Nutrition, feeding and laying hen welfare
W. L. Bryden A C , X. Li
A C , X. Li  A , I. Ruhnke
A , I. Ruhnke  B , D. Zhang A and S. Shini A
B , D. Zhang A and S. Shini A
A University of Queensland, School of Agriculture and Food Sciences, Gatton, Qld 4343, Australia.
B University of New England, School of Environmental and Rural Science, Armidale, NSW 2350, Australia.
C Corresponding author. Email: w.bryden@uq.edu.au
Animal Production Science 61(10) 893-914 https://doi.org/10.1071/AN20396
Submitted: 6 July 2020 Accepted: 9 September 2020 Published: 23 February 2021
Journal Compilation © CSIRO 2021 Open Access CC BY-NC-ND
Abstract
The relationship between nutrition and welfare is usually considered to be a direct result of supplying the hen with adequate quantities of feed and water. This simple notion of freedom from hunger and thirst belies the fact that nutrients play a pivotal role in the body’s response to challenges whether they relate to ambient temperature, gastrointestinal health, pathogen exposure, metabolic disorders, or social and mental stress. In all instances, maintaining homeostasis and allowing for physiologic response is dependent on an adequate and balanced nutrient supply. It is accepted that most laying hens are fed a complete diet, formulated commercially to provide the required nutrients for optimal health, egg production and welfare. In other words, the laying hen, irrespective of her housing, does not experience hunger or thirst. However, despite adequate nutrient and water supply, certain senarios can significantly affect and alter the nutrient requirements of the hen. Furthermore, the chemical composition and also the physical form of feed can significantly contribute to prevent or treat welfare and health conditions and is, therefore, a highly relevant tool to ensure and maintain an adequate welfare status. Therefore, this review takes a broader perspective of nutritional welfare and considers the nutrition of hens managed in different production systems in relation to nutritional physiology, gut microbiota, stress, metabolic disorders and feeding management.
Keywords: amino acid, calcium, diet, energy, gut health, gut microbiota, immunity, inflammation, metabolic disorder, nutritional physiology, pasture, phosphorus, stress.
Introduction
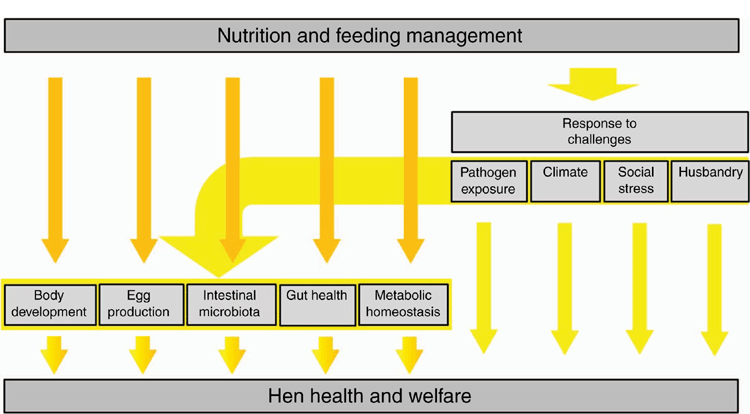
The supply of feed to laying hens is the most significant cost of egg production and diets are formulated to meet the nutrient requirements of the hen at least cost. The modern bird has been selected for high productivity (Underwood et al. 2021) and if this genetic potential is to be met without compromising welfare, feeding and housing must be optimal. Diets for laying hens are formulated to ensure maximum egg output. In general, nutritional recommendations for laying hens consider breed and housing conditions. However, as most husbandry situations are complex, adjustments to the recommended daily energy and nutrient intakes may need to be made to meet specific production objectives (Fig. 1). As conditions change in the hen house or on the range, including stage of production and season of the year, the amount of each nutrient required will change and, in some cases, dietary imbalances occur that impair productivity and compromise welfare.
The five freedoms are commonly used as a framework to evaluate welfare (see Webster 2001, 2016). It is obvious that freedom from hunger and thirst involves nutrition and feeding, but it is less obvious that the other four freedoms, including freedom from discomfort, freedom from pain, injury and disease, freedom to express normal behaviour, and freedom from discomfort and stress have nutritional components that also must be satisfied. In this regard, feed provides the nutrients and substrates essential for the maintenance of homeostasis. Moreover, there is increasing evidence that diet and the gastrointestinal microbiota combine to play a central role not only in physical but also mental wellbeing, with obvious implications for bird health and welfare (see Villageliũ and Lyte 2017; Kraimi et al. 2019).
