307 TRANSGENIC OVINE EMBRYOS BY ARTIFICIAL INSEMINATION, IN VITRO FERTILIZATION AND INTRACYTOPLASMIC SPERM INJECTION
F. Pereyra-Bonnet A , A. Gibbons B , M. Cueto B , R. Fernández-Martín A and D. Salamone AA Lab. Animal Biotechnology, FA-UBA, Argentina;
B Lab. Reproduction in Small Ruminants, INTA, Bariloche, Argentina
Reproduction, Fertility and Development 21(1) 250-251 https://doi.org/10.1071/RDv21n1Ab307
Published: 9 December 2008
Abstract
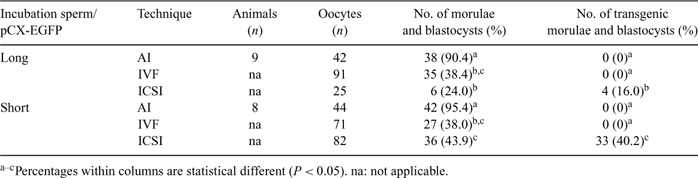
Nowadays, transgenesis in animals constitutes an important tool for pharmacological protein production and livestock improvement. In 1971 Brackett first described that heterologous DNA can be introduced into a mammalian oocyte using sperm cells as vectors. We evaluated the capacity of AI, IVF and ICSI to produce transgenic embryos, in ovine, using sperm that had been exposed to a pCX-EGFP plasmid in Long and Short incubation protocols. The pCX-EGFP plasmid contains an enhanced green fluorescent protein gene (egfp) under the chimeric cytomegalovirus-IE-chicken β-actin enhancer-promoter control. Sperm/pCX-EGFP incubation was carried out by Long Incubation (2 h at 17°C in 200 μL of SFM medium: 100 mL contains glucose 1.2 g, Na citrate 1.0 g, EDTA 0.4 g, Citric acid 0.3 g, Trizma 0.6 g) and Short Incubation (5 min at 0–5°C in 10–100 μL of extender medium: 100 mL contains Na Citrate 2.8 g and EDTA 4 mg). For AI, Merino sheep (n = 17) were superovulated and inseminated with fresh semen (200 millions sperm/sheep) from eight Merino rams. The embryos were recovered by flushing the uterine horns by standard procedures. In IVF and ICSI, slaughterhouse oocytes were fertilized with frozen/thawed sperm. IVF was carried out in Brackett-Oliphant medium with 5 mm of caffeine, 20 IU mL–1 of heparin with 20 million sperm mL–1 during 5 h in an atmosphere of 5% CO2 in air. In ICSI, the spermatozoon was immobilized by breaking its tail and injected into MII oocytes. Immediately the oocytes were activated by incubation in TALP-HEPES with 5 μm ionomycin for 4 min, cultured in TCM199 for 3 h and transferred to a droplet of 1.9 mm DMAP for 3 h. Maturation and cultivation conditions were determined by standard operating procedures. All embryos were exposed to blue light (488 nm) to determine the percentage of morulae/blastocysts showing green fluorescence. Results are shown in Table 1. Statistical analysis was done by Fisher test. AI and IVF were able to produce a high percentage of morula and blastocyst stage, but were unable to produce transgenic embryos. In contrast, regardless of the sperm/plasmid incubation protocol, high percentages of transgenic morulae and blastocysts were always obtained by ICSI and the highest rate was achieved with Short Incubation (P < 0.05). In order to demonstrate ICSI-Short incubation embryo viability, two-day-old non-selected fluorescence embryos (n = 45), were transferred into the oviducts of five surrogate mothers. Pregnancy was diagnosed at day 25 (2/5; 40%), and one normal female lamb was recently born (1/5; 20%). In conclusion, our results show that in ovine, ICSI seems to be the only method for producing transgenic embryos using sperm cells as vectors. In addition the offspring born confirm the viability of these embryos.
|


