Transcriptional and epigenetic control of cell fate decisions in early embryos
Ramiro AlberioSchool of Biosciences, University of Nottingham, Nottingham, LE12 5RD, UK. Email: ramiro.alberio@nottingham.ac.uk
Reproduction, Fertility and Development 30(1) 73-84 https://doi.org/10.1071/RD17403
Published: 4 December 2017
Journal Compilation © IETS 2018 Open Access CC BY
Abstract
Mammalian embryo development is characterised by regulative mechanisms of lineage segregation and cell specification. A combination of carefully orchestrated gene expression networks, signalling pathways and epigenetic marks defines specific developmental stages that can now be resolved at the single-cell level. These new ways to depict developmental processes have the potential to provide answers to unresolved questions on how lineage allocation and cell fate decisions are made during embryogenesis. Over the past few years, a flurry of studies reporting detailed single-cell transcription profiles in early embryos has complemented observations acquired using live cell imaging following gene editing techniques to manipulate specific genes. The adoption of this newly available toolkit is reshaping how researchers are designing experiments and how they view animal development. This review presents an overview of the current knowledge on lineage segregation and cell specification in mammals, and discusses some of the outstanding questions that current technological advances can help scientists address, like never before.
Additional keywords: epiblast, gastrulation, lineage segregation, primordial germ cells, stem cells.
Regulative development in the early embryo
Mammalian embryo development follows a regulatory series of lineage segregation events that culminate with the specification of more than 200 different cell types that form the adult animal. How the embryo transits through these successive developmental stages and what mechanisms determine how cell fate decisions are made is of central importance for mammalian developmental biology and is pivotal to innovation in biotechnology and regenerative medicine.
Development from the zygote to the blastocyst stage is morphologically similar across mammalian species, although detailed molecular analysis has revealed important differences in gene expression and epigenetic regulation. The 1-cell zygote, a transcriptionally silent cell, initiates reductionist divisions that are initially controlled by maternally inherited RNAs and proteins. During this initial period there is no evidence that the early blastomeres are different from each other (i.e. they can all give rise to all the cells of the blastocyst; Motosugi et al. 2005). At the 2-cell (rodents) and 4- to 8-cell stage (other mammals), major embryonic genome activation (EGA) takes place, which is critical for the culmination of cleavage divisions (Evsikov et al. 2006; Yan et al. 2013; Cao et al. 2014). In the mouse, the 8-cell stage marks the first morphological reorganisation of the blastomeres, evidenced by the process of compaction, which become polarised with an apical surface containing microvilli and separated from a basolateral membrane by adherens and tight junctions (Johnson and Ziomek 1983; Korotkevich et al. 2017). The apical domain of blastomeres confers differential contractility, and cells with reduced contractility contribute to the inner cell mass (ICM), suggesting mechanical sensing is important during lineage segregation (Maître et al. 2016). Two additional rounds of cleavage divisions result in more cells contributing to the outside and inside compartments before the onset of cavitation at around the 32-cell stage. In other mammals, the appearance of microvilli has been reported at the 16-cell (human, porcine, bovine) or 32-cell (rabbit) stage (Reima et al. 1993; Koyama et al. 1994; Nikas et al. 1996). Similarly, compaction does not start until the 16-cell stage in humans (Steptoe et al. 1971) and 32-cell stage in bovine (Van Soom et al. 1997) and porcine (Reima et al. 1993) embryos. The outside and inside cells have their fates determined during the process of cavitation, wherein a fluid-filled cavity forms as the prelude to the segregation of the trophectoderm (TE) and the ICM, respectively.
During early cleavage divisions, the mammalian embryo is highly adaptable to the removal or addition of blastomeres, although in mice epigenetic differences become apparent in blastomeres of 4-cell embryos, which influence how the pluripotency factors octamer-binding transcription factor 4 (Oct4) and SRY (sex determining region Y)-box 2 (Sox2) contribute to the commitment of ICM cells during the first lineage segregation (Goolam et al. 2016; White et al. 2016). The highly heterogeneous gene expression (see later) and the plastic properties of the preimplantation embryo that enable adaptation to the removal of blastomeres provide additional evidence for the highly regulative nature of early mammalian development.
Totipotent blastomeres segregate into embryonic and extraembryonic precursors
A unique property of mammalian embryos is the segregation of extra-embryonic lineages concurrent with the emergence and expansion of the pluripotent epiblast. This totipotent property is the exclusive feature of cleaving (2- to 4-cell embryos) blastomeres, and much emphasis has been devoted to understanding how these cells are regulated at the transcriptional and epigenetic levels. Totipotent blastomeres have an open chromatin configuration that includes primarily heterochromatic regions, consistent with a stage-specific transcriptional profile (Liu et al. 2004; Ahmed et al. 2010). This unique chromatin configuration may be needed for the extensive reprogramming of epigenetic marks of germline chromatin before ‘zygotic’ modifications are re-established. Indeed, epigenetic modifiers are highly expressed in early cleavage stage embryos only to decrease by the blastocyst stage, reflecting a high degree of chromatin plasticity before lineage segregation (Burton et al. 2013). Among the many unique transcriptional features concomitant with epigenetic reprogramming is the activation of 2-cell-specific genes and transposable elements (TrE; Peaston et al. 2004). TrE sequences exhibit an open chromatin configuration, and because some of these (e.g. Short Interspersed Nuclear Elements (SINE) and endogenous retroviruses subfamily (ERVLs)) are enriched at transcriptional start sites (TSS) of 2-cell-specific genes, it was suggested that they may play a role during EGA (Wu et al. 2016). In contrast, long interspersed nuclear element (LINE1) elements, another type of TrE more widely distributed throughout the genome and highly expressed in early embryos, seem to play a role in regulating global transcription factor accessibility by mediating de- and recondensation of chromatin as an integral part of embryo development (Jachowicz et al. 2017). Furthermore, the open chromatin configuration leading to murine endogenous retrovirus with leucine tRNA (MERV-L) expression is regulated by histone-modifying enzymes (Macfarlan et al. 2012), and is consistent with the broad enrichment for trimethylation (me3) of lysine 4 of histone H3 (H3K4) at large genomic loci detected during EGA (Dahl et al. 2016; Liu et al. 2016; Zhang et al. 2016). Capturing this totipotent cell state in culture has been a challenge and almost a serendipitous observation; however, mouse embryonic stem cells (ESCs) with features of the ‘2-cell (2C)-like’ state do appear under regular culture conditions and show totipotent features (Macfarlan et al. 2012). These cells can also be induced in vitro by depletion of the chromatin assembly complex, chromatin assembly complex 1 (CAF1), and they acquire chromatin configuration equivalent to early cleavage embryos (Ishiuchi et al. 2015), indicating that an increase in chromatin compaction is indicative of restricted developmental potential. In addition to the changes in nuclear configuration and gene expression, extensive epigenetic reprogramming characterised by DNA demethylation, histone replacement (H3.3 with H3.1/2) and histone demethylation (H3K64 me3, K3K9 me3 and H4K20 me3) are detected in early cleavage stage blastomeres, further demonstrating the unique epigenetic features that characterise this period of development.
First lineage segregation
The gradual polarisation of blastomeres from the 8-cell stage (in mice) to the onset of cavitation marks the first lineage segregation and is correlated with the expression of specific transcription factors that play key roles during specification. Cdx2, activated by Tead4 in the outer cells as a result of an inactive Hippo signalling pathway, begins to express ubiquitously at the 8-cell stage and becomes restricted to the outer cells at the 16- to 32-cell stage (Dietrich and Hiiragi 2007). Together with additional factors such as Gata3 and Eomes they participate in the gradual establishment of the mouse TE lineage (Ralston and Rossant 2008; Nishioka et al. 2009; Pfeffer and Pearton 2012). As demonstrated by experimental manipulations many decades ago, the specification of the TE lineage is gradual, because outer cells are not lineage restricted until the 32-cell stage in the mouse (Tarkowski and Wroblewska 1967; Pedersen et al. 1986; Suwińska et al. 2008; Szczepanska et al. 2011). The combined activation of key regulators of the TE lineage is accompanied by epigenetic restriction regulated by the activation of genes like Elf5, which is controlled by DNA methylation in the mouse TE (Ng et al. 2008). Elf5 acts as a lineage gatekeeper by preventing the precocious differentiation of trophoblast stem cells (Pearton et al. 2014). Thus, the transition from totipotency towards TE provides a good example of ‘canalisation’, whereby cells are gradually fated to a specific lineage through activation of a master regulator (Cdx2) that activates downstream effectors (Gata3, Eomes, Elf5), which, in turn, establish a feedback loop to maintain and propagate this lineage. The establishment of gene regulatory networks (GRN) with positive feedback loops that directly influence cell fate determination and regulate development has been described in other systems and is a recurrent feature of developmental processes (Peter and Davidson 2017). Interestingly, TE segregation in other mammals does not follow the same gene expression pattern as in mice, reflecting the differences in TE physiology. For example, TE cells in the human, bovine and porcine express OCT4 at the blastocyst stage (Kirchhof et al. 2000; Cauffman et al. 2005). In the bovine, blastocyst-derived TE cells are able to contribute to the hypoblast lineage (Berg et al. 2011). Similarly, in humans, blastocyst-derived TE cells are able to reconstitute a blastocyst (De Paepe et al. 2013), suggesting that although early TE cells are fated to become trophectoderm, they do not become lineage restricted until later in development. This observation is consistent with the recent reports indicating that TE, epiblast and primitive endoderm segregate concurrently in the human embryo (Petropoulos et al. 2016).
CDX2 starts to be expressed at the blastocyst stage in the human, bovine and porcine (Kuijk et al. 2008; Niakan and Eggan 2013), although RNA sequencing (RNA-Seq) from single cells and bulk transcriptomes demonstrates that expression is significantly lower than in mice (Berg et al. 2011; Cao et al. 2014; Blakeley et al. 2015). Consistent with these findings, CDX2 protein is detected in a subset of TE cells of early porcine blastocysts (Liu et al. 2015; Bou et al. 2017) and becomes confined to all TE cells in expanded blastocysts (Kuijk et al. 2008; Bou et al. 2017). Thus, CDX2 in porcine embryos does not play a critical role during TE segregation, but is essential for polarity and maintenance of the TE lineage (Bou et al. 2017), similar to observations in the bovine (Berg et al. 2011). Indeed, CDX2 was shown to repress OCT4 expression in the TE (Bou et al. 2016), therefore contributing to lineage restriction of the TE. EOMES and ELF5 in the porcine (Cao et al. 2014; Valdez Magaña et al. 2014) and bovine (Pearton et al. 2011) TE do not follow the expression profile described in mice, suggesting that the TE GRN has evolved differently in species with different placentas.
Instructive program of ICM segregation into hypoblast and epiblast
Concurrent with the segregation of the TE lineage, the ICM forms in the inside of the blastocyst. This compact mass of cells expresses the pluripotency genes Oct4 and Sox2, which are also expressed throughout cleavage stages in mice. In contrast, Nanog is first expressed at the 8-cell stage and becomes restricted to the ICM in the mouse (Guo et al. 2010). Gata6, the first marker of primitive endoderm (PE), also starts to be expressed in all cells at the 8-cell stage; however, by the 32-cell stage (Embryonic Day (E) 3.5), it resolves into a mutually exclusive pattern with Nanog, known as a ‘salt and pepper’ pattern, marking the hypoblast and epiblast lineages respectively (Chazaud et al. 2006; Plusa et al. 2008). These cells will segregate to their definitive positions by processes involving cell induction, active migration and apoptosis (Plusa et al. 2008; Meilhac et al. 2009). By E4.5, epiblast cells expressing Nanog are segregated from the hypoblast layer expressing Gata6, and other markers such as Pdgfrα, Sox17, and Gata4 (Plusa et al. 2008; Guo et al. 2010; Artus et al. 2011). In contrast with these observations in mice, NANOG protein in human, bovine and porcine embryos is first detected in late blastocysts (Kuijk et al. 2008; Cauffman et al. 2009; Khan et al. 2012). In human, bovine and porcine embryos, GATA6 is expressed in cleavage stages and all cells of the blastocyst, therefore it is not a suitable markers for PE segregation (Kimber et al. 2008; Roode et al. 2012; Cao et al. 2014; Wei et al. 2017). Thus, if the equivalent NANOG and GATA6 antagonistic relationship exists during epiblast and PE segregation in non-rodent mammals, it must be controlled by NANOG; however, this has not been determined. Instead, SOX17 and GATA4 have been found more reliable markers of this lineage in the human, bovine and porcine (Rodríguez et al. 2012; Roode et al. 2012; Niakan and Eggan 2013; McLean et al. 2014).
