Aestivation dynamics of bogong moths (Agrotis infusa) in the Australian Alps and predation by wild pigs (Sus scrofa)
Peter Caley A C and Marijke Welvaert A BA Commonwealth Scientific and Industrial Research Organisation, GPO Box 1700, Canberra, ACT 2601, Australia.
B Present Address: University of Canberra, Research Institute for Sport and Exercise, Canberra, ACT 2601, Australia.
C Corresponding author: Email: peter.caley@csiro.au
Pacific Conservation Biology 24(2) 178-182 https://doi.org/10.1071/PC18007
Submitted: 18 January 2018 Accepted: 9 March 2018 Published: 16 April 2018
Journal compilation © CSIRO 2018 Open Access CC BY-NC-ND
Abstract
We document predation of aestivating bogong moths (Agrotis infusa) by wild pigs (Sus scrofa) at a location in the Australian Alps. This is the first known record of pigs preying on bogong moths. Wild pigs are recent colonisers of the region, though already the population appears seasonally habituated to foraging on aestivating moths. This is indicative of adaptation of a feral animal undertaking dietary resource switching within what is now a modified ecosystem and food web. The significance of this predation on moth abundance is unclear. Long-term monitoring to compare numbers of moths with historical surveys undertaken before the colonisation by wild pigs will require that they are excluded from aestivation sites. Our surveys in 2014–15 observed bogong moths to arrive about one month earlier compared with a similar survey in 1951–52, though to also depart earlier.
Additional keywords: climate change, migration, monitoring.
Introduction
The annual migration of bogong moths (Agrotis infusa) from the inland plains to the alpine regions of south-eastern Australia is a remarkable natural phenomenon, with many similarities to the monarch butterfly (Danaus plexippuss) migrations in North America (Warrant et al. 2016). In behaviours first described in detail by Common (1954), the spring generation of moths migrate from the inland plains to the mountains, where they aestivate gregariously over summer in crevices and caverns at or close to the top of the mountain summits. During late summer and autumn, the moths return to the inland breeding grounds, when dicotyledonous annuals suitable as a larval foodsource have germinated (Common 1954). The reliance of bogong moths on the highest parts of the Australian High Country for summer aestivation suggests that a warming climate may impact on this species (Green 2010), with knock-on effects to species that are heavily dependent on the moths as a source of food, such as the mountain pigmy possum (Burrayms parvus) (Broome 2001). The abundance of bogong moths could also be strongly influenced by land-use changes in the other parts of their range in inland Australia, climate-driven or otherwise.
Aboriginal people incorporated the annual migration of bogong moths to Australia‘s high country into their yearly foraging activity and culture (Flood 1996). More recently, the spring arrival of the moths has become a feature of the cultural calendar amongst the current predominately non-Aboriginal community. Indeed, the spring arrival of the bogong moths is detectable on social media (Welvaert et al. 2017).
Invasive animals quickly adapt to exploiting local food sources, and are often dietary generalists. For example, during summer months red foxes (Vulpes vulpes) inhabiting areas above 1500 m altitude in the Australian High Country extensively feed on bogong moths and grasshoppers (Green and Osborne 1981). Such predation on bogong moth populations has probably been occurring since the early 1900s when foxes first colonised the region (Short 1998). The colonisation of Australia’s High Country by wild pigs (Sus scrofa) is a much more recent event. It appears that wild pigs were first introduced to the northern section of the Australian Alps in 1959 as a result of escapees from a vehicle accident (Hone 2002). They have subsequently colonised virtually all of the region (Saunders and Kay 1996; Hone 2002). The wild pig is a particularly adaptable, highly invasive, vertebrate species that is well known for exploiting seasonally available high-value food sources, whether in its native (e.g. Schley and Roper 2003) or introduced ranges (e.g. Caley 1997).
There are little published data on long-term trends in the number of bogong moths aestivating in the Australian High Country. Indeed, when recently describing the aestivation behaviour of bogong moths in the Australian High Country, Warrant et al. (2016) relied heavily on the work of Common (1954), who intensively surveyed the coverage of moths on a single rock face within an overhanging shelter near the summit of Mount Gingera in 1951–52 and 1952–53. There is a need to develop an approach for monitoring numbers of bogong moths in the Australian High Country, if long-term changes in moth numbers are to be detectable.
