Spawning area and season of butterfly kingfish (Gasterochisma melampus), a large scombrid adapted to cooler temperate southern water
Tomoyuki Itoh A C and Shiro Sawadaishi BA National Research Institute of Far Seas Fisheries, Japan Fisheries Research and Education Agency, 5-7-1 Orido, Shimizu, Shizuoka, Shizuoka, 424-8633, Japan.
B Marine Fisheries Research and Development Center, Japan Fisheries Research and Education Agency, 2-3-3 Minato-mirai, Nishi, Yokohama, Kanagawa, 220-6115, Japan. [Retired].
C Corresponding author. Email: itou@fra.affrc.go.jp
Marine and Freshwater Research 69(1) 16-23 https://doi.org/10.1071/MF17077
Submitted: 18 March 2017 Accepted: 1 June 2017 Published: 21 August 2017
Journal compilation © CSIRO 2018 Open Access CC BY-NC-ND
Abstract
In the present study we investigated spawning of the butterfly kingfish (Gasterochisma melampus), a Scombridae species distributed in circumpolar temperate waters of the Southern Hemisphere in the Atlantic, Indian and Pacific oceans. Using data from 25 564 individuals collected by longline operations from 1987 to 1996, analysis of the gonadosomatic index, maturity based on oocyte size and the presence of hydrated eggs revealed that the spawning area was between longitude 85 and 130°W and latitude 28 and 41°S in the south-east Pacific Ocean, and that the spawning season was from mid-April to mid-July. Length–frequency data suggested that larger fish arrived and spawned earlier, whereas smaller fish did so later. The species has distinctive reproductive characteristics compared with other Scombridae: it produces large hydrated eggs 1.6 mm in diameter, sea surface temperatures in the spawning area were as low as 14–18°C and more than 80% of fish were female. The south-east Pacific Ocean may be the only (and is at least the major) spawning area of the species. Butterfly kingfish is a single stock that migrates to the Atlantic, Indian and Pacific oceans to feed and returns to the south-east Pacific Ocean to spawn.
Additional keywords: reproductive biology.
Introduction
There have been few studies of the ecosystems in the circumpolar temperate waters of the Southern Hemisphere at latitudes between 30 and 50°S extending across the Atlantic, Indian and Pacific oceans (Itoh and Sakai 2016). Representative large pelagic fish species distributed in this area are the southern bluefin tuna (Thunnus maccoyii), albacore (Thunnus alalunga), butterfly kingfish (Gasterochisma melampus), slender tuna (Allothunnus fallai), opah (Lampris guttatus), blue shark (Prionace glauca) and the porbeagle (Lamna nasus). Although commercially important species such as tunas have been studied well (e.g. Caton 1994; Murray 1994), there have been few studies of other species, such as the butterfly kingfish (but see Semba et al. (2013) for the porbeagle).
The butterfly kingfish is a large Scombridae species that reaches a fork length (FL) of 190 cm; the genus Gasterochisma has only this single species. The fish’s silver body is covered with large cycloid scales and has many distinctive morphological characteristics that differentiate it from other Scombridae (Collette et al. 2001). The general distribution of the species was reported by Warashina and Hisada (1972) and its sporadic occurrence off Argentina, Brazil and Hawaii has been reported (Ito et al. 1994; Rotundo et al. 2015). The fish’s physiology is interesting in that it has a brain heater (Carey 1982). The taxonomic position of the species, in terms of the evolution of endothermy, has been somewhat controversial (Block et al. 1993; Collette et al. 2001). There is a consensus that the species belongs to the Scombridae; however, studies based on morphology have hypothesised that it belongs to the independent subfamily Gasterochismatinae (Kohno 1984; Ito et al. 1994; Collette et al. 2001), whereas studies in molecular biology have hypothesised that it belongs to the subfamily Scombrinae, with tunas and mackerels (Block et al. 1993; Ito et al. 1994; Collette et al. 2001; Qiu et al. 2014).