The competence for PE segregation is dependent on Nanog expression in mouse, because Nanog−/− ESCs fail to differentiate into Gata4-expressing cells, suggesting a paracrine mechanism of PE cell induction (Messerschmidt and Kemler 2010). A key candidate signalling pathway first investigated was the fibroblast growth factor (FGF)/mitogen-activated protein kinase (MAPK)/GRB2 pathway because mouse mutants for these molecules failed to develop a PE (Feldman et al. 1995; Arman et al. 1998; Cheng et al. 1998). Indeed, pharmacological inhibition of the FGF/MAPK pathway using small molecules prevents the differentiation of the PE, whereas supplementation of culture media with FGF led to a shift of all ICM cells towards the PE lineage (Nichols et al. 2009; Yamanaka et al. 2010). Thus, FGF4 acts as an instructing paracrine signal that determines the segregation of PE cells in the mouse. The process by which this is modulated is dictated by gradual production of FGF4 by early ICM cells internalised during cavitation of the embryo. FGF4 is then taken up during a subsequent cell division by cells containing higher levels of FGF receptor (FGFR) 2 on their cell surface, and therefore able to respond to the ligand (Morris et al. 2013). This proposed mechanism of epiblast and PE segregation suggests a bias of cells contributing to the ICM to become PE. However, the role of FGF4 during mouse ICM segregation has recently been extended, showing that it promotes expansion and maturation of the epiblast primarily through its interaction with FGFR1 (Kang et al. 2017; Molotkov et al. 2017). A secondary role of FGF4 is to induce the specification of PE, which is dependent on two receptors of the FGF family: FGFR1 and FGFR2 (Kang et al. 2017). Thus, these studies suggest that the outputs of FGF signalling in promoting expansion of epiblast cells and the segregation of the PE during these early embryonic stages depends on the availability of different FGFRs and the intracellular regulation of extracellular signal-regulated kinase (ERK) signalling. These new findings perhaps help us interpret the differences reported in the response to FGF signalling in other species. In human, bovine and porcine embryos, inhibition of the MAPK/ERK signalling pathway using the small molecule inhibitor PD0325901 does not prevent the complete ablation of the PE lineage when using equivalent doses of the inhibitor as in mice (Kuijk et al. 2012; Rodríguez et al. 2012; Roode et al. 2012). However, when the inhibitor is used at an increased concentration (25-fold higher), the PE is completely eliminated in bovine and porcine embryos (McLean et al. 2014; P. Ramos Ibeas and R. Alberio, unpubl. obs.). This could be due to an indirect effect or due to a reduced sensitivity of bovine and porcine embryonic cells to these compounds, because they may not rely on FGFR1/2 for the segregation of the PE lineage. Interestingly, in human embryos FGFR1 is not detected in the ICM, but instead it is preferentially expressed in the polar trophectoderm (Niakan and Eggan 2013). FGFR2 is also not present in human blastocysts (Kunath et al. 2014). Similarly, in the porcine, FGFR1 and FGFR2 are not detected in the ICM, but are highly expressed in the late E11 epiblast and the TE (Hall et al. 2009; Valdez Magaña et al. 2014). Interestingly, FGFR4 is detected in the ICM of bovine and porcine embryos (Wei et al. 2017; P. Ramos Ibeas and R. Alberio, unpubl. obs.), suggesting that FGF signalling can trigger a response in the ICM via this receptor. Indeed, we showed previously that blocking FGFRs using a pan-FGFR inhibitor affects the number of cells in the porcine ICM (Rodríguez et al. 2012); however, this was not observed in bovine embryos (McLean et al. 2014). Different FGFRs have been shown to preferentially use different signalling pathways following ligand binding (Brewer et al. 2016). FGFR4 has been associated with signal transducer and activator of transcription (STAT) 3, phospholipase Cγ and c-Jun N-terminal kinase (JNK) signalling, all of which are active pathways in naïve epiblasts in mice (Nichols and Smith 2009). Indeed, in the bovine, inhibition of JNK and p38/MAPK leads to a reduction in NANOG expression (McLean et al. 2014). Similarly, inhibition of STAT3 in bovine and porcine embryos results in reduced ICMs and lower NANOG expression (Meng et al. 2015; P. Ramos Ibeas and R. Alberio, unpubl. obs.). Based on these observations, it will be interesting to assess the role of FGFR4 during the segregation of epiblast from PE in larger mammals, and determine whether it participates in the establishment of pluripotency. Interestingly, another cytokine reported to stimulate STAT3 signalling is colony-stimulating factor (CSF) 2, which promotes proliferation of the ICM in human, porcine and bovine embryos (Sjöblom et al. 1999; Cui et al. 2004; Loureiro et al. 2009). Embryos produced in vitro supplemented with CSF also result in increased pregnancy rates (Sjöblom et al. 2005; Loureiro et al. 2009). It was also shown that CSF2 downregulates the MAPK/ERK pathway in bovine embryos (Ozawa et al. 2016), suggesting that it may promote epiblast expansion by modulating MAPK; however, it is not clear whether CSF2 causes shifts in the proportion of epiblasts versus PE in embryos. Because naïve pluripotent cells from non-rodent species cannot be easily captured (Guo et al. 2016), stimulation of these alternative signalling pathways could be considered in future ESC derivation attempts. Thus, the combined roles of CSF2 and FGFR4 in promoting STAT3 signalling while dampening MAPK during embryo emergence of the pluripotent ICM development deserve closer examination.
The role of other signalling pathways in hypoblast segregation is less explored. Inhibition of transforming growth factor β (TGFB)/activin A signalling using the inhibitor SB431542 (40 µM) between Days 5 and 7 in the human led to a reduced number of NANOG- and SOX17-positive cells (Blakeley et al. 2015). In bovine and porcine embryos, such an effect was not observed; however, the dose of the inhibitor was lower (20 µM), thus it is possible that higher concentrations of this inhibitor are needed (Kuijk et al. 2012; Rodríguez et al. 2012). Indeed, human embryos treated with 10 µM SB431542 had increased numbers of NANOG-positive cells (Van der Jeught et al. 2014). However, this is difficult to reconcile with the observation that inhibition of cultured porcine epiblast with 20 µM SB431542 for 6 days results in neural differentiation (Alberio et al. 2010). A similar observation was reported with human (h) ESCs (Van der Jeught et al. 2014). It is possible that the effect of the inhibitors in isolated epiblasts or hESC lines is much more robust than within the embryo proper, where additional signalling pathways may be operating, and therefore higher inhibitor concentrations may be required. Based on the expression of key components of the TGFB/activin pathway, it is likely that this signalling pathway is operational in early ICM cells in non-rodent mammals. Recent single-cell transcriptome studies have shown expression of NODAL, TGFB1, and ACVR1 in human, Cynomolgus monkey, bovine and porcine embryos (Cao et al. 2014; Blakeley et al. 2015; Nakamura et al. 2016; Petropoulos et al. 2016; Wei et al. 2017; P. Ramos Ibeas and R. Alberio, unpubl. obs.). Further studies are needed to determine the role of this pathway during establishment of pluripotency and segregation of PE.
The WNT signalling pathway is dispensable for lineage segregation in the mouse embryo (Biechele et al. 2011). In human, stimulation of WNT signalling together with mitogen-activated protein kinase kinase (MEK) inhibition does not affect the proportion of cells contributing to the epiblast or PE (Roode et al. 2012). Similarly, in bovine and porcine embryos, no effect of segregation is detected (Kuijk et al. 2012; Rodríguez et al. 2012). Interestingly, in bovine embryos it was also reported that WNT inhibition using DKK1 promotes differentiation into TE and PE cells (Denicol et al. 2014). WNT signalling has also been linked to promoting TE differentiation in the human embryo (Krivega et al. 2015).
Transcriptional network of metastable pluripotent states in the embryo
As discussed above, during the emergence of pluripotency, the triad of core transcription factors OCT4, SOX2 and NANOG is first detected in the ICM. Together, these factors establish a network of interactions that prevents differentiation and, at the same time, promotes the maintenance of pluripotency and self-renewing potential in ESCs (Boyer et al. 2005). In mouse embryos, the triad of gene expression is maintained for a brief period between E3.5 and E7.5, and during this period pluripotent stem cells with different morphological, molecular and developmental properties can be established (Brons et al. 2007; Nichols and Smith 2009; Osorno et al. 2012; Savatier et al. 2017). This brief developmental window is the critical period during which the embryo establishes and expands the pluripotent epiblast before engaging in differentiation during gastrulation. The expression of the core transcription factors is critically important for establishing and maintaining pluripotency, as demonstrated by knockout experiments in mice (Nichols et al. 1998; Mitsui et al. 2003). However, the expression of these factors in vivo varies between species, and how they participate in maintaining the pluripotency network is not well understood. Recent evidence from clustered regularly interspaced short palindromic repeat (Crispr)–Crispr-associated (Cas) 9 genome-edited human zygotes shows that OCT-4-null cells downregulate CDX2 and NANOG, and blastocyst development is impaired (Fogarty et al. 2017). The equivalent knockout in mouse embryos did not interfere with blastocyst formation, and orthologous genes were unaffected (Fogarty et al. 2017). This clearly highlights the important species-specific differences regulating early development in mammals, and a deeper understanding of these mechanisms across mammals will inform us of the evolutionary paths that were selected in different species, and will provide more relevant models of human development.
To begin to build detailed knowledge of the embryology of different species, new high-throughput single-cell transcriptomic and epigenomic platforms have been used to gather comprehensive high-resolution data of developmental progression in mammalian embryos (Xue et al. 2013; Cao et al. 2014; Blakeley et al. 2015; Boroviak et al. 2015; Clark et al. 2016; Nakamura et al. 2016; Petropoulos et al. 2016). The first studies performed in mouse embryos showed great heterogeneity of gene expression in individual cells, lending further support to the idea of regulative and stochastic processes regulating cell lineage allocation in early embryos (Dietrich and Hiiragi 2007). Indeed, single-cell transcript analysis showed that heterogeneous populations in the ICM stochastically segregate into epiblast and PE following reinforcement by antagonistic signalling (Ohnishi et al. 2014). Later studies comparing human and mouse early embryos showed broad conservation of pluripotency network; but, more importantly, they also revealed novel pathways and gene profiles present in human embryos (Blakeley et al. 2015). In addition to the expression of the core pluripotency genes NANOG, OCT4 and SOX2, many additional genes are shared between species, including KLF4, GDF3, DPPA2 and HESX1. Although KLF17 has been reported as a novel pluripotency marker in the human (Blakeley et al. 2015), it is not expressed in the mouse embryo (Blakeley et al. 2015). Other genes, like KLF2, ESRRB and BMP4, are not expressed in human ICM (Blakeley et al. 2015), whereas they are expressed in the mouse (Blakeley et al. 2015). In the porcine embryo, ESRRB, and KLF4 expression was also detected in the ICM (Cao et al. 2014). Similarly, PRDM14 is expressed in mouse and human embryos, but in porcine embryos PRDM14 is delayed to the expanded blastocyst stage and increases in the late epiblast stage (Cao et al. 2014; Kobayashi et al. 2017). Following analysis of components of different signalling pathways, it is apparent that human and mouse embryos are very different. Genes indicative of an active Janus tyrosine kinases (Jak)/Stat3 pathway, such as Lifr, Lif and Jak1/2 are upregulated, whereas the Jak/Stat repressor suppressor of cytokine signalling 3 (Socs3) is downregulated in the mouse (E3.5) early epiblast (Boroviak et al. 2014). Equally, TGFB/Nodal/activin A signalling components are not upregulated until the E4.5 stage in the mouse (Boroviak et al. 2014). In human embryos, ACVRL1, NODAL and LEFTY are expressed in the ICM, and this signalling pathway is apparently functional (Blakeley et al. 2015; Petropoulos et al. 2016), indicating an important divergence in the signalling requirements of emerging pluripotent cells between these species. These findings in the human are consistent with observations in porcine embryos demonstrating an active TGFB/Nodal/activin A signalling from the blastocyst stage until the late epiblast (van de Pavert et al. 2001; Alberio et al. 2010). However, a note of caution about comparing species and stages is needed, because the kinetics of development in the mouse are much shorter than in larger mammals, and perhaps some of the comparisons sometimes skip important intermediate stages that may exist in species with longer intervals between one stage and the next. For example, data from marmoset embryos collected at early, mid and late blastocyst stages show a gradual upregulation of the TGFB/Nodal/activin A signalling, concomitant with a gradual reduction in WNT signalling (Boroviak et al. 2015). Jak/Stat3 signalling was not reported in the marmoset. Thus, because of the extended period of ICM and epiblast development in larger mammals compared with mice, it is possible that intermediate, transitional stages transit the period of naïve towards lineage primed epiblast. Some evidence of this possibility has been reported, with all the caveats of in vitro systems, from ESC lines. A variety of different metastable states that resemble either the early naïve epiblast or more advanced developmental stages like the postimplantation epiblast have been captured under different conditions (Savatier et al. 2017), suggesting that the embryonic stage selected for ESC derivation can affect the efficiency of this process. Indeed, cells derived from an early developmental stage and placed in a culture system compatible with an advanced developmental stage of pluripotency can promote the conversion of these cells towards a more advanced developmental state, yet the opposite is less straightforward without forced gene expression (Guo et al. 2009; Greber et al. 2010; Boroviak et al. 2014). Indeed, in humans, where the naïve state has not been identified in the embryo proper, conventional hESCs exhibit properties of primed cells. There is only a single report of isolation of hESCs in naïve conditions, and these cells are unstable in culture (Guo et al. 2016). This suggests that either we do not yet have a good understanding of the requirements of human naïve cells or that this state does not exist in this species and cannot be captured without genetic manipulation (Theunissen et al. 2014; Takashima et al. 2015). These naïve hESCs show limited chimeric potential and are epigenetically unstable (Pastor et al. 2016; Theunissen et al. 2016). A recent study has presented some evidence of developmental correlates been between monkeys (Cynomolgus monkey), human stem cells and mouse embryonic cells, showing that conventional hESCs are more aligned with monkey postimplantation embryo cells, whereas naïve hESCs are more aligned with the monkey ICM and preimplantation epiblast (Nakamura et al. 2016). That analysis also showed that a stable pluripotency network is maintained for approximately 1 week in the monkey. Thus, it is possible that in humans and primates (and other non-rodent mammals) emergent pluripotent cells (~8- to 12-cells) transit through a naïve state during the establishment of pluripotency, but exit this stage within one cell division to then gradually establish the TGFB/Nodal/activin A pathway that promotes cell growth, expansion of the epiblast and finally lineage priming in preparation for gastrulation. To test this possibility further, studies investigating this important window of development in human and other large mammalian species are needed to provide a high-resolution transcription profile that depicts the early molecular program of pluripotency.
