescence are often inter-related (Jones 2004). During ripening, a variety of physical and biochemical mechanisms change the structure of cell walls in fleshy fruits with textural results ranging from crisp to melting or soft and deformable (Brummell 2006).The deterioration of plant membranes during senescence is well documented and several enzymes are involved (Paliyath and Droillard 1992). Research has focussed on the post harvest period, but interestingly, enzymes of the lipoxygenase family have been associated with membrane lipid peroxidation in ripening saskatoons (Rogiers et al. 1998) and volatile aroma or flavour development in tomatoes (Chen et al. 2004) and kiwifruit (Zhang et al. 2006a). It has been noted that aroma compounds in wine grapes increase late in ripening when sugar accumulation has slowed or stopped and non- anthocyanin glycosides rise sharply at around 100 DAA in Shiraz (Coombe and McCarthy 1997).
In this context, another important issue raised by our results is whether flavour development in Shiraz grapes can be attributed to the significant loss of cell integrity within the body of the pericarp and the ensuing degradation processes. According to this data, it is more than loss of compartmentation in the mesocarp; it is extensive lysing and mixing of cell contents. The trend for ‘hang time’ while the winemakers wait for ‘ripe flavours’ in this variety would also contribute to the loss of cell vitality by harvest. The highly organised pattern of cell death in the premium Shiraz fruit suggests that this idea should be examined.
In conclusion, the variety dependent vitality changes in the pericarp of grapevine berries late in maturity seems to be linked to the strategy of berry water balance in the particular variety. We hypothesise that there are two strategies: (1) cell death in the mesocarp and loss of osmotically competent membranes requires concomitant reduction in the hydraulic conductance of the pathway via the xylem back to the vine; (2) continued cell vitality and osmotically competent membranes that can allow continued hydraulic conductance to the vine. A miss-match in the timing of strategy 1 could cause substantial backflow to the vine and loss of berry weight. For strategy 2, there is a greater danger of berry splitting upon the vine attaining high water potentials. Our data also strongly suggests that closer examination of the cell death processes in wine grapes would contribute significantly to understanding the final phases of flavour development.
Acknowledgements
This research was supported by the Commonwealth Cooperative Research Centre Program and conducted through the CRC for Viticulture with support from Australia’s grape growers and winemakers through their investment body the Grape and Wine Research and Development Corporation, with matching funds from the Federal Government. Also, thanks to Professor Ken Shackel for sharing the FDA method developed in his laboratory at Davis, University of California.
Adams DO
(2006) Phenolics and ripening in grape berries. American Journal of Enology and Viticulture 57, 249–256.

Bernas T,
Zarebski M,
Cook RR, Dobrucki JW
(2004) Minimising photobleaching during confocal microscopy of fluorescent probes bound to chromatin: role of anoxia and photon flux. Journal of Microscopy 215, 281–296.
| Crossref | GoogleScholarGoogle Scholar | PubMed |

Bondada BR,
Matthews MA, Shackel KA
(2005) Functional xylem in the post-veraison grape berry. Journal of Experimental Botany 56, 2949–2957.
| Crossref | GoogleScholarGoogle Scholar | PubMed |

Brummell DA
(2006) Cell wall disassembly in ripening fruit. Functional Plant Biology 33, 103–119.
| Crossref | GoogleScholarGoogle Scholar |

Chen G,
Hackett R,
Walker D,
Taylor A,
Lin Z, Grierson D
(2004) Identification of a specific isoform of tomato lipoxygenase (TomloxC) involved in the generation of fatty acid-derived flavor compounds. Plant Physiology 136, 2641–2651.
| Crossref | GoogleScholarGoogle Scholar | PubMed |

Coombe BG
(1992) Research on development and ripening of the grape berry. American Journal of Enology and Viticulture 43, 101–110.

Coombe BG, McCarthy MG
(1997) Identification and naming of the inception of aroma development in ripening grape berries. Australian Journal of Grape and Wine Research 3, 18–20.
| Crossref | GoogleScholarGoogle Scholar |

Cosa G,
Focsaneanu KS,
McLean JRN,
McNamee JP, Scalano JC
(2001) Photophysical properties of fluorescent DNA-dyes bound to single-and double-stranded DNA in aqueous buffered solution. Photochemistry and Photobiology 73, 585–599.
| Crossref | GoogleScholarGoogle Scholar | PubMed |

Creasy G,
Price SF, Lombard PB
(1993) Evidence for xylem discontinuity in pinot noir and merlot grapes: dye uptake and mineral composition during berry maturation. American Journal of Enology and Viticulture 44, 187–192.