In commercial tuna longline fishing, the butterfly kingfish has been a bycatch of southern bluefin tuna fishing by the Japanese fleet since the 1970s and is retained in the vessels for market, although the price received is not particularly high (Warashina and Hisada 1972). Because it appeared that the butterfly kingfish was distributed at high densities in the south-east Pacific Ocean, which is not a fishing ground for southern bluefin tuna, a research project was run for 10 years from 1987 by the Japan Marine Fishery Resources Research Center (JAMARC; now the Marine Fisheries Research and Development Center of the Fisheries and Education Research Agency) to explore the fishing grounds where butterfly kingfish could become the main target species. During these research cruises, the spawning area was located through an analysis of captured fish with ripe ovaries. The results have not been published before in an international peer-reviewed scientific journal, although they have been reported in domestic documents (e.g. Anonymous 1997). Herein we reanalyse the data, report on the spawning season, area and fish size and discuss the characteristic spawning ecology of this species.
Materials and methods
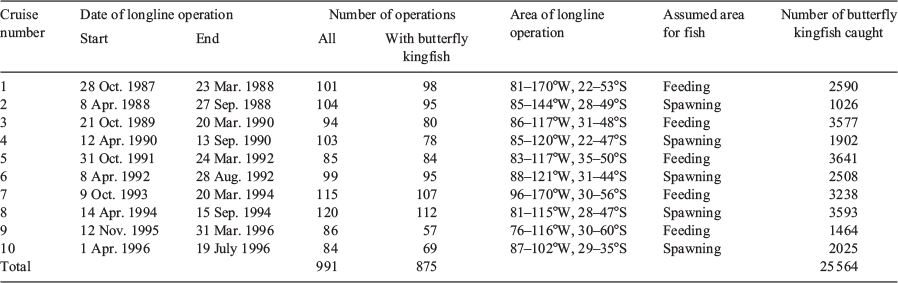
There were 10 longline research cruises from 1987 to 1996 (Table 1). One vessel, which was usually used for commercial tuna longline fishing, was chartered for each cruise and longline operations were conducted in the same way as for commercial operations. In the usual pattern, a total of 2400 hooks was used for each operation, and between 10 and 15 branch lines were used between two floats that set the hooks at depths of 80–220 m; this depth range was based on calculations of the catenary curve of the longline (Yoshihara 1951). Five of the ten cruises aimed to catch spawning fish. All applicable international, national and institutional guidelines for the care and use of animals were followed.
|
When an individual butterfly kingfish was caught, its FL (cm) and weight (kg) were measured and its sex identified by gonad examination. Both ovaries were weighed and their weights summed (in tens of grams) and maturation was classified into the following five stages by macroscopic visual observation of the fish while still on board: (1) immature, with no oocytes; (2) most advanced oocytes 0.2–0.3 mm in diameter; (3) most advanced oocytes 0.4–0.5 mm in diameter; (4) most advanced oocytes 0.8–1.1 mm in diameter or larger, translucent and easy to separate from the surrounding tissue, the ovaries are enlarged and the ovarian membrane thin and fragile; and (5) ovaries larger than Maturity Stage 1 but smaller than Maturity Stage 4, with an increased size of the ovarian cavity.
The presence of hydrated eggs was recorded arbitrarily. Seventeen females were found that had hydrated eggs in their ovaries; this is classified as Maturity Stage 4 and is strong evidence of spawning having taken place. Samples were cut from one ovary from five of these fish and preserved in 5% formalin in seawater on-board. Later, in the laboratory, the diameter of 100 randomly selected hydrated eggs from each sample was measured at a resolution of 0.1 mm using a micrometer under a dissecting microscope.
In the present data analysis, the gonadosomatic index (GSI) was used as the criterion of maturation rather than the aforementioned maturation stages used in on-board gonad observations because the on-board researchers differed from cruise to cruise and the classifications they made may have been inconsistent. The GSI was calculated as follows:
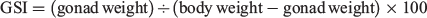
Judgment of maturity solely on the basis of the GSI is problematic in that the GSI cannot distinguish a spent ovary from an immature ovary; however, the GSI is effective if data on the size of the most advanced oocyte, or histological observations, are also used (Schaefer 2001). We attempted to evaluate maturity by approximating the relationship between GSI and the proportion of females in Maturity Stage 4 using a logistic model and a binomial distribution based on the dataset from 1994, the year in which the largest dataset was obtained. The GSI at which 80% of fish in Maturity Stage 4 met the criteria for maturity was used, with GSIs at which 20 and 50% of fish in Maturity Stage 4 met the criteria were used for comparison.