Concerns of laying hen welfare have focussed on housing systems, environmental enrichment and other management strategies. The major variable for the laying hen is the management system (housing) under which it is maintained (Edwards and Hemsworth 2021). Understandably, significant amounts of money have been invested by egg producers to upgrade caged-hen facilities and to develop colony-caged, barn, and free-range systems to improve hen welfare and meet consumer expectations. The aim of egg production is to achieve the best bird performance and feed utilisation, while providing adequate hen welfare. Therefore, for some husbandry conditions and specific production objectives, adjustments of the recommended daily energy and nutrient intakes need to be made to meet these demands and manage associated challenges. However, there is limited information available on the nutritional welfare of laying hens. Within this context, the nutrition of hens managed in caged and non-caged production systems is reviewed. Nutrient requirements are discussed in relation to nutritional physiology, stress, metabolic disorders and feeding management.
Diet composition and nutrient availability
Arguably, more is known about poultry nutrition than the nutrition of most other species and this has contributed to the success of the poultry industry. With chicken meat production, nutrition research has sought to optimise growth and development, while the aim of laying hen nutrition research has been to maximise egg production or reproductive performance. However, to achieve maximum egg output, bird health and welfare must be optimised. The role of diet composition in achieving this is discussed in this section. The growing pullet is discussed in a later section.
Energy
Laying hens require energy for maintenance, determined by metabolic body mass, and for production (Leeson and Summers 2009). The energy requirements for production are primarily for daily egg production and body mass increase between the onset of sexual maturity and the attainment of mature bodyweight (BW), including feather growth (Tauson and Svensson 1980). Genotype and housing also mediate energy requirements along with several variable factors, including hen activity, ambient temperature and plumage condition that change during the production cycle (Peguri and Coon 1991, 1993).
The housing system is a major determinant of hen behaviour and activity. Tiller (2001) suggested that the maintenance energy required for hens housed in barn and free-range conditions is higher than for caged hens. While several studies have determined that hens kept in non-cage systems, such as organic, barn or free-range production, require an additional 10–15% more energy, hens may compromise egg production so as to meet their energy demands and, therefore, the expected egg output is reduced (Tiller 2001; Aerni et al. 2005; Leenstra et al. 2012; Leinonen et al. 2012; MacLeod 2013).
Energy requirement adjustments are also required for changes in ambient temperature (GfE 1999). An important component of this adjustment is plumage condition, which provides insulation and allows hens to regulate energy loss. A loss of feathers due to abrasion, feather pecking or moulting has to be compensated with increased levels of dietary energy to maintain body temperature, hen health and welfare. Moreover, additional dietary energy is required for the dissipation of body heat when the ambient temperature exceeds 22°C, as frequently occurs in many areas of Australia and Asia.
Laying hens are able to adjust their feed intake, to some degree, according to their energy requirements. Hens reduce feed intake when dietary energy increases. In contrast, if dietary energy content is too low, layers are unlikely to increase their feed intake sufficiently to overcome the deficit (Leeson and Summers 2009). Throughout the production cycle, the energy requirements of laying hens remain relatively constant as the demand for body maintenance increases with age and this is offset by a decrease in egg production. Nevertheless, hen age is positively correlated with egg weight and egg mass, resulting in reduced eggshell thickness and, subsequently, a reduced number of sellable whole eggs in older flocks (Harms et al. 1982).