Epigenetic landscape of pluripotency
Accompanied by the distinctive gene expression profile of naïve and primed states of pluripotency, important epigenetic differences have been described. Naïve mouse ESCs exhibit reduced levels of H3K27 me3 at repressed promoters, global DNA hypomethylation (Marks et al. 2012; Habibi et al. 2013) mediated by reduced Uhrf1 protein, Dnmt1 activity and H3K9 dimethylation (me2) compared with ESCs grown in serum containing media (von Meyenn et al. 2016). Similarly, although higher-order chromatin is more relaxed in early cleavage stages, DNA hypersensitive sites (DHS) are found to increase in the morula stage, indicating increased chromatin accessibility during this period of lineage segregation (Lu et al. 2016). Indeed, many of the DHS are detected at Oct4-binding sites, suggesting that transcription factor expression during lineage specification contributes to creating a highly dynamic chromatin state that facilitates lineage priming. This is also consistent with the evidence that key transcription factors (TF) can impose new chromatin configuration by recruiting additional TF; for example, Oct4 and Sox2 dimerisation at target domains can nucleate enhanceosome assembly (Chen et al. 2014). Similarly, during the transition from naïve to primed pluripotency, Oct4 and Sox2 continue to be expressed, but their association with Otx changes targets specificity leading to new epiblast-like cell-specific enhancers (Buecker et al. 2014). Interestingly, although the transcriptome of murine (m) ESCs and epiblast stem cells (EpiSCs) is remarkably similar and the number of DHS at promoter regions is also equivalent, the enhancer landscape is sharply modified, with almost half these regions losing activity during EpiSC conversion (Factor et al. 2014). Thus, enhancer switching, such as the well-described differential regulation of the Oct4 enhancer (Yeom et al. 1996), represents an example of what probably applies more broadly to other enhancer regions during the transition from naïve to primed pluripotency. Enhancer switching requires topological changes of the chromatin to enable target recognition, and it has been shown that this major chromatin reorganisation can be triggered by external stimuli. Indeed, switching media from leukaemia inhibitory factor (LIF) to basic (b) FGF conditions rapidly triggers the transitions towards primed pluripotency (Guo et al. 2009). Thus, chromatin provides an additional level of regulation in cellular response mediating lineage specification.
Transcriptional and epigenetic regulation of the mammalian germline
In the developmental continuum of the embryo, a key event, known as gastrulation, consists in laying the foundations for the segregation of all the lineages that contribute to the make-up of the fetus (Wolpert 2008). This process has been largely studied in the mouse due to the accessibility of embryos and the possibility of performing advanced genetic studies (Arnold and Robertson 2009). However, most embryologists are familiar with some important structural differences between the early gastrulating mouse embryo and other mammalian species. Mouse and rat embryos develop as conical structures, also referred to as ‘cup-shape embryos’, which are distinct from the flat embryonic disc of other eutherian mammals, including human and domestic animal embryos. Understanding the geometries of these different embryos is critical for elucidating the inductive processes that mediate lineage specification. A careful evaluation of the interactions of the epiblast with neighbouring extra-embryonic tissues suggested that these tissue interactions are under considerably different influences, placing strong emphasis for diversifying the tissues on a proximodistal axis in mice compared with a radial axis (peripheral to central axis) in other mammals (Behringer et al. 2000). Indeed, the formation of the extra-embryonic mesoderm, one of the earliest epiblast derivatives, shows striking differences between human and mouse embryos. In the mouse, these cells derive from cells that ingress through the primitive streak, whereas in humans these tissues segregate before the appearance of a node (Eakin and Behringer 2004). Indeed, studies in porcine embryos have shown that precursors of the extra-embryonic mesoderm delaminate from the epiblast, rather than migrate through the streak (Fléchon et al. 2004). This important spatial difference in tissue emergence highlights key differences between mice and non-rodent species that may underlie divergent gene regulatory networks. For example, some evidence shows that the roles of NODAL and BMP gradients in establishing the anterior–posterior axis is unique to mice, whereas the role of NODAL in epiblast and visceral endoderm seems to be conserved in eutherian mammals (Yoshida et al. 2016). One of the key embryological structures that is also unique to mouse embryos is the extra-embryonic ectoderm (ExE), a derivative of the polar trophoblast. The ExE is a source of BMP, critical for mesoderm formation and germ cell specification in mice (Winnier et al. 1995; Lawson et al. 1999). Given the critical role of this tissue in patterning the mouse embryo, it seemed paradoxical that this is a unique structure to a subset of rodents, like mice and rats. We set out to investigate the source of BMP in embryos developing a flat morphology and showed that in the porcine embryo the primary source of BMP2 is the primitive endoderm of pre-primitive streak stage embryos, which is followed soon after by BMP4 produced by the extra-embryonic mesoderm (Valdez Magaña et al. 2014). This was later shown to coincide with upregulation of BRA (T), a key mesoderm differentiation marker (Yoshida et al. 2016).
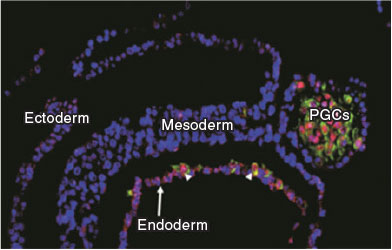
In mice, BMP induces germ cell specification from posterior proximal epiblast cells by segregating lineage-restricted cells that activate the germ cell program through a tripartite gene expression network established by Prdm14, Blimp1 and Tfap2c (Yamaji et al. 2008; Magnúsdóttir et al. 2013). The key function of this network is to inhibit the somatic differentiation program and to promote the upregulation of pluripotency genes and epigenetic reprogramming. Reactivation of pluripotency genes Sox2 and Nanog is a hallmark of mouse primordial germ cells (PGC) development and their expression is maintained until these cells reach the gonads (Yamaguchi et al. 2005; Campolo et al. 2013). The significance of pluripotent gene expression in PGC development is unclear, but mouse mutants for these genes show defects due to cell loss by apoptosis and reduced cell proliferation (Yamaguchi et al. 2009), suggesting that these genes act as survival factors and in the expansion of the initial pool of PGCs. Owing to the striking difference in BMP4 expression between murine and porcine embryos, we asked whether these divergent BMP signalling domains could have resulted in an altered program of PGC specification from other precursors. Evidence from in vitro differentiation of hESCs showed that the program of PGC specification is determined by activation of SOX17 and BLIMP1 (Irie et al. 2015). These findings were confirmed in Cynomolgus monkeys, suggesting they may be shared among primates (Sasaki et al. 2016). We used this new paradigm to interrogate the porcine embryo during the early stages of gastrulation and confirmed that the sequence of initial SOX17 followed by BLIMP1 activation marks the PGCs in the porcine embryo (Kobayashi et al. 2017). Importantly, these cells are found in the posterior end of the embryo where the nascent mesodermal derivatives emerge (Fig. 1). This is consistent with the area where BMP4 is produced in the porcine embryo (Valdez Magaña et al. 2014; Yoshida et al. 2016). Thus, this shows that PGCs in large mammals emerge from a ‘pluripotent mesoderm’ precursor population in response to BMP4. Interestingly, evidence from organisms with inductive mechanisms of germ cell specification, such as the basal amphibian Ambystoma mexicanum (axolotl), also show the presence of a pluripotent mesoderm from which PGCs are specified upon BMP4 induction (Chatfield et al. 2014). These observations have led to the hypothesis that in species with a regulative mode of development, the PGCs arise from a pool of progenitors after the onset of gastrulation, consistent with the presence of a stem cell niche. In species with early germ cell lineage segregation, which is the most prevalent among extant species, liberation of genetic constraints has enabled the rewiring of existing gene regulatory networks, resulting in increased adaptability and speciation (Johnson and Alberio 2015). Indeed, within mammals it seems that mice have established a novel gene regulatory network of germline development that diverges from that described in humans, monkeys and pigs. In addition to the changes in the TF network, we showed that the onset of epigenetic reprogramming, a key event that defines germline development, is different in human and pigs. PRDM14, a key molecule responsible for initiating epigenetic reprogramming in the mouse, is weakly expressed in early human, monkey and porcine PGCs. Furthermore, DNA demethylation in mouse PGCs is primarily driven by passive cell division-coupled dilution and by repression of Dnmt3A/B dependent on Blimp1 and Prdm14 (Kagiwada et al. 2013; Ohno et al. 2013). Active DNA demethylation mediated by the enzymatic conversion of 5-methylcytosine (5-meC) to 5-hydroxymethylcytosine (5-hmeC) is first detected in late migratory mouse PGCs, when they start to colonise the genital ridges (Hackett et al. 2013; Yamaguchi et al. 2013). In a human PGC differentiation model, high levels of TeT1 expression are detected at Day 2 and higher 5-hmeC is detected at Day 4 of induction in cells that presumably represent premigratory stages and have initiated epigenetic reprogramming, as demonstrated by reduced UHRF1, H3K9 me2, and DNMT3A (Irie et al. 2015). Similarly, in porcine embryos, higher levels of TET1 and 5-hmeC and lower levels of UHRF1 and H3K9 me2 are detected in early premigratory PGCs (Kobayashi et al. 2017), indicating that as soon as the PGC program is induced following the activation of SOX17 and BLIMP1, the epigenetic machinery is activated in a cell cycle-independent manner, as demonstrated by the marked reduction in DNA replication in nascent PGCs. Collectively, the evidence at the level of gene expression, onset of epigenetic reprogramming and cell cycle arrest demonstrates fundamental differences in the program that ensues upon germ cell specification in large mammals. It will be interesting to investigate whether equivalent differences also apply to other programs of development in species with conserved embryology and, more generally, what the evolutionary consequences are of changes in the GRNs regulating early gastrulation for adaptation and speciation.
|
Concluding remarks
The highly regulative mechanisms directing mammalian development have been studied in great detail in the mouse, but more recently efforts have been directed towards large mammalian species, which, in many examples, are showing closer relevance to the developmental mechanisms of humans. A better knowledge of large animal embryology offers new opportunities for investigating basic mechanisms of development, but can also have applications for improving assisted reproductive technologies and understanding the effects of these technologies on the health status of these animals in adulthood. The availability of full genome sequences of domestic animals and the incorporation of gene-editing technologies in the battery of approaches for disrupting gene function in zygotes, combined with single-cell transcriptomics and new live imaging techniques, are revolutionising the way we study developmental biology in large mammals. Many old questions and new questions that have arisen from recent discoveries can be addressed using elegant and sophisticated molecular approaches. Is naïve pluripotency a feature of rodent embryos or has it not been observed or captured in large mammals due to the poor resolution of available techniques? How did TE segregation evolve in different mammals? Do epiblast cells transit through metastable states of pluripotency in vivo? If so, how important is this for the developmental potential of different lineages during gastrulation? These are examples of some questions that biologists should be able to address using the new toolkit available for dissecting the intricate mechanisms of embryo development.