The spatial and temporal occurrences of mature fish were examined on the basis of the GSI by aggregating the data by 1° of latitude and 5° of longitude, or by month divided into one-third intervals (i.e. Days 1–10, Days 11–20 and Day 21 to the end of the month); the spawning area and season were then estimated accordingly. Furthermore, we analysed the relationship between length–frequency and the proportion of mature fish in the spawning area, compared the water temperatures (sea surface temperature at noon during the longline operation) inside and outside the spawning area, mapped the distribution of monthly catch per unit effort (CPUE; the number of butterfly kingfish caught by 1000 hooks) and examined the sex ratio by size. Analyses were performed using R, ver. 3.2.3 (R Foundation for Statistical Computing, Vienna, Austria).
Results
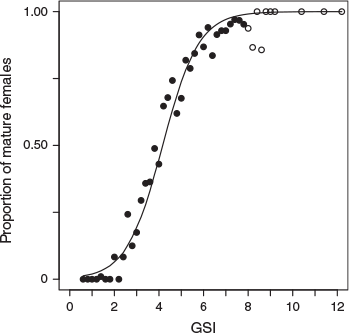
The parameter values for the logistic model that approximated the proportion of mature females (Maturity Stage 4) from the GSI were estimated as follows (Fig. 1):
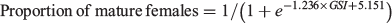
|
In the model, 80, 50 and 20% of females were mature at GSI = 5.3, 4.2 and 3.0 respectively. In the following description, GSI >5 is used as the maturity criterion; data for females with GSI >3 and >4 are presented in the figures for comparison.
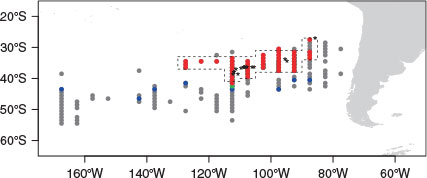
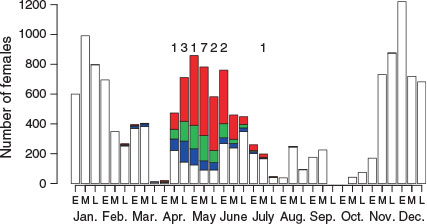
The spatial range of the butterfly kingfish caught extended from 76 to 169°W and from 28 to 54°S. Mature females (GSI >5) occurred in the northern part of this range, from 85 to 130°W and from 28 to 41°S (Fig. 2). Mature females were found from mid-April to mid-July and most frequently in mid-May (Fig. 3). This range was validated by the occurrence of ovaries containing hydrated eggs (see numbers in Fig. 3).
|
|
The smallest mature female with GSI >5 had a FL of 103 cm. In the mature group, substantial numbers of females were found with FL >120 cm (Fig. 4). It would not have been appropriate to estimate the length at which 50% of the population reached maturity (Schaefer 2001) from our data, because we did not know the number of females distributed in the spawning ground as a proportion of the total population. In our analysis of length–frequency according to one-third month intervals, larger fish were abundant in mid-April, when the mode of the FL was 165 cm; the fish then became smaller as the month progressed, reaching a FL mode of 135 cm in mid-June. The proportion of mature females was high in all size classes in the early period, from mid-April to late May; among larger fish (>150 cm FL) this proportion decreased from early June onward. Therefore, these observations may indicate that larger fish arrived and spawned early in the spawning season, whereas smaller fish spawned later in the season. However, the larger fish seemed to stay in the spawning area after spawning.
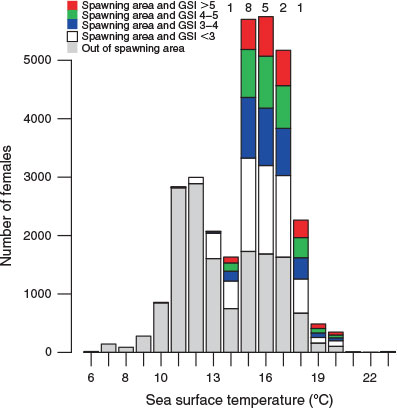
The temperature of the water where mature fish were found (as inferred from the sea surface temperatures during longline operations) was frequently in the range 14–18°C, whereas the temperature outside the spawning area was lower (Fig. 5). The suitability of this temperature range for spawning was further supported by the presence of fish with ovaries containing hydrated eggs (see numbers in Fig. 5).
