34 OVINE SPERM IS HIGHLY SUSCEPTIBLE TO ATTACK BY HYDROGEN PEROXIDE
E. G. A. Perez A , M. Nichi A , F. A. Oliveira Neto B , R. O. C. Silva A , A. Dalmazzo A , C. H. C. Viana B , D. M. C. Pesce B , P. A. A. Góes A , G. H. Ferreira B , V. H. Barnabe A and R. C. Barnabe AA University of São Paulo, São Paulo, Brazil;
B Pontifícia Universidade Católica, Poços de Caldas, MG, Brazil
Reproduction, Fertility and Development 22(1) 175-175 https://doi.org/10.1071/RDv22n1Ab34
Published: 8 December 2009
Abstract
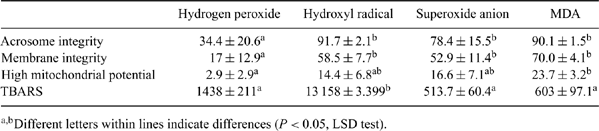
Ram sperm membrane displays a particular lipid composition, especially regarding the high quantity of polyunsaturated cholesterol. This trait improves membrane fluidity; however, the spermatozoa become more susceptible to the attack of reactive oxygen species (ROS), which may lead to structural and functional damage, impairment or even impeded fecundity. The aim of the present experiment was to study the resistance of ovine spermatozoa to different ROS. Sperm samples from 4 rams were collected using an artificial vagina. Sperm samples were then incubated (1 h, 37°C) with four ROS inducer mechanisms: xanthine/xanthine oxidase (produces superoxide anion), hydrogen peroxide (4 mM), ascorbate/ferrous sulfate (4 mM; produces hydroxyl radical), and malondialdehyde (MDA, lipid peroxidation product). Samples were analysed using the 3-3′ diamino benzidine (DAB) stain as an index of mitochondrial activity, the eosin nigrosin stain as an index of membrane integrity; the simple stain (fast green/Bengal rose) as an index of acrosome integrity; and the measurement of thiobarbituric acid reactive substances (TBARS) as an index of lipid peroxidation. Results showed that acrosome and membrane integrity as well as mitochondrial potential were highly impaired by hydrogen peroxide, which was not the case for the other ROS (Table 1). Surprisingly, TBARS production was higher in samples incubated with ascorbate and ferrous sulfate (hydroxyl radical). Furthermore, sperm showing impaired mitochondrial potential were negatively correlated with membrane and acrosome integrities (r = -0.83, P < 0.0001 and r = -0.62, P = 0.01, respectively). Results of the present experiment suggest that semen of rams is extremely susceptible to attack by hydrogen peroxide. However, the mechanism by which this substance impairs sperm quality apparently does not involve oxidative stress, because no increase in TBARS was observed. Despite the necessity of further studies to investigate how hydrogen peroxide negatively influences sperm function, the use of catalase and glutathione peroxidase, important hydrogen peroxide scavengers, appears to be an alternative to improve the quality of ram sperm.
|
The authors thank Nutricell for the media used in the experiment and CAPES for financial support.