Protein and amino acids
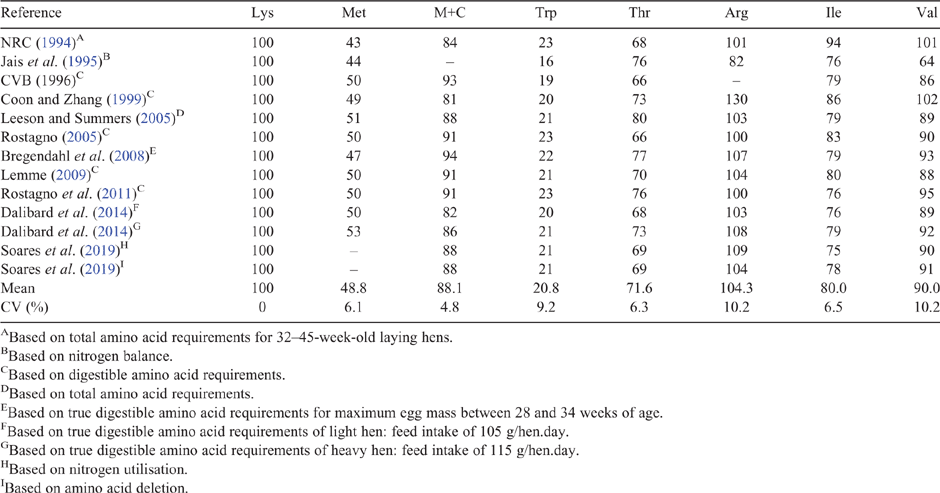
While protein is a critical component of a layer diet, the balance of essential amino acids is crucial when formulating diets (Leeson and Summers 2009). The ideal protein concept was developed in the 1950s, as a way to define the essential amino acid requirements of birds for protein accretion and maintenance (Scott et al. 1969). Although sulfur amino acids (methionine and cystine) are considered first limiting amino acids in most commercial laying hen diets, lysine is used as the reference amino acid for this concept (NRC 1994). The requirements for all other indispensable amino acids are expressed as a percentage of lysine and calculated values for caged hens, as shown in Table 1; presumably, free-range hens have a similar ideal amino acid ratio. The values reported span some 20 years and show some variation due to genetic changes in laying stock and different methods of calculation. Synthetic or crystalline amino acids are added to diets to ensure that the ideal amino acid profile is achieved, thus permitting lower levels of protein to be fed. In some countries, synthetic amino acids are banned in organic poultry farming and this could result in the feeding of imbalanced diets, especially in relation to methionine, with a negative impact on bird health and welfare (Burley et al. 2016; van Krimpen et al. 2016).
Dietary content of balanced protein affects both egg number and egg size. For each 0.05% increase in sulfur-containing amino acids above 0.23%, the egg weight of layers increases by 0.7 g and the addition of methionine results in an almost linear increase of egg weight (Calderon and Jensen 1990; Waldroup and Hellwig 1995). However, if the concentration of these amino acids is at the lower limit of the requirement, reduced egg number is observed. Hens will also try to maintain amino acid requirements by increasing their feed intake, resulting in an overall increase in energy consumption. Hens maintained in free-range conditions have higher crude protein requirements for maintenance, most likely reflecting increased feed intake to meet the greater energy expenditure (Leenstra et al. 2014; de Almeida Brainer et al. 2016).
Specific amino acids have been shown to modifiy animal behaviour in many species. In caged laying hens, increasing lysine and methionine concentrations ameliorate the negative effects of stocking density on cannibalism and mortality (Balnave and Robinson 2000). Khattak and Helmbrecht (2019) demonstrated that the tryptophan requirements of hens housed in colony cages at peak production were higher than current NRC (1994) recommendations. In barn systems, dietary supplementation with tryptophan was shown to alleviate stress in layers, and be an effective treatment for hysteria (Laycock and Ball 1990). Hens fed additional tryptophan generally show decreased stress, and when also supplemented with insoluble fibre, have a reduced occurrence of cannibalism (Mousavi et al. 2018). Obviously, tryptophan availability in layer diets has important implications for hen welfare.
Generally, hens housed in cages are at a lower risk of infectious (Noormohammadi 2021) and parasitic diseases (Groves 2021) than are hens housed in free-range systems. The increased disease burden experienced by free-range hens may increase their requirement for amino acids to support an upregulation of the immune response (Klasing 2007). However, some metabolic conditions appear predominantly in caged hens. Low protein and high energy diets have been implicated in the development of the fatty liver haemorrhagic syndrome (FLHS) in caged layers, as discussed below.
Calcium and phosphorus
Calcium (Ca) and phosphorus (P) are key nutrients in layer diets. These minerals have many essential roles in metabolism, especially in bone development and eggshell formation (Etches 1987; Bar 2009). In the 25 years since the NRC (1994) prescribed the poultry requirements for these minerals, the genetics of layers have changed dramatically, housing and management have improved and, subsequently, diet requirements have changed. Within this context, it is likely that the altered requirements for Ca and P has direct implications for hen health and welfare. We have recently reviewed Ca and P metabolism in laying hens and concluded that the concentrations of both nutrients in layer diets could be substantially decreased without a negative effect on the bird (Li et al. 2017). Furthermore, bone quality, including mineral composition, appears not to be affected by the degree of range use when an aviary system is available in the shed (Kolakshyapati et al. 2019; Sibanda et al. 2020b).