Dreier LP,
Hunter JJ, Ruffner HP
(1998) Invertase activity, grape berry development and cell compartmentation. Plant Physiology and Biochemistry 36, 865–872.
| Crossref | GoogleScholarGoogle Scholar |

Dreier LP,
Stoll GS, Ruffner HP
(2000) Berry ripening and evapotranspiration in Vitis vinifera L. American Journal of Enology and Viticulture 51, 340–346.

Findlay N,
Oliver KJ,
Nii N, Coombe BG
(1987) Solute accumulation by grape pericarp cells. Journal of Experimental Botany 38, 668–679.
| Crossref | GoogleScholarGoogle Scholar |

Greenspan MD,
Shackel KD, Matthews MA
(1994) Developmental changes in the diurnal water budget of the grape berry exposed to water deficits. Plant, Cell & Environment 17, 811–820.
| Crossref | GoogleScholarGoogle Scholar |

Greenspan MD,
Schultz HR, Matthews MA
(1996) Field evaluation of water transport in grape berries during water deficits. Physiologia Plantarum 97, 55–62.
| Crossref | GoogleScholarGoogle Scholar |

Hardie WJ, O’Brien TP
(1988) Considerations of the biological significance of some volatile constituents of grape (Vitis spp.). Australian Journal of Botany 36, 107–117.
| Crossref | GoogleScholarGoogle Scholar |

Hardie WJ,
O’Brien TP, Jaudzems VG
(1996) Morphology, anatomy and development of the pericarp after anthesis in grape, Vitis vinifera L. Australian Journal of Grape and Wine Research 2, 97–142.
| Crossref | GoogleScholarGoogle Scholar |

Jones KH, Senft JA
(1985) An improved method to determine cell viability by simultaneous staining with fluorescein diacetate-propidium iodide. Journal of Histochemistry and Cytochemistry 33, 77–79.
| PubMed |

Keller M,
Smith JP, Bondada BR
(2006) Ripening grape berries remain hydraulically connected to the shoot. Journal of Experimental Botany 57, 2577–2587.
| Crossref | GoogleScholarGoogle Scholar | PubMed |

Kral T,
Widerak K,
Langner M, Hof M
(2005) Propidium iodide and PicoGreen as dyes for the DNA fluorescence correlation spectroscopy measurements. Journal of Fluorescence 15, 179–183.
| Crossref | GoogleScholarGoogle Scholar | PubMed |

Lang A, Düring H
(1991) Partitioning control by water potential gradient:evidence for compartmentation breakdown in grape berries. Journal of Experimental Botany 42, 1117–1122.
| Crossref | GoogleScholarGoogle Scholar |

Lang A, Thorpe MR
(1989) Xylem, phloem and transpiration flows in a grape: application of a technique for measuring the volume of attached fruits to high resolution using Archimedes’ principle. Journal of Experimental Botany 40, 1069–1078.
| Crossref | GoogleScholarGoogle Scholar |

Loureiro J,
Rodriguez E,
Dolezel J, Santos C
(2006) Flow cytometric and microscopic analysis of the effect of tannic acid on plant nuclei and estimation of DNA content. Annals of Botany 98, 515–527.
| Crossref | GoogleScholarGoogle Scholar | PubMed |

McCarthy MG
(1999) Weight loss from ripening berries of shiraz grapevines (Vitis vinifera L. cv. Shiraz). Australian Journal of Grape and Wine Research 5, 10–16.
| Crossref | GoogleScholarGoogle Scholar |

McCarthy MG, Coombe BG
(1999) Is weight loss in ripening grape berries cv. Shiraz caused by impeded phloem transport? Australian Journal of Grape and Wine Research 5, 17–21.
| Crossref | GoogleScholarGoogle Scholar |

Noirot M,
Barre P,
Duperray C,
Louarn J, Hamon S
(2003) Effects of caffeine and chlorogenic acid on propidium iodide accessibilty to DNA: consequences on genome size evaluation in coffee tree. Annals of Botany 92, 259–264.
| Crossref | GoogleScholarGoogle Scholar | PubMed |

Ojeda H,
Deloire A,
Carbonneau A,
Ageorges A, Romieu C
(1999) Berry development of grapevines: relations between the growth of berries and their DNA content indicate cell multiplication and enlargement. Vitis 38, 145–150.