In this study we revisit the cave surveyed by Common (1954) as part of a broader survey of dynamics of bogong moths aestivating on Mount Gingera, Brindabella Ranges, Australia. In doing so we provide alternative explanations for temporal variation in the abundance of aestivating moths, and note the predation of bogong moths by wild pigs in the cave described by Common (1954).
Materials and methods
Study area
The study focused on the flat-topped summit on Mount Gingera (1857 m above sea level; 35°34′S, 148°47′E) within the Brindabella Ranges bordering the Australian Capital Territory and New South Wales, in south-eastern Australia. Scattered granite outcrops occur along the length of the summit, and on the surrounding slopes. Vegetation is a mix of woodland dominated by snowgum (Eucalyptus pauciflora) and subalpine grassland communities. Snow cover typically persists on the shaded side of the summit from July to October. Weather data are available from an Australian Government Bureau of Meterology station (site name: Mount Ginini AWS; site no: 070349) on the neighbouring Mount Ginini at 1760 m above sea level. For the period 2004–17, the coldest month was July (mean minimum = −2.4°C, mean maximum = 2.4°C), and the hottest month was January (mean minimum = 9.9°C, mean maximum = 20.8°C). It would be expected, based on the adiabatic lapse rate, for Mount Gingera to experience temperatures ~0.6°C colder than these. The annual precipitation averaged 1053 mm, with rainfall spread reasonably evenly throughout the year apart from autumn, which is drier.
Aestivating moth survey
Prior to any moth arrival and snow cover, 37 potential aestivating sites associated with granite boulders and boulder piles were identified along a north–south transect along the summit ridge of Mount Gingera. Sites were chosen either on the basis of evidence of previous moth use (e.g. moth detritus being visible), or rock topography (crevices, overhangs, caves) similar to those sites with previous evidence of use. The rationale for the latter was that in some sites evidence of previous occupation is removed by natural factors (wind, water, etc.).
During each survey all crevices were searched with a torch, and the volume of moths estimated with the aid of a tape measure. A depth of 2 cm was used for moths covering faces. Searches and scoring were undertaken by the same observer (PC) in both years.
Wild pig monitoring
Whilst undertaking moth aestivation surveys during late 2014 there was considerable sign (tracks, faeces) of wild pigs inside and around the chief observation cave (hereafter ‘Moth Cave’) studied by Common (1954). There was also considerable sign of pigs foraging as far as they could reach under granite boulder piles hosting aestivating bogong moths. Wild pigs were not present on Mount Gingera in the early 1950s at the time of the pioneering surveys of Common (1954).
To monitor use of the Moth Cave by wild pigs, and other wildlife, during 2015 we installed a camera-trap (Reconyx HC500 Hyperfire Trail) to monitor the entrance to the cave for the period 17 December 2015 until 9 January 2016. The camera uses passive infrared motion detection, and to be triggered requires an object different from the background temperature to enter one of the detection bands. The camera was located ~1 m from the cave entrance and programmed to take a burst of 10 images upon each triggering (with no ‘stand-down’ period). Subsequently, the camera was stolen from a second placement inside the cave. The lack of a ‘stand down’ period created problems with the volume of images captured (>33 000), as the pigs spent of lot of time moving in and out of the cave entrance. To facilitate processing, bursts of images were thinned to a minimum time interval of 2 min for species identification and analysis. This resulted in 416 camera trigger events over a c. 23-day period, of which 372 trigger events involved a visible animal of sorts (including moths).
Results
General
Moths first arrived in 2014 between 4 (absent) and 9 (present) October. For 2015 the window of arrival was 29 September to 6 October. On both occasions, the first site housing moths was at the south-western side of the summit boulder pile. The distance separating the initial clumps between the two years was less than 1 m, which seemed remarkable.