Cereal grains and their by-products are the major ingredients in layer diets. Cereals have low concentrations of P predominantly bound to phytate. Diets, therefore, require supplementation with inorganic P and phytase to provide adequate available P to the hen (Li et al. 2016a). Phosphorus availability can also be impaired by elevated dietary Ca concentrations, which increase the pH of digesta and as a result, decrease P absorption and retention (Li et al. 2016b). Furthermore, the high gastrointestinal pH caused by high dietary Ca concentrations decreases pepsin activity in the proventriculus and gizzard, thus reducing digestibility of protein. In contrast, high dietary phytate concentrations bind Ca, forming non-absorbable complexes (Bryden et al. 2007). High plasma P concentrations also decrease gastrointestinal absorption of Ca. Therefore, an optimum dietary Ca to P ratio is essential for bone health, egg production and, ultimately, hen welfare. Vitamin D3 regulates Ca and P absorption, storage and mobilisation, but excess availability of vitamin D3 provides no additional advantage for egg production (Li et al. 2017). In general, hens can tolerate a wider range of Ca concentrations, when dietary P concentrations are high (Li et al. 2017).
Most of our understanding of Ca and P metabolism has been determined with broilers, and similar research is required for layers. Layers have a much higher Ca requirement and Ca metabolism is also modulated by oestrogen. Currently, total dietary Ca concentrations are used for diet formulation but, for accurate formulation, availability of different Ca sources is required. This will improve the precision of feed formulation and bird welfare. Moreover, as Neijat et al. (2011) concluded following their study of Ca and P dynamics of layers housed in conventional and enriched cages, nutrient utilisation, along with welfare, should be assessed when comparing different housing systems.
Vitamins, minerals and feed additives
Minimum dietary concentrations of vitamin that prevent clinical signs of deficiency may not support optimum performance and welfare (Leeson et al. 1995; Weber 2009). This is due to continuous genetic improvement of hens, changes in the nutrient availability of feed ingredients, and ongoing changes in hen management, which all contribute to the demand for micronutrients, especially vitamins. Intensive egg production increases metabolic, social, environmental and disease stresses, resulting in increased requirements for vitamin and trace minerals. For example, vitamin E, vitamin C, selenium and zinc are antioxidants that limit radical damage and oxidative stress and help sustain health and longevity (Attia et al. 2016). Improved egg quality can be achieved if supra-nutritional levels of vitamin E are added to layer diets. However, very little research has been conducted on the implications of adding high concentrations of one vitamin for the absorption and metabolism of other vitamins. Weber (2009) reviewed the requirements of laying hens for vitamins and concluded that high producing layers require 5–10 times the minimum requirements determined by the NRC (1994), so as to sustain productivity and maintain welfare. The possibility of suboptimal vitamin and trace-mineral intake can be avoided by adding sufficient quantities with approporiate safety margins, as a premix to diets.
Along with a premix, poultry diets are commonly supplemented with a variety of other feed additives to improve the nutritional characteristics of the diet, hen performance, and egg quality. These include nutritional additives (amino acids, vitamin and minerals, prebiotics, and probiotics), products to add nutritive value to the egg (so called ‘designer eggs’), and products that affect the visual characteristics of eggs by improving yolk colour. The various yolk pigments added to layer diets are to meet consumer expectations and have no relevance to hen health or welfare.
The use of feed enzymes for improving the digestibility of nutrients and dietary energy content has become common practice over the past 30 years. Dietary enzymes lower feed costs and benefit the environment. For example, exogenous feed enzymes such as phytase increase dietary P availability, thus reducing the need to add inorganic P to diets, and, subsequently, decrease the amount of P excreted. As indicated above, many factors affect phytase efficacy, including phytase source and concentration, and importantly, the dietary Ca : P ratio. Phytase is most efficacious in diets containing low available P and adequate Ca concentrations (Li et al. 2016b, 2017), whereas carbohydrases, such as xylanase and β-glucanase, increase the available energy level of the diets (Mathlouthi et al. 2002).
Mixtures of additives are sometimes used strategically to improve bird health and welfare. Probiotics, which are live bacterial cultures, along with prebiotics (a substrate for gut bacteria, usually fibre) fall into this category and are being used increasingly in layer diets to improve gut health. This is discussed further in the section on the gut microbiota and microbiome. Combinations of feed enzymes, organic acids and essential oils can reduce the incidence of gut impaction and increase nutrient digestibility (including non-starch polysaccharides) in free-range hens with access to abundant pasture (Iqbal et al. 2018, 2019a).