Paliyath G, Droillard MJ
(1992) The mechanisms of membrane deterioration and disassembly during senescence. Plant Physiology and Biochemistry 30, 789–812.

Patrick JW
(1997) Phloem unloading: sieve element unloading and post-seive element transport. Annual Review of Plant Physiology and Plant Molecular Biology 48, 191–222.
| Crossref | GoogleScholarGoogle Scholar | PubMed |

Pratt C
(1971) Reproductive anatomy in cultivated grapes – a review. American Journal of Enology and Viticulture 23, 93–109.

Price HJ,
Hodnett G, Johnston JS
(2000) Sunflower (Helianthus annus) leaves contain compounds that reduce nuclear propidium iodide fluorescence. Annals of Botany 86, 929–934.
| Crossref | GoogleScholarGoogle Scholar |

Rogiers SY,
Mohan Kumar GN, Knowles NR
(1998) Maturation and ripening of fruit of Amelanchier alnifolia Nutt. are accompanied by increasing oxidative stress. Annals of Botany 81, 203–211.
| Crossref | GoogleScholarGoogle Scholar |

Rogiers S,
Smith JA,
White R,
Keller M,
Holzapfel BP, Virgona JM
(2001) Vascular function in berries of Vitis vinifera (L) cv. Shiraz. Australian Journal of Grape and Wine Research 7, 47–51.
| Crossref | GoogleScholarGoogle Scholar |

Rogiers S,
Hatfield J,
Gunta Jaudzems V,
White R, Keller M
(2004) Grape berry cv. Shiraz epicuticular wax and transpiration during ripening and preharvest weight loss. American Journal of Enology and Viticulture 55, 121–127.

Rogiers S,
Greer D,
Hatfield J,
Orchard B, Keller M
(2006) Solute transport into Shiraz berries during development and late-ripening shrinkage. American Journal of Enology and Viticulture 57, 73–80.

Sadras V, McCarthy MG
(2007) Quantifying the dynamics of sugar concentration in berries of Vitis vinifera cv. Shiraz: a novel approach based on allometric analysis. Australian Journal of Grape and Wine Research 13, 66–71.
| Crossref | GoogleScholarGoogle Scholar |

Thomas H,
Ougham HJ,
Wagstaff C, Stead AD
(2003) Defining senescence and death. Journal of Experimental Botany 54, 1127–1132.
| Crossref | GoogleScholarGoogle Scholar | PubMed |

Thomas TR,
Matthews MA, Shackel KA
(2006) Direct in situ measurement of cell turgor in grape (Vitis vinifera L.) berries during development and in response to plant water deficits. Plant, Cell & Environment 29, 993–1001.
| Crossref | GoogleScholarGoogle Scholar | PubMed |

Tyerman SD,
Tilbrook J,
Pardo C,
Kotula L,
Sullivan W, Steudle E
(2004) Direct measurement of hydraulic properties in developing berries of Vitis vinifera L. cv Shiraz and Chardonnay. Australian Journal of Grape and Wine Research 10, 170–181.

Zhang D-P,
Li M, Wang Y
(1997) Ultrastructural changes in the mesocarp cells of grape berry during its development. Acta Botanica Sinica 39, 389–396.

Zhang B,
Chen K,
Bowen J,
Allan A,
Espley R, Karunairetnam S
(2006a) Differential expression within the LOX gene family in ripening kiwifruit. Journal of Experimental Botany 57, 3825–3836.
| Crossref | GoogleScholarGoogle Scholar | PubMed |

Zhang X-Y,
Wang X-L,
Wang X,
Xia G-H,
Pan Q-H,
Fan R-C,
Wu F-Q,
Yu X-C, Zhang D-P
(2006b) A shift of phloem unloading from symplasmic to apoplasmic pathway is involved in developmental onset of ripening in grape berry. Plant Physiology 142, 220–232.
| Crossref | GoogleScholarGoogle Scholar | PubMed |