Conflicts of interest
The author declares no conflicts of interest.
Acknowledgements
Work in the author’s laboratory is funded by Biotechnology and Biological Sciences Research Council (BBSRC; BB/M001466/1).
References
Ahmed, K., Dehghani, H., Rugg-Gunn, P., Fussner, E., Rossant, J., and Bazett-Jones, D. P. (2010). Global chromatin architecture reflects pluripotency and lineage commitment in the early mouse embryo. PLoS One 5, e10531.| Global chromatin architecture reflects pluripotency and lineage commitment in the early mouse embryo.Crossref | GoogleScholarGoogle Scholar |
Alberio, R., Croxall, N., and Allegrucci, C. (2010). Pig epiblast stem cells depend on activin/nodal signaling for pluripotency and self-renewal. Stem Cells Dev. 19, 1627–1636.
| Pig epiblast stem cells depend on activin/nodal signaling for pluripotency and self-renewal.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3cXht1Wmu7vE&md5=b0cad1e479753a950043cf8d8cb53fa8CAS |
Arman, E., Haffner-Krausz, R., Chen, Y., Heath, J. K., and Lonai, P. (1998). Targeted disruption of fibroblast growth factor (FGF) receptor 2 suggests a role for FGF signaling in pregastrulation mammalian development. Proc. Natl Acad. Sci. USA 95, 5082–5087.
| Targeted disruption of fibroblast growth factor (FGF) receptor 2 suggests a role for FGF signaling in pregastrulation mammalian development.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK1cXivV2jsLo%3D&md5=d03e87e213ac67adf138b62a76b7f5b2CAS |
Arnold, S. J., and Robertson, E. J. (2009). Making a commitment: cell lineage allocation and axis patterning in the early mouse embryo. Nat. Rev. Mol. Cell Biol. 10, 91–103.
| Making a commitment: cell lineage allocation and axis patterning in the early mouse embryo.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1MXitlGntw%3D%3D&md5=9111fc4db6c0dbdc4dc9315ba95c5c66CAS |
Artus, J., Piliszek, A., and Hadjantonakis, A. K. (2011). The primitive endoderm lineage of the mouse blastocyst: sequential transcription factor activation and regulation of differentiation by Sox17. Dev. Biol. 350, 393–404.
| The primitive endoderm lineage of the mouse blastocyst: sequential transcription factor activation and regulation of differentiation by Sox17.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3MXhtlKnt70%3D&md5=ca499e368f51da3b6041aba52bf94ed9CAS |
Behringer, R. R., Wakamiya, M., Tsang, T. E., and Tam, P. P. (2000). A flattened mouse embryo: leveling the playing field. Genesis 28, 23–30.
| A flattened mouse embryo: leveling the playing field.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DC%2BD3M%2FivFKisA%3D%3D&md5=0348a922f085bcea3cc46528fa2e7f7dCAS |
Berg, D. K., Smith, C. S., Pearton, D. J., Wells, D. N., Broadhurst, R., Donnison, M., and Pfeffer, P. L. (2011). Trophectoderm lineage determination in cattle. Dev. Cell 20, 244–255.
| Trophectoderm lineage determination in cattle.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3MXhvFeit7Y%3D&md5=39359cd401e0a820c8f8c6c5070ef594CAS |
Biechele, S., Cox, B. J., and Rossant, J. (2011). Porcupine homolog is required for canonical Wnt signaling and gastrulation in mouse embryos. Dev. Biol. 355, 275–285.
| Porcupine homolog is required for canonical Wnt signaling and gastrulation in mouse embryos.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3MXnslOlsbw%3D&md5=c619be67b0846935815d3e5c95f001b7CAS |
Blakeley, P., Fogarty, N. M., del Valle, I., Wamaitha, S. E., Hu, T. X., Elder, K., Snell, P., Christie, L., Robson, P., and Niakan, K. K. (2015). Defining the three cell lineages of the human blastocyst by single-cell RNA-seq. Development 142, 3151–3165.
| Defining the three cell lineages of the human blastocyst by single-cell RNA-seq.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28XksV2rur8%3D&md5=482fa3f89290180fdd69ca93a82d3ed4CAS |
Boroviak, T., Loos, R., Bertone, P., Smith, A., and Nichols, J. (2014). The ability of inner-cell-mass cells to self-renew as embryonic stem cells is acquired following epiblast specification. Nat. Cell Biol. 16, 516–528.
| The ability of inner-cell-mass cells to self-renew as embryonic stem cells is acquired following epiblast specification.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2cXos1eqsb0%3D&md5=b86be3d67c24be8f68147f94b2a25fdeCAS |
Boroviak, T., Loos, R., Lombard, P., Okahara, J., Behr, R., Sasaki, E., Nichols, J., Smith, A., and Bertone, P. (2015). Lineage-specific profiling delineates the emergence and progression of naive pluripotency in mammalian embryogenesis. Dev. Cell 35, 366–382.
| Lineage-specific profiling delineates the emergence and progression of naive pluripotency in mammalian embryogenesis.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2MXhvVSksr7P&md5=0bc784b1c0350179e0d9fca3b112cfdfCAS |
Bou, G., Liu, S., Guo, J., Zhao, Y., Sun, M., Xue, B., Wang, J., Wei, Y., Kong, Q., and Liu, Z. (2016). Cdx2 represses Oct4 function via inducing its proteasome-dependent degradation in early porcine embryos. Dev. Biol. 410, 36–44.
| Cdx2 represses Oct4 function via inducing its proteasome-dependent degradation in early porcine embryos.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2MXitVGgs7zP&md5=d72f2410b623d4f17cf10351195df2f5CAS |
Bou, G., Liu, S., Sun, M., Zhu, J., Xue, B., Guo, J., Zhao, Y., Qu, B., Weng, X., Wei, Y., Lei, L., and Liu, Z. (2017). CDX2 is essential for cell proliferation and polarity in porcine blastocysts. Development 144, 1296–1306.
| CDX2 is essential for cell proliferation and polarity in porcine blastocysts.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2sXhtF2gsrzO&md5=c9fcb61f8e01697a89391077a71bd55dCAS |
Boyer, L. A., Lee, T. I., Cole, M. F., Johnstone, S. E., Levine, S. S., Zucker, J. P., Guenther, M. G., Kumar, R. M., Murray, H. L., Jenner, R. G., Gifford, D. K., Melton, D. A., Jaenisch, R., and Young, R. A. (2005). Core transcriptional regulatory circuitry in human embryonic stem cells. Cell 122, 947–956.
| Core transcriptional regulatory circuitry in human embryonic stem cells.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2MXhtVOrurbJ&md5=e78ff095c08ce07c10882446a291d845CAS |
Brewer, J. R., Mazot, P., and Soriano, P. (2016). Genetic insights into the mechanisms of Fgf signaling. Genes Dev. 30, 751–771.
| Genetic insights into the mechanisms of Fgf signaling.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28Xht1KjurfN&md5=eed1041622e48dd61f52d185f2f6b14aCAS |
Brons, I. G., Smithers, L. E., Trotter, M. W., Rugg-Gunn, P., Sun, B., Chuva de Sousa Lopes, S. M., Howlett, S. K., Clarkson, A., Ahrlund-Richter, L., Pedersen, R. A., and Vallier, L. (2007). Derivation of pluripotent epiblast stem cells from mammalian embryos. Nature 448, 191–195.
| Derivation of pluripotent epiblast stem cells from mammalian embryos.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2sXnsFeisbw%3D&md5=946fa4d5038703b514bc7b4b99d28056CAS |
Buecker, C., Srinivasan, R., Wu, Z., Calo, E., Acampora, D., Faial, T., Simeone, A., Tan, M., Swigut, T., and Wysocka, J. (2014). Reorganization of enhancer patterns in transition from naive to primed pluripotency. Cell Stem Cell 14, 838–853.
| Reorganization of enhancer patterns in transition from naive to primed pluripotency.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2cXpvFSktL0%3D&md5=bcda370228629e5b054e53155fbaf2fcCAS |
Burton, A., Muller, J., Tu, S., Padilla-Longoria, P., Guccione, E., and Torres-Padilla, M. E. (2013). Single-cell profiling of epigenetic modifiers identifies PRDM14 as an inducer of cell fate in the mammalian embryo. Cell Reports 5, 687–701.
| Single-cell profiling of epigenetic modifiers identifies PRDM14 as an inducer of cell fate in the mammalian embryo.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXhslWmtr7P&md5=0f541c0a65387cd3e1e8be6a37f78b02CAS |
Campolo, F., Gori, M., Favaro, R., Nicolis, S., Pellegrini, M., Botti, F., Rossi, P., Jannini, E. A., and Dolci, S. (2013). Essential role of Sox2 for the establishment and maintenance of the germ cell line. Stem Cells 31, 1408–1421.
| Essential role of Sox2 for the establishment and maintenance of the germ cell line.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXhtlWjsL3M&md5=fa09717f4a3bda5e3865c6e6868dcd9dCAS |
Cao, S., Han, J., Wu, J., Li, Q., Liu, S., Zhang, W., Pei, Y., Ruan, X., Liu, Z., Wang, X., Lim, B., and Li, N. (2014). Specific gene-regulation networks during the pre-implantation development of the pig embryo as revealed by deep sequencing. BMC Genomics 15, 4.
| Specific gene-regulation networks during the pre-implantation development of the pig embryo as revealed by deep sequencing.Crossref | GoogleScholarGoogle Scholar |
Cauffman, G., Van de Velde, H., Liebaers, I., and Van Steirteghem, A. (2005). Oct-4 mRNA and protein expression during human preimplantation development. Mol. Hum. Reprod. 11, 173–181.
| Oct-4 mRNA and protein expression during human preimplantation development.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2MXitV2jtr8%3D&md5=41b200ab62473793fdcc3c3fb39c93b8CAS |
Cauffman, G., De Rycke, M., Sermon, K., Liebaers, I., and Van de Velde, H. (2009). Markers that define stemness in ESC are unable to identify the totipotent cells in human preimplantation embryos. Hum. Reprod. 24, 63–70.
| Markers that define stemness in ESC are unable to identify the totipotent cells in human preimplantation embryos.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXhsFWjtbfJ&md5=d296cd094fedf1554c2ea62ec1bd83dfCAS |
Chatfield, J., O’Reilly, M. A., Bachvarova, R. F., Ferjentsik, Z., Redwood, C., Walmsley, M., Patient, R., Loose, M., and Johnson, A. D. (2014). Stochastic specification of primordial germ cells from mesoderm precursors in axolotl embryos. Development 141, 2429–2440.
| Stochastic specification of primordial germ cells from mesoderm precursors in axolotl embryos.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2cXht1WkurrL&md5=4af323ffb79205751b692e87270ec28aCAS |
Chazaud, C., Yamanaka, Y., Pawson, T., and Rossant, J. (2006). Early lineage segregation between epiblast and primitive endoderm in mouse blastocysts through the Grb2–MAPK pathway. Dev. Cell 10, 615–624.
| Early lineage segregation between epiblast and primitive endoderm in mouse blastocysts through the Grb2–MAPK pathway.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD28XltVyktro%3D&md5=8f1b3339a54ca64b349b7b01000284f7CAS |
Chen, J., Zhang, Z., Li, L., Chen, B. C., Revyakin, A., Hajj, B., Legant, W., Dahan, M., Lionnet, T., Betzig, E., Tjian, R., and Liu, Z. (2014). Single-molecule dynamics of enhanceosome assembly in embryonic stem cells. Cell 156, 1274–1285.
| Single-molecule dynamics of enhanceosome assembly in embryonic stem cells.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2cXksVWrtbg%3D&md5=0094dc478920284498419cd6993b8ae2CAS |
Cheng, A. M., Saxton, T. M., Sakai, R., Kulkarni, S., Mbamalu, G., Vogel, W., Tortorice, C. G., Cardiff, R. D., Cross, J. C., Muller, W. J., and Pawson, T. (1998). Mammalian Grb2 regulates multiple steps in embryonic development and malignant transformation. Cell 95, 793–803.
| Mammalian Grb2 regulates multiple steps in embryonic development and malignant transformation.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK1MXivVKi&md5=da58c75e964f9922a0c2ae5dc0361f49CAS |
Clark, S. J., Lee, H. J., Smallwood, S. A., Kelsey, G., and Reik, W. (2016). Single-cell epigenomics: powerful new methods for understanding gene regulation and cell identity. Genome Biol. 17, 72.