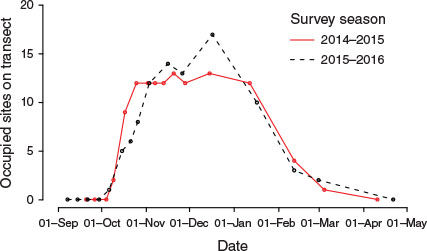
Most of the aestivation sites were in narrow cracks (<10 cm) (e.g. Fig. 1). Of the 37 potential sites identified prior to the survey, a maximum of 35% were occupied in 2014–15 during mid-November and mid-December, and 46% were occupied in 2015–16 in mid-December (Fig. 2).

|
|
Seasonality
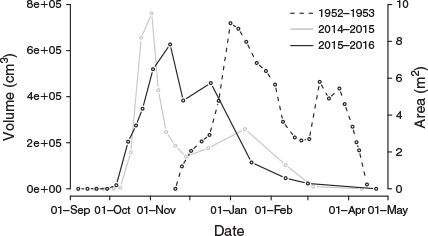
Compared with the 1951–52 and 1952–53 seasons observed by Common (1954), the moths arrived a month earlier and departed a month early (Fig. 3). The intraseasonal variation in abundance was broadly similar, with a two-peaked pattern that was evident in both the number of occupied sites and the volume of moths.
|
In both years nearly all aestivating moths had departed by early March, with all departed by the end of April (Fig. 3), a month earlier than observed by Common (1954).
Notably, over the aestivation period, the number of moths at some sites changed suddenly.
Predation
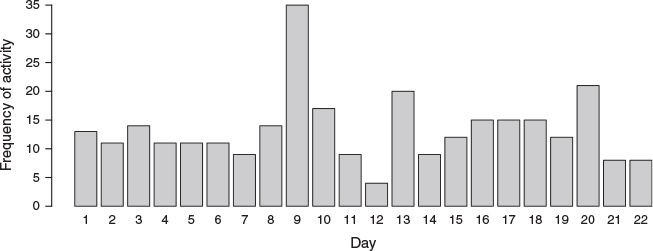
Pigs were observed (based on their tracks) to regularly visit the Moth Cave before the arrival of moths. After the arrival of the moths, wild pigs used the moth cave and its immediate surroundings extensively, especially a small group of pregnant sows (Fig. 4) and lactating sows and their piglets (Fig. 5). They were recorded visiting the cave every day for the 24 days that the camera was operating, with no obvious trend in visitation rate over time (Fig. 6). This always included multiple visits on the same day (mean = 13 visits day−1, range = 4–35 visits day−1, n = 22 full calendar days) (Fig. 6). Interestingly, few, if any, boars were caught on camera, although they were seen in the vicinity at other times. Clearly, the bogong moth resource was being heavily utilised by sows and their offspring.

|

|
|
At no time were there ever any moths on the roof of the moth cave, as had been observed by Common (1954). In fact, there were no moths present on any rock surface within the shelter that was accessible to a pig snout. In contrast, the inaccessible (to pigs) corners of the cave were completely covered in moths during both years.
Additional species observed at the cave entrance include the pied currawong (Strepera graculina), red fox (one trigger event), feral cat (Felis catus) (one trigger event) and an unidentified (image resolution too poor) bat (eight trigger events).
Discussion
This study has identified further ecological pressures on the bogong moth population, and documented an approach for monitoring changes in an index of abundance on Mount Gingera. Being one of the northernmost, and hence climatically marginal, mountain-top aestivation sites for this species, Mount Gingera is a key site for long-term monitoring. We note that within the summer aestivation period, movement of moths is not fully understood, and this affects the interpretation of data. For example, Common (1954) inferred that the two-peaked pattern of abundance in the moth cave could have been caused by moths using Mount Gingera as a staging post for travelling further south to the slightly higher peaks of the Main Range. Common (1954) also observed the mid-summer decline to coincide with moths being observed flying towards the north, though presumably not migrating. The extent of between-site movement over the aestivation period that we have documented provides an alternative explanation. It is possible that moths are moving deeper into cracks that are not observable, though whether such sites exist is difficult to tell. There were, however, several deep, narrow and shaded aestivation sites that were observable for which moth aggregations disappeared early in the period of aestivation. We expected such sites to remain inhabited if moths were shifting deeper for reasons of thermoregulation. The intraseasonal movements and their reasons remain unclear.