Water
Water is the most important nutrient as it has a central role in all aspects of metabolism (Leeson et al. 1976) and is critical for bird welfare. It is difficult to access water requirements, as water intake is modulated by age, feed intake, stage of production, ambient temperature, water temperature and quality (Leeson et al. 1976). The intake of water and feed are directly related; hens that drink less water will also consume less feed, and, subsequently, egg production declines. Presumably, current high producing laying hens have higher metabolic demands for water than did earlier layer strains. Water intake is a sensitive indicator of bird health and, therefore, monitoring water intake of a flock is a useful guide to changes in bird welfare (Leeson and Summers 2009). Hens must have access to water of high quality at all times. However, there are situations during transport and illness when birds may be deprived of water (Rault et al. 2016).
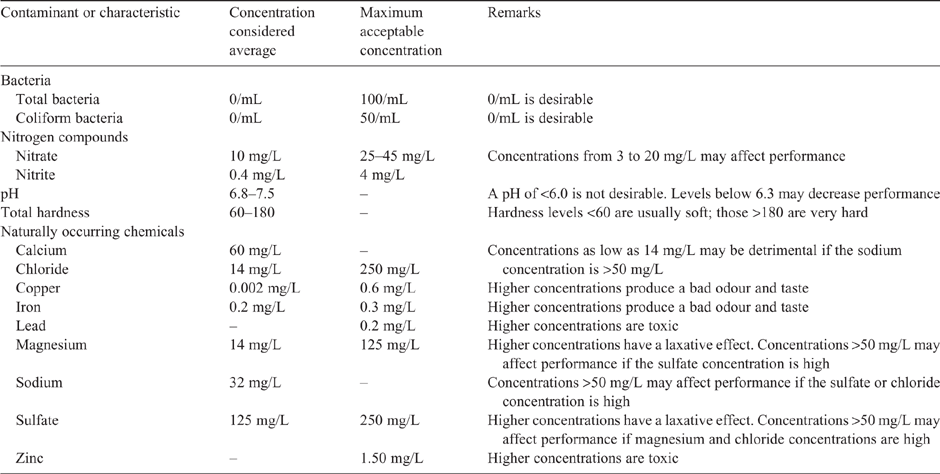
Poultry farm water in Australia can come from many sources, including surface water such as dams, lakes and rivers, but also underground water or treated town water; all vary greatly in quality (DAFF 2009). Water quality should be tested, and if concentrations of nitrites, chloride (Cl) and other minerals or bacterial contamination exceed the values in Table 2, the water needs to be treated accordingly (Leeson and Summers 2009). High concentrations of total dissolved solids or high salinity, predominantly high concentrations of Ca, magnesium (Mg) and sodium (Na) salts, are the most common cause of harmful effects in poultry, and guidelines are shown in Table 3. High concentrations of Na, Cl or Ca in drinking water require changes in dietary nutrient composition to avoid excessive mineral intake. Most of the literature on Ca and Mg in water for laying hens was generated in the 1970s or earlier (Scott et al. 1969) and, therefore, may not be appropriate for modern high-producing laying hens. For example, there is a lack of research on the impact of mineral concentrations in drinking water on mineral metabolism or bone quality. Therefore, there is a need to determine the effect of water pH and mineral (Ca, Mg and sulfate, SO4) concentrations on the welfare of hens. Moreover, the contribution of drinking-water concentrations of Ca and P to the homeostasis of these two minerals in laying hens (discussed above) has been largely neglected.
|

|
In many countries, underground water is a common source of drinking water for poultry and is usually high in NaCl, Ca, Mg and potassium (K; Balnave and Scott 1986). For example, concentrations of Na and Cl as high as 570 and 2000 mg/L respectively, have been recorded in Australian bore water (Zhang 1993). This can be of concern since saline drinking water has been shown to have an adverse effect on eggshell quality, while having little effect on feed intake, egg production or egg weight (Zhang 1993). Similar effects of saline drinking water on eggshell quality were reported in Israel (Yoselewitz et al. 1993), Iran (Pourreza et al. 1994) and Germany (Khalafalla and Bessei 1997). However, research conducted in the USA (Maurice 1989; Damron 1998) did not find reduced eggshell quality when hens drank saline drinking water. Different responses may be due to genetic sensitivity to NaCl, as Yoselewitz and Balnave (1990) showed different sensitivities among six Australian layer strains. Furthermore, the degree of sensitivity observed among strains also existed among hens of the same strain. A reduction in bicarbonate ions in the shell gland (Balnave et al. 1989; Roberts and Balnave 1992), and not Ca availability (Brackpool et al. 1996), appears to explain the poor shell quality in hens receiving saline drinking water. Therefore, measures that increase blood bicarbonate concentrations may improve eggshell quality. Supplying ammonium bicarbonate, ascorbic acid and various zinc supplements to hens receiving saline drinking water improved shell quality (Zhang 1993). Presumably, the beneficial response to these supplements also had a positive effect on hen welfare as it would have affected acid-base homeostasis.