|
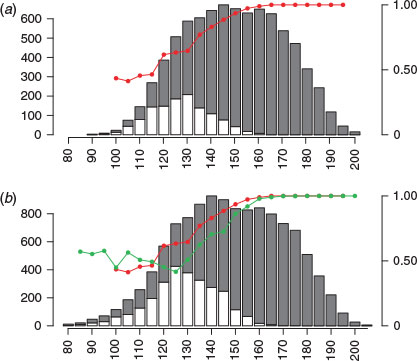
Sex was skewed towards female (85.3% in the spawning area and 77.1% outside the spawning area; Fig. 6). The percentage of females in the spawning area was ~50% up to 125 cm FL; it then increased linearly and reached almost 100% at 160 cm FL. The percentage of large males (e.g. 140 cm FL) was larger outside than inside the spawning area.
|
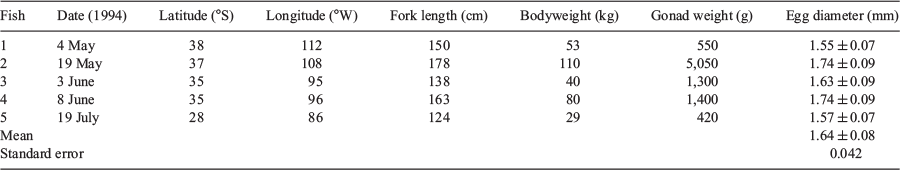
Examination of the monthly CPUE distribution showed that the area in which fish were caught moved to the spawning area from the south in April and were widely distributed in the spawning area in May, which may indicate fish movement (Fig. 7). The area in which fish were caught was concentrated in the eastern half of the spawning area in June and July, but note that longline operations were not conducted in the western half of the area in these months. Fish were still being caught in the spawning area in August and September. The distribution of mature females was similar to that of the CPUE. Hydrated eggs were spherical, with individual mean diameters ranging from 1.55 to 1.74 mm; the mean diameter of eggs from five individuals was 1.64 mm (Table 2).
|
Discussion
From our data, we determined the previously unknown spawning season and area of butterfly kingfish. The dataset contained information on a large number of fish sampled over 10 years, throughout most of the year, from an extensive area of the south-eastern Pacific Ocean. In some months there were areas where no surveys were conducted. However, because the survey covered 10 years and actively targeted butterfly kingfish, we consider it is highly unlikely that butterfly kingfish would have been distributed at high temporal and spatial densities in areas of the south-eastern Pacific Ocean missed by the survey.
It is possible that the spawning area and season were wider than those shown in the present study, and it is possible that they varied annually with the environmental conditions, stock status or both. Logbook data of Japanese longline fishery contain details of the butterfly kingfish catch since 1994. The data showed that, from June to August, there were some butterfly kingfish catches at additional times and in additional locations as bycatch of fishing targeting other tuna species in the area east to north-east of our delineated spawning area, although these catches were small in number and low in CPUE. Note that commercial fishing targeting butterfly kingfish in the south-eastern Pacific Ocean has not been developed further since these data were obtained; therefore, no further substantial data for analysis from this area have been obtained.
We found that females were dominant (>80% of fish). This female dominance is not limited to the spawning area or the south-east Pacific Ocean: it also occurs in the feeding grounds in the Atlantic, Indian and south-western Pacific oceans (T. Itoh, unpubl. data). This is unusual in the Scombridae, in which the number of males and females is generally equal, or is otherwise skewed towards a higher proportion of males in the group of large individuals (Schaefer 2001). However, in wahoo (Acanthocybium solandri) there is a skew towards females (76%; Zischke et al. 2013). In large pelagic species (although not Scombridae), the swordfish (Xiphias gladius) and the blue marlin (Makaira nigricans) show a skew towards females in larger fish, and the sex ratio differs by area (Arocha and Lee 1996; Shimose et al. 2012). In the aforementioned studies, the proportion of males was higher in the spawning areas, at lower latitudes, suggesting that the distribution was segregated by sex and that males were resident in the spawning areas all year round. However, in the case of butterfly kingfish, the percentage of males in the spawning area was smaller than that of females; males accounted for <20% of fish in both spawning and feeding grounds. There are several possible reasons for this difference: the original number of males may have been small, male mortality may be higher or males may have been distributed in places where we did not sample. Regardless, further studies are warranted.