Our study is the first known record of pigs preying on bogong moths. Wild pigs are recent colonisers of the region, though already the population appears seasonally habituated to foraging on aestivating moths. It has previously been reported that wild pigs utilised higher-altitude regions of Namadgi National Park and neighbouring Kosciusko National Park during the summer months (John McIlroy, unpubl. data) (Saunders 1988). During April 2017, a visit to the summit of another prominent peak in the southern Brindabella Ranges, Mount Gudgenby (~1740 m above sea level; 35°46′S, 148°55′E) revealed extensive signs of pigs, indicative of prolonged use over the previous summer. This strongly suggests that the pigs were utilising bogong moth resources atop Mount Gudgenby in a similar manner to what we observed on Mount Gingera. Our study shows that for at least a portion of this pig population in the Australian High Country, aestivating bogong moths are one of the food sources driving this seasonal shift in home-range use. This is indicative of adaptation of a feral animal undertaking dietary resource switching within what is now a modified ecosystem and food web. The parallels in resource utilisation with the original indigenous inhabitants of the region are striking (see Flood 1996). Currently, most sites (rock faces) with aestivating bogong moths are inaccessible to wild pigs, although there is possible circularity in this inference. Hence the impact of the pigs on moth abundance is unclear.
The frequency of recorded visits by red foxes (only a single trigger event in nearly a month) was less than we expected based on the high frequency of bogong moths in the summer diet of foxes (Green and Osborne 1981). A possible explanation for this is that the foxes may have been avoiding interactions with the wild pigs that had taken up residence. The red fox is capable of preying on piglets, hence their presence may not be tolerated by sows. We do not have the data to test this idea quantitatively.
The bogong moth has been described as a keystone species of Australian alpine habitats in recognition of its role in transporting energy and nutrients (Green 2011). Being reliant on the highest points in the Australian Alps for summer aestivation, bogong moths are clearly a species at risk of suffering under a warming climate (Green 2010), with possible cascading effects to species such as the mountain pigmy possum whose summer diet is heavily reliant on aestivating moths (Broome 2001). Although we have noted that the single site used by Common (1954) is probably insufficient on its own to monitor year-to-year trends in moth abundance, it would be straight-forward to prevent pigs, but not moths from entering (e.g. using pig-proof mesh). This would enable legitimate comparison of future data collected within the moth cave with those of Common (1954).
It certainly appears that during the years 2014 and 2015, moths arrived about a month earlier atop Mount Gingera than observed during 1951–52 and 1952–53; this would make sense in a warming climate with a shorter period of snow cover, though clearly the strength of this inference is limited by the number of samples. In contrast, Green (2010) observed that bogong moths were not responding to earlier snow melt, at least at altitudes 1800–2228 m around Mount Kosciuszko, on the Main Range ~150 km south-south-west of Mount Gingera. The same logic, however, doesn‘t explain why it appears that the moths on Mount Gingera are departing earlier – in fact, it would predict the reverse.
The bogong moth population faces challenges across its range, and long-term monitoring would appear prudent. The aestivation sites atop Mount Gingera would appear to be at the lower end of the preferred thermal conditions (and, by analogy, altitude) for the moths, and hence be a key monitoring location for detecting changes in response to a warming climate.
Conflicts of interest
None to report.
Acknowledgements
Staff from the Australian Capital Territory Parks and Conservation Service assisted with access to the study site. Data collection was facilitated by the Australian Government's Cooperative Research Centres Program (Plant Biosecurity CRC Project 1029). Funding for analysis and write-up was provided by the CSIRO. Sam Nelson assisted with analysing camera trap data. Natasha Herron and Jessica Caley assisted with data collection. Sophie De Meyer kindly gave permission for use of the photo in Figure 1. Comments from Matt Colloff and Warren Muller improved an earlier version of the manuscript.