| Single-cell epigenomics: powerful new methods for understanding gene regulation and cell identity.Crossref | GoogleScholarGoogle Scholar |
Cui, X. S., Lee, J. Y., Choi, S. H., Kwon, M. S., Kim, T., and Kim, N. H. (2004). Mouse granulocyte–macrophage colony-stimulating factor enhances viability of porcine embryos in defined culture conditions. Anim. Reprod. Sci. 84, 169–177.
| Mouse granulocyte–macrophage colony-stimulating factor enhances viability of porcine embryos in defined culture conditions.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2cXmsVGjs70%3D&md5=1e55780e362f18eb0c22f2f1dd9cf5f6CAS |
Dahl, J. A., Jung, I., Aanes, H., Greggains, G. D., Manaf, A., Lerdrup, M., Li, G., Kuan, S., Li, B., Lee, A. Y., Preissl, S., Jermstad, I., Haugen, M. H., Suganthan, R., Bjoras, M., Hansen, K., Dalen, K. T., Fedorcsak, P., Ren, B., and Klungland, A. (2016). Broad histone H3K4me3 domains in mouse oocytes modulate maternal-to-zygotic transition. Nature 537, 548–552.
| Broad histone H3K4me3 domains in mouse oocytes modulate maternal-to-zygotic transition.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28XhsFWiurnI&md5=5c867a72a40c1ea6f435fc6293ce75c1CAS |
De Paepe, C., Cauffman, G., Verloes, A., Sterckx, J., Devroey, P., Tournaye, H., Liebaers, I., and Van de Velde, H. (2013). Human trophectoderm cells are not yet committed. Hum. Reprod. 28, 740–749.
| Human trophectoderm cells are not yet committed.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXislOmtb8%3D&md5=70026d31dc2c98e8fd354d5e323822f2CAS |
Denicol, A. C., Block, J., Kelley, D. E., Pohler, K. G., Dobbs, K. B., Mortensen, C. J., Ortega, M. S., and Hansen, P. J. (2014). The WNT signaling antagonist Dickkopf-1 directs lineage commitment and promotes survival of the preimplantation embryo. FASEB J. 28, 3975–3986.
| The WNT signaling antagonist Dickkopf-1 directs lineage commitment and promotes survival of the preimplantation embryo.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2cXhsFanur%2FM&md5=d0ee4b0ab24b0bab372889b33611334cCAS |
Dietrich, J. E., and Hiiragi, T. (2007). Stochastic patterning in the mouse pre-implantation embryo. Development 134, 4219–4231.
| Stochastic patterning in the mouse pre-implantation embryo.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXmtFegtw%3D%3D&md5=71757b0dd53aff8eb0a44676881e116aCAS |
Eakin, G. S., and Behringer, R. R. (2004). Diversity of germ layer and axis formation among mammals. Semin. Cell Dev. Biol. 15, 619–629.
| Diversity of germ layer and axis formation among mammals.Crossref | GoogleScholarGoogle Scholar |
Evsikov, A. V., Graber, J. H., Brockman, J. M., Hampl, A., Holbrook, A. E., Singh, P., Eppig, J. J., Solter, D., and Knowles, B. B. (2006). Cracking the egg: molecular dynamics and evolutionary aspects of the transition from the fully grown oocyte to embryo. Genes Dev. 20, 2713–2727.
| Cracking the egg: molecular dynamics and evolutionary aspects of the transition from the fully grown oocyte to embryo.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD28XhtV2hsLjN&md5=594d5aa651d32e84b00f392780eed29eCAS |
Factor, D. C., Corradin, O., Zentner, G. E., Saiakhova, A., Song, L., Chenoweth, J. G., McKay, R. D., Crawford, G. E., Scacheri, P. C., and Tesar, P. J. (2014). Epigenomic comparison reveals activation of “seed” enhancers during transition from naive to primed pluripotency. Cell Stem Cell 14, 854–863.
| Epigenomic comparison reveals activation of “seed” enhancers during transition from naive to primed pluripotency.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2cXpvFSktLs%3D&md5=4195f22f6829caf4bbf5ed42d35bf597CAS |
Feldman, B., Poueymirou, W., Papaioannou, V. E., DeChiara, T. M., and Goldfarb, M. (1995). Requirement of FGF-4 for postimplantation mouse development. Science 267, 246–249.
| Requirement of FGF-4 for postimplantation mouse development.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK2MXjt1ensbo%3D&md5=a92b21f8165554cf36d7479993c66ad6CAS |
Fléchon, J. E., Degrouard, J., and Fléchon, B. (2004). Gastrulation events in the prestreak pig embryo: ultrastructure and cell markers. Genesis 38, 13–25.
| Gastrulation events in the prestreak pig embryo: ultrastructure and cell markers.Crossref | GoogleScholarGoogle Scholar |
Fogarty, N. M. E., McCarthy, A., Snijders, K. E., Powell, B. E., Kubikova, N., Blakeley, P., Lea, R., Elder, K., Wamaitha, S. E., Kim, D., Maciulyte, V., Kleinjung, J., Kim, J. S., Wells, D., Vallier, L., Bertero, A., Turner, J. M. A., and Niakan, K. K. (2017). Genome editing reveals a role for OCT4 in human embryogenesis. Nature 550, 67–73.
| Genome editing reveals a role for OCT4 in human embryogenesis.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2sXhsFehtrzI&md5=36543ec33550e77d9f2324aa0fd043c7CAS |
Goolam, M., Scialdone, A., Graham, S. J. L., Macaulay, I. C., Jedrusik, A., Hupalowska, A., Voet, T., Marioni, J. C., and Zernicka-Goetz, M. (2016). Heterogeneity in Oct4 and Sox2 targets biases cell fate in 4-cell mouse embryos. Cell 165, 61–74.
| Heterogeneity in Oct4 and Sox2 targets biases cell fate in 4-cell mouse embryos.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28XltVamu70%3D&md5=e73d838690b8589da78381d0c242fd1fCAS |
Greber, B., Wu, G., Bernemann, C., Joo, J. Y., Han, D. W., Ko, K., Tapia, N., Sabour, D., Sterneckert, J., Tesar, P., and Scholer, H. R. (2010). Conserved and divergent roles of FGF signaling in mouse epiblast stem cells and human embryonic stem cells. Cell Stem Cell 6, 215–226.
| Conserved and divergent roles of FGF signaling in mouse epiblast stem cells and human embryonic stem cells.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3cXlt1KmsrY%3D&md5=d47cf74e0f93dea6384dd565158b897fCAS |
Guo, G., Yang, J., Nichols, J., Hall, J. S., Eyres, I., Mansfield, W., and Smith, A. (2009). Klf4 reverts developmentally programmed restriction of ground state pluripotency. Development 136, 1063–1069.
| Klf4 reverts developmentally programmed restriction of ground state pluripotency.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1MXlt1Oqt7g%3D&md5=7bbda9496a20c136e99c3a5fcbed7f59CAS |
Guo, G., Huss, M., Tong, G. Q., Wang, C., Li Sun, L., Clarke, N. D., and Robson, P. (2010). Resolution of cell fate decisions revealed by single-cell gene expression analysis from zygote to blastocyst. Dev. Cell 18, 675–685.
| Resolution of cell fate decisions revealed by single-cell gene expression analysis from zygote to blastocyst.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3cXmsF2mtbo%3D&md5=36cbdc7c050aa5d8e855a2ded0ec29c3CAS |
Guo, G., von Meyenn, F., Santos, F., Chen, Y., Reik, W., Bertone, P., Smith, A., and Nichols, J. (2016). Naive pluripotent stem cells derived directly from isolated cells of the human inner cell mass. Stem Cell Reports 6, 437–446.
| Naive pluripotent stem cells derived directly from isolated cells of the human inner cell mass.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28Xjs1Ghur8%3D&md5=14e5fcb144f2bc13c2122ac77e8353d4CAS |
Habibi, E., Brinkman, A. B., Arand, J., Kroeze, L. I., Kerstens, H. H., Matarese, F., Lepikhov, K., Gut, M., Brun-Heath, I., Hubner, N. C., Benedetti, R., Altucci, L., Jansen, J. H., Walter, J., Gut, I. G., Marks, H., and Stunnenberg, H. G. (2013). Whole-genome bisulfite sequencing of two distinct interconvertible DNA methylomes of mouse embryonic stem cells. Cell Stem Cell 13, 360–369.
| Whole-genome bisulfite sequencing of two distinct interconvertible DNA methylomes of mouse embryonic stem cells.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXhtV2rur3L&md5=73fff5e70702aa16f058cacaf72da1e1CAS |
Hackett, J. A., Sengupta, R., Zylicz, J. J., Murakami, K., Lee, C., Down, T. A., and Surani, M. A. (2013). Germline DNA demethylation dynamics and imprint erasure through 5-hydroxymethylcytosine. Science 339, 448–452.
| Germline DNA demethylation dynamics and imprint erasure through 5-hydroxymethylcytosine.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXhtFyntL8%3D&md5=f2729cd2aab4c5005495ddc6ebb088a8CAS |
Hall, V. J., Christensen, J., Gao, Y., Schmidt, M. H., and Hyttel, P. (2009). Porcine pluripotency cell signaling develops from the inner cell mass to the epiblast during early development. Dev. Dyn. 238, 2014–2024.
| Porcine pluripotency cell signaling develops from the inner cell mass to the epiblast during early development.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1MXhtVKrsbbO&md5=b52bdf56fde36b3a8c1b38bfcfb7f901CAS |
Irie, N., Weinberger, L., Tang, W. W., Kobayashi, T., Viukov, S., Manor, Y. S., Dietmann, S., Hanna, J. H., and Surani, M. A. (2015). SOX17 is a critical specifier of human primordial germ cell fate. Cell 160, 253–268.
| SOX17 is a critical specifier of human primordial germ cell fate.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2MXnslGlsA%3D%3D&md5=36e24b099dada6a206881ffa0cd028b3CAS |
Ishiuchi, T., Enriquez-Gasca, R., Mizutani, E., Boskovic, A., Ziegler-Birling, C., Rodriguez-Terrones, D., Wakayama, T., Vaquerizas, J. M., and Torres-Padilla, M. E. (2015). Early embryonic-like cells are induced by downregulating replication-dependent chromatin assembly. Nat. Struct. Mol. Biol. 22, 662–671.
| Early embryonic-like cells are induced by downregulating replication-dependent chromatin assembly.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2MXht1entLfJ&md5=1601306665a7fe9986ae1048996d8409CAS |
Jachowicz, J. W., Bing, X., Pontabry, J., Boskovic, A., Rando, O. J., and Torres-Padilla, M. E. (2017). LINE-1 activation after fertilization regulates global chromatin accessibility in the early mouse embryo. Nat. Genet. 49, 1502–1510.
| LINE-1 activation after fertilization regulates global chromatin accessibility in the early mouse embryo.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2sXhtl2gtL%2FO&md5=ecc154e29f9f87a59547c304e09bd16aCAS |
Johnson, A. D., and Alberio, R. (2015). Primordial germ cells: the first cell lineage or the last cells standing? Development 142, 2730–2739.
| Primordial germ cells: the first cell lineage or the last cells standing?Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2MXhs1enu77E&md5=053bf0d7458a345f9a59eadc90a48a2eCAS |
Johnson, M. H., and Ziomek, C. A. (1983). Cell interactions influence the fate of mouse blastomeres undergoing the transition from the 16- to the 32-cell stage. Dev. Biol. 95, 211–218.
| Cell interactions influence the fate of mouse blastomeres undergoing the transition from the 16- to the 32-cell stage.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DyaL3s7jt1Oqtg%3D%3D&md5=6c13a6a533232b94dfc1760d524a0e4eCAS |
Kagiwada, S., Kurimoto, K., Hirota, T., Yamaji, M., and Saitou, M. (2013). Replication-coupled passive DNA demethylation for the erasure of genome imprints in mice. EMBO J. 32, 340–353.
| Replication-coupled passive DNA demethylation for the erasure of genome imprints in mice.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC38XhvVektL7P&md5=3796561d277acd969fdd12ec9f593026CAS |
Kang, M., Garg, V., and Hadjantonakis, A. K. (2017). Lineage establishment and progression within the inner cell mass of the mouse blastocyst requires FGFR1 and FGFR2. Dev. Cell 41, 496–510.e5.
| Lineage establishment and progression within the inner cell mass of the mouse blastocyst requires FGFR1 and FGFR2.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2sXos1Wjsrw%3D&md5=68cd929ed7f31b355454ac55cc6a99f3CAS |
Khan, D. R., Dube, D., Gall, L., Peynot, N., Ruffini, S., Laffont, L., Le Bourhis, D., Degrelle, S., Jouneau, A., and Duranthon, V. (2012). Expression of pluripotency master regulators during two key developmental transitions: EGA and early lineage specification in the bovine embryo. PLoS One 7, e34110.