The sea surface temperature in the spawning area of the butterfly kingfish, between 14 and 18°C, was the second lowest of the Scombridae, next to that of the Atlantic mackerel (Scomber scombrus), which was between 5 and 23°C (Studholme et al. 1999). In all species except the slender tuna in the tribe Thunnini, including the genera Thunnus, Katsuwonus, Euthynnus and Auxis, spawning takes place at sea surface temperatures of ~24°C or higher. Reproduction at high temperatures permits high spawning rates, high reproductive output and rapid growth of offspring (Schaefer 2001). The spawning areas of fish in these genera are restricted to high-temperature regions, even though the adults of some of these species, such as the southern bluefin tuna, are well adapted to cold water, permitting them to exploit productive temperate waters. The butterfly kingfish may choose a different strategy, namely having a body adapted to cold water, with a brain heater and a thick fat layer beneath the scales (Collette et al. 2001); by spawning in cooler water, it avoids competition with the Thunnini in their early life stages.
The spawning area we delineated is located in the subtropical convergence zone. The whole range of the butterfly kingfish caught in the south-eastern Pacific was in the area between the North Subtropical Front and the Subantarctic Front (Belkin and Gordon 1996). The spawning area was in the lowest latitudinal part of this area, where the highest sea surface temperatures occur during the month in which spawning was observed.
A review of egg diameter in 288 pelagic species found that the majority were in the range between 0.6 and 1.6 mm (Ahlstrom and Moser 1980). In most Scombridae species, including Thunnus and Scomber, the diameter of hydrated eggs is ~1.0 mm (e.g. Fritzsche 1978; Studholme et al. 1999; Margulies et al. 2007), although Japanese Spanish mackerel (Scomberomorus niphonius) has an egg diameter of 1.6 mm (Yokogawa 1995). Therefore, at 1.6 mm in diameter, the hydrated egg of the butterfly kingfish is one of the largest among the Scombridae.
It has been hypothesised that the butterfly kingfish branched early on from the scombrid stem and underwent its own specialisations in morphology and distribution (Kohno 1984; Block et al. 1993). Spawning at lower temperatures, a large egg and female dominance are likely to be mutually related factors in the reproductive strategy of butterfly kingfish: larval survival rates are increased by the production of large eggs to compensate for the slow larval growth at low temperatures, and female dominance compensates for the fact that, if eggs are larger, then fewer are produced. This is an additional specialisation of the species.
Butterfly kingfish are also distributed in temperate waters of the Atlantic, Indian and south-western Pacific oceans in the Southern Hemisphere (Warashina and Hisada 1972). Fish in these areas are much smaller than in the south-eastern Pacific Ocean, ranging from 80 to 160 cm FL, with a length mode of ~120 cm FL. Spawning, as evidenced by the finding of fish with fully developed ovaries, has not yet been shown to occur in these areas. There are areas other than the south-eastern Pacific Ocean that have sea surface temperatures in the range 14–18°C between April and June, but longline fishery data show that the number of butterfly kingfish caught in these areas is small and the CPUEs are low (T. Itoh, unpubl. data). It is highly likely that the south-eastern Pacific is the major spawning area of the species in light of the large catch number and high catch rate. Therefore, we consider that there is a single global stock of butterfly kingfish and that this fish migrates to the Atlantic, Indian and south-western Pacific oceans to feed, returning to the south-eastern Pacific Ocean to spawn. Note, however, that it is still unclear whether the spawning area is the only one or whether other (even small-scale) spawning areas exist. Examination of the ovaries of fish of this species from the Atlantic, Indian and south-western Pacific is warranted to resolve this question.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Acknowledgements
The authors thank the crew of the research vessels (F/V Sumiyoshi-maru 8, 18, 52 and 71) and the researchers (A. Ohno, S. Shigeno, A. Ueda, S. Miyagawa, M. Satani). The authors also thank the Marine Fisheries Research and Development Center of the Japan Fisheries Research and Education Agency, which allowed us to use their data. The authors are grateful to Dr Y. Tsuda and Dr Y. Semba for their assistance with the creation of the dataset. This study was supported by a grant from the Japan Fisheries Agency. The authors appreciate the work of the two anonymous reviewers whose comments improved the manuscript.