References
Broome, L. S. (2001). Density, home range, seasonal movements and habitat use of the mountain pygmy-possum Burramys parvus (Marsupialia: Burramyidae) at Mount Blue Cow, Kosciuszko National Park. Austral Ecology 26, 275–292.| Density, home range, seasonal movements and habitat use of the mountain pygmy-possum Burramys parvus (Marsupialia: Burramyidae) at Mount Blue Cow, Kosciuszko National Park.Crossref | GoogleScholarGoogle Scholar |
Caley, P. (1997). Movements, activity patterns and habitat use of feral pigs (Sus scrofa) in a tropical habitat. Wildlife Research 24, 77–87.
| Movements, activity patterns and habitat use of feral pigs (Sus scrofa) in a tropical habitat.Crossref | GoogleScholarGoogle Scholar |
Common, I. F. B. (1954). A study of the ecology of the adult bogong moth, Agrotis infusa (Boisd) (Lepidoptera: Noctuidae), with special reference to its behaviour during migration and aestivation. Australian Journal of Zoology 2, 223–263.
| A study of the ecology of the adult bogong moth, Agrotis infusa (Boisd) (Lepidoptera: Noctuidae), with special reference to its behaviour during migration and aestivation.Crossref | GoogleScholarGoogle Scholar |
Flood, J. (1996). ‘Moth Hunters of the Australian Capital Territory.’ (Published privately: Canberra.)
Green, K. (2010). Alpine taxa exhibit differing responses to climate warming in the Snowy Mountains of Australia. Journal of Mountain Science 7, 167–175.
| Alpine taxa exhibit differing responses to climate warming in the Snowy Mountains of Australia.Crossref | GoogleScholarGoogle Scholar |
Green, K. (2011). The transport of nutrients and energy into the Australian Snowy Mountains by migrating bogong moths Agrotis infusa. Austral Ecology 36, 25–34.
| The transport of nutrients and energy into the Australian Snowy Mountains by migrating bogong moths Agrotis infusa.Crossref | GoogleScholarGoogle Scholar |
Green, K., and Osborne, W. (1981). The diet of foxes, Vulpes vulpes (L.), in relation to abundance of prey above the winter snowline in New South Wales. Wildlife Research 8, 349–360.
| The diet of foxes, Vulpes vulpes (L.), in relation to abundance of prey above the winter snowline in New South Wales.Crossref | GoogleScholarGoogle Scholar |
Hone, J. (2002). Feral pigs in Namadgi National Park, Australia: dynamics, impacts and management. Biological Conservation 105, 231–242.
| Feral pigs in Namadgi National Park, Australia: dynamics, impacts and management.Crossref | GoogleScholarGoogle Scholar |
Saunders, G. R. (1988). The ecology and management of feral pigs in New South Wales. M.Sc. Thesis, Macquarie University, Sydney.
Saunders, G., and Kay, B. (1996). Movements and home ranges of feral pigs (Sus scrofa) in Kosciusko National Park, New South Wales. Wildlife Research 23, 711–719.
| Movements and home ranges of feral pigs (Sus scrofa) in Kosciusko National Park, New South Wales.Crossref | GoogleScholarGoogle Scholar |
Schley, L., and Roper, T. J. (2003). Diet of wild boar Sus scrofa in western Europe, with particular reference to consumption of agricultural crops. Mammal Review 33, 43–56.
| Diet of wild boar Sus scrofa in western Europe, with particular reference to consumption of agricultural crops.Crossref | GoogleScholarGoogle Scholar |
Short, J. (1998). The extinction of rat-kangaroos (Marsupialia: Potoroidae) in New South Wales, Australia. Biological Conservation 86, 365–377.
| The extinction of rat-kangaroos (Marsupialia: Potoroidae) in New South Wales, Australia.Crossref | GoogleScholarGoogle Scholar |
Warrant, E., Frost, B., Green, K., Mouritsen, H., Dreyer, D., Adden, A., Brauburger, K., and Heinze, S. (2016). The Australian Bogong moth Agrotis infusa: a long-distance nocturnal navigator. Frontiers in Behavioral Neuroscience 10, .
| The Australian Bogong moth Agrotis infusa: a long-distance nocturnal navigator.Crossref | GoogleScholarGoogle Scholar |
Welvaert, M., Al-Ghattas, O., Cameron, M., and Caley, P. (2017). Limits of use of social media for monitoring biosecurity events. PLoS One 12, e0172457.
| Limits of use of social media for monitoring biosecurity events.Crossref | GoogleScholarGoogle Scholar |