| Expression of pluripotency master regulators during two key developmental transitions: EGA and early lineage specification in the bovine embryo.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC38XlsFamsbk%3D&md5=eedd6c894b2a5d22b2f19cb38e5a56d7CAS |
Kimber, S. J., Sneddon, S. F., Bloor, D. J., El-Bareg, A. M., Hawkhead, J. A., Metcalfe, A. D., Houghton, F. D., Leese, H. J., Rutherford, A., Lieberman, B. A., and Brison, D. R. (2008). Expression of genes involved in early cell fate decisions in human embryos and their regulation by growth factors. Reproduction 135, 635–647.
| Expression of genes involved in early cell fate decisions in human embryos and their regulation by growth factors.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXls1Wqur4%3D&md5=8f54bd37fafe795e8504104557192cf8CAS |
Kirchhof, N., Carnwath, J. W., Lemme, E., Anastassiadis, K., Scholer, H., and Niemann, H. (2000). Expression pattern of Oct-4 in preimplantation embryos of different species. Biol. Reprod. 63, 1698–1705.
| Expression pattern of Oct-4 in preimplantation embryos of different species.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD3cXosVKhtrY%3D&md5=22cb3839e2b34ea2d4e10999b2f0de3cCAS |
Kobayashi, T., Zhang, H., Tang, W. W. C., Irie, N., Withey, S., Klisch, D., Sybirna, A., Dietmann, S., Contreras, D. A., Webb, R., Allegrucci, C., Alberio, R., and Surani, M. A. (2017). Principles of early human development and germ cell program from conserved model systems. Nature 546, 416–420.
| Principles of early human development and germ cell program from conserved model systems.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2sXpt1Oqtbs%3D&md5=71396156cd0f486e4f7e7c11734d664dCAS |
Korotkevich, E., Niwayama, R., Courtois, A., Friese, S., Berger, N., Buchholz, F., and Hiiragi, T. (2017). The apical domain is required and sufficient for the first lineage segregation in the mouse embryo. Dev. Cell 40, 235–247.e7.
| The apical domain is required and sufficient for the first lineage segregation in the mouse embryo.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2sXisVCqurk%3D&md5=d77da1dbaeb12e878f3e970472437158CAS |
Koyama, H., Suzuki, H., Yang, X., Jiang, S., and Foote, R. H. (1994). Analysis of polarity of bovine and rabbit embryos by scanning electron microscopy. Biol. Reprod. 50, 163–170.
| Analysis of polarity of bovine and rabbit embryos by scanning electron microscopy.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DyaK2c7ks1ajsw%3D%3D&md5=02974857440e215359e50ff51f960b47CAS |
Krivega, M., Essahib, W., and Van de Velde, H. (2015). WNT3 and membrane-associated beta-catenin regulate trophectoderm lineage differentiation in human blastocysts. Mol. Hum. Reprod. 21, 711–722.
| WNT3 and membrane-associated beta-catenin regulate trophectoderm lineage differentiation in human blastocysts.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2sXhsVGgsbzM&md5=75ce7a8d6c5bf6efb15c00fa30fd7da3CAS |
Kuijk, E. W., Du Puy, L., Van Tol, H. T., Oei, C. H., Haagsman, H. P., Colenbrander, B., and Roelen, B. A. (2008). Differences in early lineage segregation between mammals. Dev. Dyn. 237, 918–927.
| Differences in early lineage segregation between mammals.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXltFaktbY%3D&md5=00dcf92eecffcc6518036bce906b5e31CAS |
Kuijk, E. W., van Tol, L. T., Van de Velde, H., Wubbolts, R., Welling, M., Geijsen, N., and Roelen, B. A. (2012). The roles of FGF and MAP kinase signaling in the segregation of the epiblast and hypoblast cell lineages in bovine and human embryos. Development 139, 871–882.
| The roles of FGF and MAP kinase signaling in the segregation of the epiblast and hypoblast cell lineages in bovine and human embryos.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC38Xmsl2hu7c%3D&md5=906de039634fae68877672286bfd6426CAS |
Kunath, T., Yamanaka, Y., Detmar, J., MacPhee, D., Caniggia, I., Rossant, J., and Jurisicova, A. (2014). Developmental differences in the expression of FGF receptors between human and mouse embryos. Placenta 35, 1079–1088.
| Developmental differences in the expression of FGF receptors between human and mouse embryos.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2cXhs1Kmtr7I&md5=8b90da1a98a9d4e6422a4e291d7d0c75CAS |
Lawson, K. A., Dunn, N. R., Roelen, B. A., Zeinstra, L. M., Davis, A. M., Wright, C. V., Korving, J. P., and Hogan, B. L. (1999). Bmp4 is required for the generation of primordial germ cells in the mouse embryo. Genes Dev. 13, 424–436.
| Bmp4 is required for the generation of primordial germ cells in the mouse embryo.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK1MXhs1ajtL0%3D&md5=8431e87c942be4bca6c2a21fb9eed87cCAS |
Liu, H., Kim, J. M., and Aoki, F. (2004). Regulation of histone H3 lysine 9 methylation in oocytes and early pre-implantation embryos. Development 131, 2269–2280.
| Regulation of histone H3 lysine 9 methylation in oocytes and early pre-implantation embryos.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2cXltlGrtrs%3D&md5=756ec95fe4b704afde95bcc0b386cc69CAS |
Liu, S., Bou, G., Sun, R., Guo, S., Xue, B., Wei, R., Cooney, A. J., and Liu, Z. (2015). Sox2 is the faithful marker for pluripotency in pig: evidence from embryonic studies. Dev. Dyn. 244, 619–627.
| Sox2 is the faithful marker for pluripotency in pig: evidence from embryonic studies.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2MXlt1Wlsrw%3D&md5=5d422fdc5f9ac3c5ebbb7d33859aa040CAS |
Liu, X., Wang, C., Liu, W., Li, J., Li, C., Kou, X., Chen, J., Zhao, Y., Gao, H., Wang, H., Zhang, Y., Gao, Y., and Gao, S. (2016). Distinct features of H3K4me3 and H3K27me3 chromatin domains in pre-implantation embryos. Nature 537, 558–562.
| Distinct features of H3K4me3 and H3K27me3 chromatin domains in pre-implantation embryos.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28XhsFWiurbO&md5=050ef23d9b30446ccfaa70cc8851f0acCAS |
Loureiro, B., Bonilla, L., Block, J., Fear, J. M., Bonilla, A. Q., and Hansen, P. J. (2009). Colony-stimulating factor 2 (CSF-2) improves development and posttransfer survival of bovine embryos produced in vitro. Endocrinology 150, 5046–5054.
| Colony-stimulating factor 2 (CSF-2) improves development and posttransfer survival of bovine embryos produced in vitro.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1MXhsVCht7bF&md5=c662f1929cf665c12c19a680a7ceb919CAS |
Lu, F., Liu, Y., Inoue, A., Suzuki, T., Zhao, K., and Zhang, Y. (2016). Establishing chromatin regulatory landscape during mouse preimplantation development. Cell 165, 1375–1388.
| Establishing chromatin regulatory landscape during mouse preimplantation development.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28XpslaltrY%3D&md5=2a03e394d88c694e437603e17eb52ce6CAS |
Macfarlan, T. S., Gifford, W. D., Driscoll, S., Lettieri, K., Rowe, H. M., Bonanomi, D., Firth, A., Singer, O., Trono, D., and Pfaff, S. L. (2012). Embryonic stem cell potency fluctuates with endogenous retrovirus activity. Nature 487, 57–63.
| 1:CAS:528:DC%2BC38XpvVSmsb4%3D&md5=8c26dbf9335025e7ded33657b131aaa4CAS |
Magnúsdóttir, E., Dietmann, S., Murakami, K., Günesdogan, U., Tang, F., Bao, S., Diamanti, E., Lao, K., Gottgens, B., and Azim Surani, M. (2013). A tripartite transcription factor network regulates primordial germ cell specification in mice. Nat. Cell Biol. 15, 905–915.
| A tripartite transcription factor network regulates primordial germ cell specification in mice.Crossref | GoogleScholarGoogle Scholar |
Maître, J. L., Turlier, H., Illukkumbura, R., Eismann, B., Niwayama, R., Nédélec, F., and Hiiragi, T. (2016). Asymmetric division of contractile domains couples cell positioning and fate specification. Nature 536, 344–348.
| Asymmetric division of contractile domains couples cell positioning and fate specification.Crossref | GoogleScholarGoogle Scholar |
Marks, H., Kalkan, T., Menafra, R., Denissov, S., Jones, K., Hofemeister, H., Nichols, J., Kranz, A., Stewart, A. F., Smith, A., and Stunnenberg, H. G. (2012). The transcriptional and epigenomic foundations of ground state pluripotency. Cell 149, 590–604.
| The transcriptional and epigenomic foundations of ground state pluripotency.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC38Xmt1Gqtbw%3D&md5=d57b725eb0fbb07713536b2f378a7b3aCAS |
McLean, Z., Meng, F., Henderson, H., Turner, P., and Oback, B. (2014). Increased MAP kinase inhibition enhances epiblast-specific gene expression in bovine blastocysts. Biol. Reprod. 91, 49.
| Increased MAP kinase inhibition enhances epiblast-specific gene expression in bovine blastocysts.Crossref | GoogleScholarGoogle Scholar |
Meilhac, S. M., Adams, R. J., Morris, S. A., Danckaert, A., Le Garrec, J. F., and Zernicka-Goetz, M. (2009). Active cell movements coupled to positional induction are involved in lineage segregation in the mouse blastocyst. Dev. Biol. 331, 210–221.
| Active cell movements coupled to positional induction are involved in lineage segregation in the mouse blastocyst.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1MXpt1Wmtbo%3D&md5=39e3b1387fdfaf1fbcd436f3ab1c502dCAS |
Meng, F., Forrester-Gauntlett, B., Turner, P., Henderson, H., and Oback, B. (2015). Signal inhibition reveals JAK/STAT3 pathway as critical for bovine inner cell mass development. Biol. Reprod. 93, 132.
| Signal inhibition reveals JAK/STAT3 pathway as critical for bovine inner cell mass development.Crossref | GoogleScholarGoogle Scholar |
Messerschmidt, D. M., and Kemler, R. (2010). Nanog is required for primitive endoderm formation through a non-cell autonomous mechanism. Dev. Biol. 344, 129–137.
| Nanog is required for primitive endoderm formation through a non-cell autonomous mechanism.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3cXhtVClu77M&md5=f9689ae1e061d013beb5276d718c823dCAS |
Mitsui, K., Tokuzawa, Y., Itoh, H., Segawa, K., Murakami, M., Takahashi, K., Maruyama, M., Maeda, M., and Yamanaka, S. (2003). The homeoprotein Nanog is required for maintenance of pluripotency in mouse epiblast and ES cells. Cell 113, 631–642.
| The homeoprotein Nanog is required for maintenance of pluripotency in mouse epiblast and ES cells.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD3sXksFehur4%3D&md5=3fea1e5e26e7e5747bc6fa8674baf450CAS |
Molotkov, A., Mazot, P., Brewer, J. R., Cinalli, R. M., and Soriano, P. (2017). Distinct requirements for FGFR1 and FGFR2 in primitive endoderm development and exit from pluripotency. Dev. Cell 41, 511–526.e4.
| Distinct requirements for FGFR1 and FGFR2 in primitive endoderm development and exit from pluripotency.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2sXos1Wjs74%3D&md5=8458a335174b6dd9de4bc9c394000466CAS |
Morris, S. A., Graham, S. J., Jedrusik, A., and Zernicka-Goetz, M. (2013). The differential response to Fgf signalling in cells internalized at different times influences lineage segregation in preimplantation mouse embryos. Open Biol. 3, 130104.
| The differential response to Fgf signalling in cells internalized at different times influences lineage segregation in preimplantation mouse embryos.Crossref | GoogleScholarGoogle Scholar |
Motosugi, N., Bauer, T., Polanski, Z., Solter, D., and Hiiragi, T. (2005). Polarity of the mouse embryo is established at blastocyst and is not prepatterned. Genes Dev. 19, 1081–1092.
| Polarity of the mouse embryo is established at blastocyst and is not prepatterned.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2MXkt1Cgsrk%3D&md5=e8118f8b691a85928d413ce8bc324bfaCAS |
Nakamura, T., Okamoto, I., Sasaki, K., Yabuta, Y., Iwatani, C., Tsuchiya, H., Seita, Y., Nakamura, S., Yamamoto, T., and Saitou, M. (2016). A developmental coordinate of pluripotency among mice, monkeys and humans. Nature 537, 57–62.