Gut microbiota and microbiome
Microbial activity in the gastrointestinal tract has a significant influence on bird metabolism and wellbeing. The relationship between host and microbiota can be either commensal, symbiotic or pathogenic. Microbiota refers to the community of microorganisms within the gastrointestinal environment, while microbiome describes the collective genomes of all these microbial species. The intestinal microbiota is dynamic as a result of interactions with the host, diet and environment, including the bedding material microbiome (Pan and Yu 2014); dietary components are the main modulators of the gut microbiome. Nutritional interactions that occur between the intestinal wall and gut microbes play an important role in modulating gut colonisation by either beneficial, commensal or pathogenic bacteria, as they are consistently competing for nutrients and gut attachment sites. It has been established that the gut microbiota can have a profound effect on the development of the immune system and, thus, strongly influence optimal health and productivity (Shang et al. 2018). Understanding these relationships is important for promoting hen health and productivity.
Many benefits have been attributed to gut microbiota for both nutrition and protection of poultry health, including a reduction of pathogen colonisation (competitive exclusion), production of B and K vitamins, volatile fatty acids (acetic acid, butyric acid and propionic acid), organic acids (e.g. lactic acid), and antimicrobial compounds (e.g. bacteriocins), induction of non-pathogenic immune (Shang et al. 2018) and endocrine (Villageliũ and Lyte 2017) responses, and modulation of behaviour and welfare (Kraimi et al. 2019). Many gastrointestinal bacteria are able to metabolise otherwise indigestible β-linked dietary polysaccharides, which are then fermented to volatile fatty acids (Annison et al. 1968). These can be utilised by the host as energy and carbon sources (Koutsos and Arias 2006; Tellez et al. 2006); butyrate is an important energy source for the gut wall. Gastrointestinal microbiota also contribute to the host’s nitrogen metabolism as some bacterial protein can be a source of amino acids for the host (Metges 2000). However, the degree to which microbes might influence avian nutrition and metabolism is poorly understood. It is doubtful that all of the end products of microbial metabolism will be available to the host, as most microbial activity occurs in the caeca and despite the bird’s ability to cause retrograde movement of digesta to sites of absorption in the avian gut. A high inter-individual variation of intestinal microbiota can be observed, which may result in a high individual variation of nutrient metabolism or feed utilisation (Sonnenburg and Bäckhed 2016).
The gut microbiome of all animals can be affected by diet composition, including the major nutrients, antinutritive factors and feed additives, especially probiotics (Bajagal et al. 2016). Different dietary interventions directed at the gut microbiota are now used to enhance hen production and reduce infection risk, while maximising feed utilisation. There is recent evidence supporting the use of probiotics for laying hens to improve productivity and egg quality (Mahdavi et al. 2005; Mátéová et al. 2009), prevent gut and reproductive infections (Shini et al. 2013), and improve performance and bone mineralisation (Yan et al. 2019). Probiotics contain live bacterial cultures, but how they induce their effects remains unclear. However, as we unravel the interactions between diet and the gut microbiota, it should be possible to refine layer diets and further improve hen wellbeing.
Nutritional physiology
The laying hen life cycle can be conveniently divided into pre- and post-attainment of sexual maturity. In this section, the impact of nutrition during pullet growth and development, and during egg production, is disussed. Many authors, including Janczak and Riber (2015) and Jongman (2021), have discussed the many factors that determine the development of the laying hen, and came to the conclusion that pullet rearing has a significant impact on subsequent egg production and welfare of the mature laying hen.
Growth and development
A strong correlation exists between body development during rearing and laying performance and also the ability to extend lay beyond 80 weeks of age (Pottgüter 2016). Key targets of pullet rearing include reaching specified BWs at specific ages, flock uniformity of at least 85%, a well developed digestive tract, and the establishment of good feeding behaviour patterns. Feeding techniques and the nutrients provided are key factors in achieving these targets. Factors that adversely affect pullet welfare may compromise body composition and reduce flock uniformity. Control of BW and flock uniformity are, therefore, crucial aspects for the successful growth and development of the laying hen.