References
Ahlstrom, E. H., and Moser, H. G. (1980). Characters useful in identification of pelagic marine fish eggs. California Cooperative Oceanic Fisheries Investigations Reports 21, 121–131.Anonymous (1997) Report of the research for new fishing resource of butterfly kingfish in 1995 and 1996. Japan Marine Fisheries Research and Development Center. [In Japanese, title translated by T. Itoh].
Arocha, F., and Lee, D. W. (1996). Maturity at size, reproductive seasonality, spawning frequency, fecundity and sex ratio in swordfish from the northwest Atlantic. International Commission for the Conservation of Atlantic Tunas Collective Volume of Scientific Papers 45, 350–357.
Belkin, I. M., and Gordon, A. L. (1996). Southern Ocean fronts from the Greenwich meridian to Tasmania. Journal of Geophysical Research 101, 3675–3696.
| Southern Ocean fronts from the Greenwich meridian to Tasmania.Crossref | GoogleScholarGoogle Scholar |
Block, B. A., Finnerty, J. R., Stewart, A. F. R., and Kidd, J. (1993). Evolution of endothermy in fish: mapping physiological traits on a molecular phylogeny. Science 260, 210–214.
| Evolution of endothermy in fish: mapping physiological traits on a molecular phylogeny.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK3sXktVahsb4%3D&md5=729fe3850bbdffa3a57920a67ba5ad77CAS |
Carey, F. G. (1982). A brain heater in the swordfish. Science 216, 1327–1329.
| A brain heater in the swordfish.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DyaL387pvFCksw%3D%3D&md5=6fffcc85e86d969ef0b8933f40039746CAS |
Caton, A. E. (Ed.) (1994). Review of aspects of southern bluefin tuna biology, population, and fisheries. In ‘Interactions of Pacific Tuna Fisheries. Proceedings of the First FAO Expert Consultation on Interactions of Pacific Tuna Fisheries’, 3–11 December 1991, Noumea, New Caledonia. (Eds R. S. Shomura, J. Majkowski, and S. Langi.) FAO Fisheries Technical Paper 336, Vol. 2, pp. 296–343. (FAO: Rome, Italy.) Available at http://www.fao.org/docrep/005/T1817E/T1817E15.htm#ch11 [Verified 21 June 2017].
Collette, B. B., Reeb, C., and Block, B. A. (2001). Systematics of the tunas and mackerels (Scombridae). In ‘Fish Physiology: Tuna: Physiology, Ecology, and Evolution’. (Eds B. A. Block and E. D. Stevens) pp. 1–33. (Academic Press: San Diego, CA, USA.)
Fritzsche, R. A. (1978). ‘Development of Fishes of the Mid-Atlantic Bight – an Atlas of Egg, Larval and Juvenile Stages. Volume 5: Chaetodontidae Through Ophidiidae.’ (US Fish and Wildlife Service.)
Ito, R. Y., Hawn, D. R., and Collette, B. B. (1994). First record of the butterfly kingfish Gasterochisma melamps (Scombridae) from the north Pacific Ocean. Japanese Journal of Ichthyology 40, 482–486.
Itoh, T., and Sakai, O. (2016). Open-ocean foraging ecology of southern bluefin tuna Thunnus maccoyii based on stomach contents. Marine Ecology Progress Series 555, 203–219.
| Open-ocean foraging ecology of southern bluefin tuna Thunnus maccoyii based on stomach contents.Crossref | GoogleScholarGoogle Scholar |
Kohno, H. (1984). Osteology and systematic position of the butterfly mackerel, Gasterochisma melampus. Japanese Journal of Ichthyology 31, 268–286.
Margulies, D., Sutter, J., and Hunt, S. (2007). Spawning and early development of captive yellowfin tuna (Thunnus albacares). Fishery Bulletin 105, 249–265.