| A developmental coordinate of pluripotency among mice, monkeys and humans.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28XhsVamtrvL&md5=f179a2ecaec92e22c2d6208bb7e91aa3CAS |
Ng, R. K., Dean, W., Dawson, C., Lucifero, D., Madeja, Z., Reik, W., and Hemberger, M. (2008). Epigenetic restriction of embryonic cell lineage fate by methylation of Elf5. Nat. Cell Biol. 10, 1280–1290.
| Epigenetic restriction of embryonic cell lineage fate by methylation of Elf5.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXhtlais7nN&md5=05a61e9dc1a76a6a3db31ba57b7b28ddCAS |
Niakan, K. K., and Eggan, K. (2013). Analysis of human embryos from zygote to blastocyst reveals distinct gene expression patterns relative to the mouse. Dev. Biol. 375, 54–64.
| Analysis of human embryos from zygote to blastocyst reveals distinct gene expression patterns relative to the mouse.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXht1Oksr4%3D&md5=670ccee0bcaf416f840b2683faa6af11CAS |
Nichols, J., and Smith, A. (2009). Naive and primed pluripotent states. Cell Stem Cell 4, 487–492.
| Naive and primed pluripotent states.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1MXnt1Ggt7c%3D&md5=c9a1f1da399988794003f5d74eb13a14CAS |
Nichols, J., Zevnik, B., Anastassiadis, K., Niwa, H., Klewe-Nebenius, D., Chambers, I., Scholer, H., and Smith, A. (1998). Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4. Cell 95, 379–391.
| Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK1cXntlCqt74%3D&md5=f9da3159e1f4ac09f842e5858fd2dc11CAS |
Nichols, J., Silva, J., Roode, M., and Smith, A. (2009). Suppression of Erk signalling promotes ground state pluripotency in the mouse embryo. Development 136, 3215–3222.
| Suppression of Erk signalling promotes ground state pluripotency in the mouse embryo.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1MXhtlyjsrjJ&md5=29fb94f22beb472a92cd1bd5a535a56fCAS |
Nikas, G., Ao, A., Winston, R. M., and Handyside, A. H. (1996). Compaction and surface polarity in the human embryo in vitro. Biol. Reprod. 55, 32–37.
| Compaction and surface polarity in the human embryo in vitro.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK28XltlKnsL8%3D&md5=fab72a36ddf18805998a2cf68289b556CAS |
Nishioka, N., Inoue, K., Adachi, K., Kiyonari, H., Ota, M., Ralston, A., Yabuta, N., Hirahara, S., Stephenson, R. O., Ogonuki, N., Makita, R., Kurihara, H., Morin-Kensicki, E. M., Nojima, H., Rossant, J., Nakao, K., Niwa, H., and Sasaki, H. (2009). The Hippo signaling pathway components Lats and Yap pattern Tead4 activity to distinguish mouse trophectoderm from inner cell mass. Dev. Cell 16, 398–410.
| The Hippo signaling pathway components Lats and Yap pattern Tead4 activity to distinguish mouse trophectoderm from inner cell mass.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1MXjs1ynsrw%3D&md5=fa95429b9f9a238df9489ad97ea52c69CAS |
Ohnishi, Y., Huber, W., Tsumura, A., Kang, M., Xenopoulos, P., Kurimoto, K., Oles, A. K., Arauzo-Bravo, M. J., Saitou, M., Hadjantonakis, A. K., and Hiiragi, T. (2014). Cell-to-cell expression variability followed by signal reinforcement progressively segregates early mouse lineages. Nat. Cell Biol. 16, 27–37.
| Cell-to-cell expression variability followed by signal reinforcement progressively segregates early mouse lineages.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXhvVGqs7%2FM&md5=a22d8f6e21c2a56fab766884ca9d210bCAS |
Ohno, R., Nakayama, M., Naruse, C., Okashita, N., Takano, O., Tachibana, M., Asano, M., Saitou, M., and Seki, Y. (2013). A replication-dependent passive mechanism modulates DNA demethylation in mouse primordial germ cells. Development 140, 2892–2903.
| A replication-dependent passive mechanism modulates DNA demethylation in mouse primordial germ cells.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXhtlCmu7fE&md5=abfad23900759444fce417b8f9382503CAS |
Osorno, R., Tsakiridis, A., Wong, F., Cambray, N., Economou, C., Wilkie, R., Blin, G., Scotting, P. J., Chambers, I., and Wilson, V. (2012). The developmental dismantling of pluripotency is reversed by ectopic Oct4 expression. Development 139, 2288–2298.
| The developmental dismantling of pluripotency is reversed by ectopic Oct4 expression.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC38XhtFOgtbfE&md5=046a1ff3c7eb764fa869381e4f7f42a0CAS |
Ozawa, M., Sakatani, M., Dobbs, K. B., Kannampuzha-Francis, J., and Hansen, P. J. (2016). Regulation of gene expression in the bovine blastocyst by colony stimulating factor 2. BMC Res. Notes 9, 250.
| Regulation of gene expression in the bovine blastocyst by colony stimulating factor 2.Crossref | GoogleScholarGoogle Scholar |
Pastor, W. A., Chen, D., Liu, W., Kim, R., Sahakyan, A., Lukianchikov, A., Plath, K., Jacobsen, S. E., and Clark, A. T. (2016). Naive human pluripotent cells feature a methylation landscape devoid of blastocyst or germline memory. Cell Stem Cell 18, 323–329.
| Naive human pluripotent cells feature a methylation landscape devoid of blastocyst or germline memory.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28Xit1Siu7k%3D&md5=7597044612ec87031d3474cffe979eaaCAS |
Pearton, D. J., Broadhurst, R., Donnison, M., and Pfeffer, P. L. (2011). Elf5 regulation in the trophectoderm. Dev. Biol. 360, 343–350.
| Elf5 regulation in the trophectoderm.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3MXhsVOjtb3J&md5=61b01713b61fa6a3be85f2c51fa9edfdCAS |
Pearton, D. J., Smith, C. S., Redgate, E., van Leeuwen, J., Donnison, M., and Pfeffer, P. L. (2014). Elf5 counteracts precocious trophoblast differentiation by maintaining Sox2 and 3 and inhibiting Hand1 expression. Dev. Biol. 392, 344–357.
| Elf5 counteracts precocious trophoblast differentiation by maintaining Sox2 and 3 and inhibiting Hand1 expression.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2cXpslKlsLk%3D&md5=8a80b01ba84b1ba8e7daa994a50d1327CAS |
Peaston, A. E., Evsikov, A. V., Graber, J. H., de Vries, W. N., Holbrook, A. E., Solter, D., and Knowles, B. B. (2004). Retrotransposons regulate host genes in mouse oocytes and preimplantation embryos. Dev. Cell 7, 597–606.
| Retrotransposons regulate host genes in mouse oocytes and preimplantation embryos.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2cXptlGjur4%3D&md5=03d9fea6d61893761d441118cdf8446fCAS |
Pedersen, R. A., Wu, K., and Balakier, H. (1986). Origin of the inner cell mass in mouse embryos: cell lineage analysis by microinjection. Dev. Biol. 117, 581–595.
| Origin of the inner cell mass in mouse embryos: cell lineage analysis by microinjection.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DyaL2s%2FgsFGrsA%3D%3D&md5=b6a3b90e5e546f9ea6cf2e4b1898b1b9CAS |
Peter, I. S., and Davidson, E. H. (2017). Assessing regulatory information in developmental gene regulatory networks. Proc. Natl Acad. Sci. USA 114, 5862–5869.
| Assessing regulatory information in developmental gene regulatory networks.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2sXpt1Ons7k%3D&md5=1ade57b35db31493418e5bb89a992502CAS |
Petropoulos, S., Edsgard, D., Reinius, B., Deng, Q., Panula, S. P., Codeluppi, S., Plaza Reyes, A., Linnarsson, S., Sandberg, R., and Lanner, F. (2016). Single-cell RNA-Seq reveals lineage and X chromosome dynamics in human preimplantation embryos. Cell 165, 1012–1026.
| Single-cell RNA-Seq reveals lineage and X chromosome dynamics in human preimplantation embryos.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28XlslWqur8%3D&md5=ddbcdad414bd4ef9d164dcc3f06d74d6CAS |
Pfeffer, P. L., and Pearton, D. J. (2012). Trophoblast development. Reproduction 143, 231–246.
| Trophoblast development.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC38XlvFOqs7c%3D&md5=b228a102d4cff0797090e9170700de5eCAS |
Plusa, B., Piliszek, A., Frankenberg, S., Artus, J., and Hadjantonakis, A. K. (2008). Distinct sequential cell behaviours direct primitive endoderm formation in the mouse blastocyst. Development 135, 3081–3091.
| Distinct sequential cell behaviours direct primitive endoderm formation in the mouse blastocyst.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXhtlansrbE&md5=819a83a3c2f95d72e44dea1583498dccCAS |
Ralston, A., and Rossant, J. (2008). Cdx2 acts downstream of cell polarization to cell-autonomously promote trophectoderm fate in the early mouse embryo. Dev. Biol. 313, 614–629.
| Cdx2 acts downstream of cell polarization to cell-autonomously promote trophectoderm fate in the early mouse embryo.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXlsFGrsA%3D%3D&md5=420b3e15a082a780798d03bf631fda03CAS |
Reima, I., Lehtonen, E., Virtanen, I., and Flechon, J. E. (1993). The cytoskeleton and associated proteins during cleavage, compaction and blastocyst differentiation in the pig. Differentiation 54, 35–45.
| The cytoskeleton and associated proteins during cleavage, compaction and blastocyst differentiation in the pig.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK2cXhtlSmt7s%3D&md5=fe59def3cfbd27be217768e509a695a7CAS |
Rodríguez, A., Allegrucci, C., and Alberio, R. (2012). Modulation of pluripotency in the porcine embryo and iPS cells. PLoS One 7, e49079.
| Modulation of pluripotency in the porcine embryo and iPS cells.Crossref | GoogleScholarGoogle Scholar |
Roode, M., Blair, K., Snell, P., Elder, K., Marchant, S., Smith, A., and Nichols, J. (2012). Human hypoblast formation is not dependent on FGF signalling. Dev. Biol. 361, 358–363.
| Human hypoblast formation is not dependent on FGF signalling.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3MXhs12hu7nO&md5=fcee03095da3314409f9ba6d1b8c0aefCAS |
Sasaki, K., Nakamura, T., Okamoto, I., Yabuta, Y., Iwatani, C., Tsuchiya, H., Seita, Y., Nakamura, S., Shiraki, N., Takakuwa, T., Yamamoto, T., and Saitou, M. (2016). The germ cell fate of cynomolgus monkeys is specified in the nascent amnion. Dev. Cell 39, 169–185.
| The germ cell fate of cynomolgus monkeys is specified in the nascent amnion.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28Xhs1ClsL7I&md5=1a9ca199bf7041c4bfde0bff99816ef9CAS |
Savatier, P., Osteil, P., and Tam, P. P. (2017). Pluripotency of embryo-derived stem cells from rodents, lagomorphs, and primates: slippery slope, terrace and cliff. Stem Cell Res. 19, 104–112.
| Pluripotency of embryo-derived stem cells from rodents, lagomorphs, and primates: slippery slope, terrace and cliff.Crossref | GoogleScholarGoogle Scholar |
Sjöblom, C., Wikland, M., and Robertson, S. A. (1999). Granulocyte–macrophage colony-stimulating factor promotes human blastocyst development in vitro. Hum. Reprod. 14, 3069–3076.
| Granulocyte–macrophage colony-stimulating factor promotes human blastocyst development in vitro.Crossref | GoogleScholarGoogle Scholar |
Sjöblom, C., Roberts, C. T., Wikland, M., and Robertson, S. A. (2005). Granulocyte–macrophage colony-stimulating factor alleviates adverse consequences of embryo culture on fetal growth trajectory and placental morphogenesis. Endocrinology 146, 2142–2153.
| Granulocyte–macrophage colony-stimulating factor alleviates adverse consequences of embryo culture on fetal growth trajectory and placental morphogenesis.Crossref | GoogleScholarGoogle Scholar |
Steptoe, P. C., Edwards, R. G., and Purdy, J. M. (1971). Human blastocysts grown in culture. Nature 229, 132–133.
| Human blastocysts grown in culture.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DyaE3M%2FmtlGlsw%3D%3D&md5=a8e1d1aef46473b764cae9e3c39e7e42CAS |
Suwińska, A., Czołowska, R., Ożdżeński, W., and Tarkowski, A. K. (2008). Blastomeres of the mouse embryo lose totipotency after the fifth cleavage division: expression of Cdx2 and Oct4 and developmental potential of inner and outer blastomeres of 16- and 32-cell embryos. Dev. Biol. 322, 133–144.