It is common practice to feed layer chickens in four phases, reflecting the different developmental stages of the pullet, for optimum pullet development. A starter diet is offered for the first 4 weeks post-hatch, with a relatively high energy content (12.3–12.4 MJ/kg), ideally as crumbles, and with an increased Ca (1.05–1.10%) concentration to promote skeletal growth (Gous and Morris 2001; Frikha et al. 2009). Depending on the chick’s BW, the starter diet may be extended to week 6 post-hatch. Grower diets are usually fed from 4 to10 weeks of age and contain slightly less energy (11.9–12.0 MJ/kg) and Ca (0.9–1.10%) than do starter diets. Adequate P concentration must be supplied, since inadequate P intake during the first half of pullet rearing results in an increased prevalence of osteoporosis in mature hens; ~95% of skeletal development is completed during the first half of pullet rearing (Pines and Reshef 2015). At 10 weeks of age, pullets are able to increase voluntary feed intake and should be offered low energy density diets to reduce the incidence of overweight birds and increase gut holding capacity. At this age, a diet to promote gastrointestinal organ development is introduced. Feeding a low energy mash (11.5 MJ/kg) encourages pullets to increase voluntary feed intake, stimulates gizzard activity, and increases digestive enzyme secretion (Yokhana et al. 2016). For this purpose, the crude fibre concentration of the developer diet may be as high as 5.5% (Pottgüter 2016). This is important for caged hens, and critical for free-range hens that may experience uncontrolled pasture intake (Ruhnke et al. 2015b).
Dietary fibre and an adequate feed particle size are key factors for gastrointestinal development (Hetland et al. 2003a; Svihus et al. 2004; Frikha et al. 2011). Coarse feed particles are especially important for the gizzard, pancreas and liver development (Engberg et al. 2002; Svihus 2011; Ruhnke et al. 2015a). Heavier gizzard and pancreas mass are associated with increased bile secretion and pancreatic enzyme activity respectively (Engberg et al. 2004; Svihus et al. 2004).
The onset of maturity in pullets begins at ~16 weeks of age. Commencement of egg production is determined by a combination of age, BW, body fat content, and increased light exposure, together with the influence of oestrogen. The duration of the pre-lay or transition period is ~10 days and, during this time, medullary bone develops, providing labile Ca storage while pullets continue to gain weight. Moderate increases of Ca, P and protein concentrations in the pre-lay diet during the transition period allow hens to meet the metabolic adjustment to lay, while maintaining overall feed intake (Leeson and Summers 2009; Thiele 2012) in conjunction with a dietary Ca supply of 2–2.5%. This diet should be offered for a further 10–14 days or until the flock reaches 2% hen house production (Coelho 2001; Pottgüter 2016).
Young layers of low BW are unable to consume sufficient feed to reach or maintain peak production. These birds use their body reserves in an attempt to meet genetically determined egg output, resulting in poor hen condition and reduced flock uniformity. The use of pre-lay diets helps offset this problem and is, therefore, essential in all production systems for optimum hen production and welfare.
Egg production and eggshell quality
Among the many factors that determine egg production and eggshell quality, with corresponding implications for hen welfare, nutrition is an important modulator (Roberts 2004). Egg weight is positively correlated with hen weight; for every 100 g live pullet weight, egg weight increases by 0.7 g during the onset of lay, with lasting, but less pronounced effects, for the duration of the laying period (Bouvarel et al. 2011). Moreover, at a given hen weight, egg weight can be increased by promoting dietary energy intake, resulting in an average of 0.96 g egg weight gain per 42 kJ additional energy intake (Jalal et al. 2006; Bouvarel et al. 2011).
Nutrition can be a tool to control egg size in aging flocks. By manipulating the diet to limit the total protein concentration to 13%, and restricting the amount of sulfur-containing amino acids, a reduction of egg size can be achieved (Calderon and Jensen 1990; Leeson and Summers 2009). Gradually decreasing the energy density of the diet during lay can control egg size, while compensatory feed intake will balance for the overall requirements. However, while the minimum demand for the essential fatty acid linoleic acid is ~1% of the diet, it is common practice to feed layers with a Phase 1 diet that contains up to 3% linoleic acid, and gradually decrease its concentration to 1% by the end of lay (Grobas et al. 1999a, 1999b, 2001; Pottgüter 2016). This gradual reduction of linoleic acid will result in consisitent egg size without promoting compensatory feed intake.