Murray, T. (1994). A review of the biology and fisheries for albacore, Thunnus alalunga, in the south Pacific Ocean. In ‘Interactions of Pacific Tuna Fisheries. Proceedings of the First FAO Expert Consultation on Interactions of Pacific Tuna Fisheries. Volume 2: Papers on Biology and Fisheries’, 3–11 December 1991. Noumea, New Caledonia. (Eds R. S. Shomura, J. Majkowski, and S. Langi.) FAO Fisheries Technical Paper 336, Vol. 2, pp. 188–206. (FAO: Rome, Italy.) Available at http://www.fao.org/docrep/005/T1817E/T1817E10.htm [Verified 21 June 2017].
Qiu, F., Kitchen, A., Burleigh, J. G., and Miyamoto, M. M. (2014). Scombroid fishes provide novel insights into the trait/rate associations of molecular evolution. Journal of Molecular Evolution 78, 338–348.
| Scombroid fishes provide novel insights into the trait/rate associations of molecular evolution.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2cXnslKjt74%3D&md5=22894895934bed1c55ea9f49507218c5CAS |
Rotundo, M. M., Júnior, T. V., and Van Sebroeck Doria, D. M. A. (2015). Report of the butterfly kingfish, Gasterochisma melampus (Scombridae: Gasterochismatinae) in southeastern Brazil. Pan-American Journal of Aquatic Sciences 10, 293–296.
Schaefer, K. M. (2001). Reproductive biology of tunas. In ‘Fish Physiology: Tuna: Physiology, Ecology, and Evolution’. (Eds B. A. Block and E. D. Stevens.) pp. 225–270. (Academic Press: San Diego, CA, USA.)
Semba, Y., Yokawa, K., Matsunaga, H., and Shono, H. (2013). Distribution and trend in abundance of the porbeagle (Lamna nasus) in the southern hemisphere. Marine and Freshwater Research 64, 518–529.
| Distribution and trend in abundance of the porbeagle (Lamna nasus) in the southern hemisphere.Crossref | GoogleScholarGoogle Scholar |
Shimose, T., Yokawa, K., Saito, H., and Tachihara, K. (2012). Sexual difference in the migration pattern of blue marlin, Makaira nigricans, related to spawning and feeding activities in the western and central North Pacific Ocean. Bulletin of Marine Science 88, 231–249.
| Sexual difference in the migration pattern of blue marlin, Makaira nigricans, related to spawning and feeding activities in the western and central North Pacific Ocean.Crossref | GoogleScholarGoogle Scholar |
Studholme, A. L., Packer, D. B., Berrien, P. L., Johnson, D. L., Zetlin, C. A., and Morse, W. W. (1999). Essential fish habitat source document: Atlantic mackerel, Scombrus, life history and habitat characteristics. NOAA Technical Memorandum NMFS-NE, 141, US Fish and Wildlife Service, Woods Hole, MA, USA.
Warashina, I., and Hisada, K. (1972). Geographical distribution and body length composition of two tuna-like fishes, Gasterochisma melamps Richardson and Allothunnus fallai Serventy, taken by Japanese tuna longline fishery. Bulletin of Far Seas Fisheries Research Laboratory 6, 51–75.
Yokogawa, K. (1995). Changes in ovarian egg number and egg diameter composition in Spanish mackerel Scomberomorus niphonius during the spawning season. Nippon Suisan Gakkai Shi 61, 689–694.
| Changes in ovarian egg number and egg diameter composition in Spanish mackerel Scomberomorus niphonius during the spawning season.Crossref | GoogleScholarGoogle Scholar |
Yoshihara, T. (1951). Distribution of fishes caught by the long line II. Vertical distribution. Nippon Suisan Gakkai Shi 16, 370–374.
| Distribution of fishes caught by the long line II. Vertical distribution.Crossref | GoogleScholarGoogle Scholar |
Zischke, M. T., Farley, J. H., Griffiths, S. P., and Tibbetts, I. R. (2013). Reproductive biology of wahoo, Acanthocybium solandri, off eastern Australia. Reviews in Fish Biology and Fisheries 23, 491–506.
| Reproductive biology of wahoo, Acanthocybium solandri, off eastern Australia.Crossref | GoogleScholarGoogle Scholar |