| Blastomeres of the mouse embryo lose totipotency after the fifth cleavage division: expression of Cdx2 and Oct4 and developmental potential of inner and outer blastomeres of 16- and 32-cell embryos.Crossref | GoogleScholarGoogle Scholar |
Szczepanska, K., Stanczuk, L., and Maleszewski, M. (2011). Isolated mouse inner cell mass is unable to reconstruct trophectoderm. Differentiation 82, 1–8.
| Isolated mouse inner cell mass is unable to reconstruct trophectoderm.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3MXnsVymt7w%3D&md5=42bb8230e05c80f747db34da6626c821CAS |
Takashima, Y., Guo, G., Loos, R., Nichols, J., Ficz, G., Krueger, F., Oxley, D., Santos, F., Clarke, J., Mansfield, W., Reik, W., Bertone, P., and Smith, A. (2015). Resetting transcription factor control circuitry toward ground-state pluripotency in human. Cell 162, 452–453.
| Resetting transcription factor control circuitry toward ground-state pluripotency in human.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2MXht1KgtLzL&md5=e76b8c96624fe66ebca958fbe3304b10CAS |
Tarkowski, A. K., and Wroblewska, J. (1967). Development of blastomeres of mouse eggs isolated at the 4- and 8-cell stage. J. Embryol. Exp. Morphol. 18, 155–180.
| 1:STN:280:DyaF1c%2Fgt1arsg%3D%3D&md5=5100175b689b42228dfdf3837a68b964CAS |
Theunissen, T. W., Powell, B. E., Wang, H., Mitalipova, M., Faddah, D. A., Reddy, J., Fan, Z. P., Maetzel, D., Ganz, K., Shi, L., Lungjangwa, T., Imsoonthornruksa, S., Stelzer, Y., Rangarajan, S., D’Alessio, A., Zhang, J., Gao, Q., Dawlaty, M. M., Young, R. A., Gray, N. S., and Jaenisch, R. (2014). Systematic identification of culture conditions for induction and maintenance of naive human pluripotency. Cell Stem Cell 15, 471–487.
| Systematic identification of culture conditions for induction and maintenance of naive human pluripotency.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2cXht1agsL%2FN&md5=d3d1872e9d2dcdb68342da9396af6e90CAS |
Theunissen, T. W., Friedli, M., He, Y., Planet, E., O’Neil, R. C., Markoulaki, S., Pontis, J., Wang, H., Iouranova, A., Imbeault, M., Duc, J., Cohen, M. A., Wert, K. J., Castanon, R., Zhang, Z., Huang, Y., Nery, J. R., Drotar, J., Lungjangwa, T., Trono, D., Ecker, J. R., and Jaenisch, R. (2016). Molecular criteria for defining the naive human pluripotent state. Cell Stem Cell 19, 502–515.
| Molecular criteria for defining the naive human pluripotent state.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28XhtFyiur%2FM&md5=d027bdbf709c093641379fdb3e893ffdCAS |
Valdez Magaña, G., Rodríguez, A., Zhang, H., Webb, R., and Alberio, R. (2014). Paracrine effects of embryo-derived FGF4 and BMP4 during pig trophoblast elongation. Dev. Biol. 387, 15–27.
| Paracrine effects of embryo-derived FGF4 and BMP4 during pig trophoblast elongation.Crossref | GoogleScholarGoogle Scholar |
van de Pavert, S. A., Boerjan, M. L., Stroband, H. W., Taverne, M. A., and van den Hurk, R. (2001). Uterine–embryonic interaction in pig: activin, follistatin, and activin receptor II in uterus and embryo during early gestation. Mol. Reprod. Dev. 59, 390–399.
| Uterine–embryonic interaction in pig: activin, follistatin, and activin receptor II in uterus and embryo during early gestation.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD3MXltVWmsL0%3D&md5=0a8640fb1e6f57173e06c9f31d052a31CAS |
Van der Jeught, M., Heindryckx, B., O’Leary, T., Duggal, G., Ghimire, S., Lierman, S., Van Roy, N., Chuva de Sousa Lopes, S. M., Deroo, T., Deforce, D., and De Sutter, P. (2014). Treatment of human embryos with the TGFbeta inhibitor SB431542 increases epiblast proliferation and permits successful human embryonic stem cell derivation. Hum. Reprod. 29, 41–48.
| Treatment of human embryos with the TGFbeta inhibitor SB431542 increases epiblast proliferation and permits successful human embryonic stem cell derivation.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXhvFequrfE&md5=b146e30dd9d0c15acfe8578fcebb6a6eCAS |
Van Soom, A., Boerjan, M. L., Bols, P. E., Vanroose, G., Lein, A., Coryn, M., and de Kruif, A. (1997). Timing of compaction and inner cell allocation in bovine embryos produced in vivo after superovulation. Biol. Reprod. 57, 1041–1049.
| Timing of compaction and inner cell allocation in bovine embryos produced in vivo after superovulation.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK2sXmvFSns7o%3D&md5=a6ba18e599e3f4aa2c1b7a3f90283d6eCAS |
von Meyenn, F., Iurlaro, M., Habibi, E., Liu, N. Q., Salehzadeh-Yazdi, A., Santos, F., Petrini, E., Milagre, I., Yu, M., Xie, Z., Kroeze, L. I., Nesterova, T. B., Jansen, J. H., Xie, H., He, C., Reik, W., and Stunnenberg, H. G. (2016). Impairment of DNA methylation maintenance is the main cause of global demethylation in naive embryonic stem cells. Mol. Cell 62, 983.
| Impairment of DNA methylation maintenance is the main cause of global demethylation in naive embryonic stem cells.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28XhtVWlurjP&md5=f3f08bb184983d17adc9eba1a6d0dd03CAS |
Wei, Q., Zhong, L., Zhang, S., Mu, H., Xiang, J., Yue, L., Dai, Y., and Han, J. (2017). Bovine lineage specification revealed by single-cell gene expression analysis from zygote to blastocyst. Biol. Reprod. 97, 5–17.
| Bovine lineage specification revealed by single-cell gene expression analysis from zygote to blastocyst.Crossref | GoogleScholarGoogle Scholar |
White, M. D., Angiolini, J. F., Alvarez, Y. D., Kaur, G., Zhao, Z. W., Mocskos, E., Bruno, L., Bissiere, S., Levi, V., and Plachta, N. (2016). Long-lived binding of Sox2 to DNA predicts cell fate in the four-cell mouse embryo. Cell 165, 75–87.
| Long-lived binding of Sox2 to DNA predicts cell fate in the four-cell mouse embryo.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28XltVamurw%3D&md5=ad697014ccedfab5f9ee6df02c683c75CAS |
Winnier, G., Blessing, M., Labosky, P. A., and Hogan, B. L. (1995). Bone morphogenetic protein-4 is required for mesoderm formation and patterning in the mouse. Genes Dev. 9, 2105–2116.
| Bone morphogenetic protein-4 is required for mesoderm formation and patterning in the mouse.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK2MXotVOnu7w%3D&md5=eba32cf9f797c768276802b45c6d9699CAS |
Wolpert, L. (2008). ‘The Triumph of the Embryo.’ (Dover Publications: New York.)
Wu, J., Huang, B., Chen, H., Yin, Q., Liu, Y., Xiang, Y., Zhang, B., Liu, B., Wang, Q., Xia, W., Li, W., Li, Y., Ma, J., Peng, X., Zheng, H., Ming, J., Zhang, W., Zhang, J., Tian, G., Xu, F., Chang, Z., Na, J., Yang, X., and Xie, W. (2016). The landscape of accessible chromatin in mammalian preimplantation embryos. Nature 534, 652–657.
| The landscape of accessible chromatin in mammalian preimplantation embryos.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28XhtVSksbjP&md5=e0c18b854c1437094dc62dfe8193b09eCAS |
Xue, Z., Huang, K., Cai, C., Cai, L., Jiang, C. Y., Feng, Y., Liu, Z., Zeng, Q., Cheng, L., Sun, Y. E., Liu, J. Y., Horvath, S., and Fan, G. (2013). Genetic programs in human and mouse early embryos revealed by single-cell RNA sequencing. Nature 500, 593–597.
| Genetic programs in human and mouse early embryos revealed by single-cell RNA sequencing.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXhtFygsLrL&md5=4d78534b57a4b0d6a116754aa563079fCAS |
Yamaguchi, S., Kimura, H., Tada, M., Nakatsuji, N., and Tada, T. (2005). Nanog expression in mouse germ cell development. Gene Expr. Patterns 5, 639–646.
| Nanog expression in mouse germ cell development.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2MXltFSlsbw%3D&md5=37d306dc1399bfe5be503a3b88f7292fCAS |
Yamaguchi, S., Kurimoto, K., Yabuta, Y., Sasaki, H., Nakatsuji, N., Saitou, M., and Tada, T. (2009). Conditional knockdown of Nanog induces apoptotic cell death in mouse migrating primordial germ cells. Development 136, 4011–4020.
| Conditional knockdown of Nanog induces apoptotic cell death in mouse migrating primordial germ cells.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3cXislSktg%3D%3D&md5=1dcaaca298c74ccce611af6a6e81bc81CAS |
Yamaguchi, S., Hong, K., Liu, R., Inoue, A., Shen, L., Zhang, K., and Zhang, Y. (2013). Dynamics of 5-methylcytosine and 5-hydroxymethylcytosine during germ cell reprogramming. Cell Res. 23, 329–339.
| Dynamics of 5-methylcytosine and 5-hydroxymethylcytosine during germ cell reprogramming.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXjsFaqtb0%3D&md5=f86df0d72a6e0b4088d9900a35febd3bCAS |
Yamaji, M., Seki, Y., Kurimoto, K., Yabuta, Y., Yuasa, M., Shigeta, M., Yamanaka, K., Ohinata, Y., and Saitou, M. (2008). Critical function of Prdm14 for the establishment of the germ cell lineage in mice. Nat. Genet. 40, 1016–1022.
| Critical function of Prdm14 for the establishment of the germ cell lineage in mice.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXptVKrsr4%3D&md5=31b310f222a3a9e4555a7068cbb2e055CAS |
Yamanaka, Y., Lanner, F., and Rossant, J. (2010). FGF signal-dependent segregation of primitive endoderm and epiblast in the mouse blastocyst. Development 137, 715–724.
| FGF signal-dependent segregation of primitive endoderm and epiblast in the mouse blastocyst.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3cXksVChu78%3D&md5=da1157a6a16fe0ce613adcbed7b3419bCAS |
Yan, L., Yang, M., Guo, H., Yang, L., Wu, J., Li, R., Liu, P., Lian, Y., Zheng, X., Yan, J., Huang, J., Li, M., Wu, X., Wen, L., Lao, K., Li, R., Qiao, J., and Tang, F. (2013). Single-cell RNA-Seq profiling of human preimplantation embryos and embryonic stem cells. Nat. Struct. Mol. Biol. 20, 1131–1139.
| Single-cell RNA-Seq profiling of human preimplantation embryos and embryonic stem cells.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXht1CgsLnO&md5=b0769540dbe89ea9068963ef0a6026e4CAS |
Yeom, Y. I., Fuhrmann, G., Ovitt, C. E., Brehm, A., Ohbo, K., Gross, M., Hubner, K., and Scholer, H. R. (1996). Germline regulatory element of Oct-4 specific for the totipotent cycle of embryonal cells. Development 122, 881–894.
| 1:CAS:528:DyaK28XhvVOrtbs%3D&md5=52e8ac06c641d09e0d543d9652bca9efCAS |
Yoshida, M., Kajikawa, E., Kurokawa, D., Tokunaga, T., Onishi, A., Yonemura, S., Kobayashi, K., Kiyonari, H., and Aizawa, S. (2016). Conserved and divergent expression patterns of markers of axial development in eutherian mammals. Dev. Dyn. 245, 67–86.
| Conserved and divergent expression patterns of markers of axial development in eutherian mammals.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2MXitVentLvM&md5=e255182a04a92458d41db999db4cff1dCAS |
Zhang, B., Zheng, H., Huang, B., Li, W., Xiang, Y., Peng, X., Ming, J., Wu, X., Zhang, Y., Xu, Q., Liu, W., Kou, X., Zhao, Y., He, W., Li, C., Chen, B., Li, Y., Wang, Q., Ma, J., Yin, Q., Kee, K., Meng, A., Gao, S., Xu, F., Na, J., and Xie, W. (2016). Allelic reprogramming of the histone modification H3K4me3 in early mammalian development. Nature 537, 553–557.
| Allelic reprogramming of the histone modification H3K4me3 in early mammalian development.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC28XhsFWiurbN&md5=a4516948740113cd9282960498108682CAS |


