184 EFFECTS OF X-RAY ELECTROMAGNETIC RADIATION ON BOVINE OOCYTE DEVELOPMENT
W. E. Snyder A , J. T. Aaltonen A , H. D. Sigal A and N. M. Loskutoff AThe Bill and Berniece Grewcock Center for Conservation & Research, Omaha’s Henry Doorly Zoo, Omaha, NE, USA
Reproduction, Fertility and Development 22(1) 250-251 https://doi.org/10.1071/RDv22n1Ab184
Published: 8 December 2009
Abstract
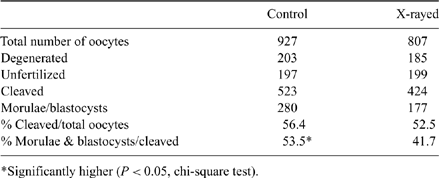
This study examined the effects of X-rays on bovine oocytes used for in vitro embryo production. In recent years, airport security has become more stringent and the use of X-ray screening may be required for all items, which could be problematic when transporting oocytes. Ionizing radiation such as X-rays are high-energy particles that can penetrate into the nucleus of a cell and cause single and double-strand breaks in the DNA chain. Oocytes exposed to X-ray radiation may have DNA damage affecting maturation, fertilization, and embryonic development. Bovine ovaries obtained from a local abattoir were used to collect immature oocytes by follicular aspiration. The oocytes were divided into a control group and an experimental group then placed in maturation medium. The oocytes in the experimental group were placed in a hospital X-ray machine with a single exposure of 100 mA, 120 kVp for 0.3 s. This exposure challenged the oocytes with a comparable, calculated amount of mrems equivalent to a single screening from an airport X-ray machine (30 mrems). After X-ray exposure, the experimental group was cultured with the control at 37°C and 5% CO2. After 18 h, cryopreserved bovine sperm were thawed and processed using BoviPure (Nidacon International, Mölndal, Sweden) density gradient centrifugation and the oocytes were inseminated with 1 × 106 sperm in fertilization medium at 37°C and 5% CO2. After 24 h, both groups were transferred to 50-μL droplets of G1 medium (Vitrolife, Göteborg, Sweden) at 37°C and 6% CO2 and both were transferred into G2 medium after 72 h. Development of the oocytes was scored after an additional 72 h. Results were based on 2 criteria: the percent cleaved (presumptive fertilization) and the percent cleaved that developed to morula and blastocyst stages. The results indicated no significant difference in the percentages of oocytes cleaving after insemination for the control and experimental groups (P = 0.403, chi-square test). However, there was a significant increase (P = 0.037, chi-square test) in the percentages of morulae and blastocysts developing in the control (non-irradiated) v. experimental (X-rayed) groups. The outcome of this study cautions on the potential damage caused to bovine oocytes used for embryo production in vitro when exposed to X-ray electromagnetic radiation during routine air transport screening operations.
|