The lipid composition of egg yolk reflects dietary fatty acids and is of interest for the production of speciality or designer eggs as nutraceuticals or functional foods (Surai and Sparks 2001). Omega-3-enriched eggs with increased quantities of α-linolenic acid, eicosapentanoeic acid and docosahexaenoic acid or conjugated linoleic acid (CLA) are of interest to health-conscious consumers. For each 1% CLA added to the hen’s diet, 50 mg CLA is transferred into the egg. In order to achieve the the target of 300 mg omega 3 fatty acids per egg, the feed should contain 10% flaxseed (Caston and Leeson 1990). Interestingly, the health benefits from consuming polyunsaturated fatty acids are well documented for humans and, presumably, the hen receives similar benefits but this does not appear to have been documented. The concentration of cholesterol in the egg (180 mg/egg) remains relatively constant.
Dietary lipids and bile acids influence carotenoid absorption and, therefore, yolk colour. However, as nearly all β-carotenes are converted into vitamin A, its contribution to yolk pigmentation is negligible (Hencken 1992). As a result, layers accumulate mainly xanthophylls such as lutein, zeaxanthin and capsanthin (Nys 2000; Surai et al. 2001; Sinanoglou et al. 2011). The accumulation of xanthophylls is crucial to meet consumer expectations of egg yolk colour. The most common plant sources used for yolk colouring include corn, corn gluten, lucerne, marigold and capsicum (Britton 1996; Lootens et al. 2004). The ingestion of pasture by free-range hens also increases the deposition of lutein and zeaxanthin in the egg yolk significantly, resulting in darker yolk colour (Karunajeewa 1978; Iqbal et al. 2018).
Yolk mottling is associated with a low intake of carotenoids, and also a lack of yellow pigments while red pigments are increased (Leeson and Summers 2009). This occurs predominantly in spring due to variable pasture availability and as a result of prolonged feed storage. Other factors that cause yolk mottling include the ingestion of gossypol, tannins, nicarbacin, mycotoxins, coccidiosis and viral diseases (i.e. Newcastle disease, infectious bronchitis) that all impair xanthophyll absorption and deposition, resulting in pale yolks (Karunajeewa et al. 1984). The smell and taste of eggs can be negatively influenced by the accumulation of aldehydes or trimethylamines. This limits the dietary inclusion of canola to less than 20%, the use of fishmeal to less than 1% and also needs to be taken into account when adding betaine or choline to layer diets.
The quality of albumen is rarely influenced by nutrition but the administration of gossypol can result in pink albumen coloration. The availability of ascorbic acid (vitamin C) has positively influenced albumen height and the Haugh unit value in heat stressed hens. This is most likely due to a reduction in free radicals that would otherwise damage the quaternary and tertiary structure of the albumen protein (Benabdeljelil et al. 1990; Ajakaiye et al. 2011).
Eggshell quality remains an ongoing concern for the egg industry. Dietary Ca and P concentrations are the key determinants of eggshell breaking strength and shell weight (Hamilton 1982). Vitamin D3 activity, which regulates Ca uptake, is modulated by hydroxylases in both the liver and kidneys. In older birds, reduced α1-hydroxylase activity in the kidney is one of the key factors considered responsible for reduced Ca utilisation (Li et al. 2017). Interference with Ca deposition may also occur due to excess Cl intake in drinking water, as discussed above. The availability of Ca for eggshell formation depends on its particle size and gizzard activity. Approximately 65% of dietary Ca should be provided as coarse (>1–2.5 mm) particles (Rao and Roland 1990; Zhang and Coon 1997; Bouvarel et al. 2011).
Modern layers utilise 2 g Ca daily for eggshell formation, with a third of the Ca being provided from medullary bone and remainder coming directly from the gastrointestinal tract (Nys and Guyot 2011). Bone quality is negatively correlated with egg mass and eggshell quality (Riczu et al. 2004; Kim et al. 2005). Therefore, providing a pre-lay diet is essential to support medullary bone formation and is a major contributing factor to ensure adequate eggshell quality throughout production (Elaroussi et al. 1994; Summers and Leeson 1994) This reduces the incidence of bone fractures and osteoporosis (discussed below), thus maintaining hen health and welfare.
Stress and metabolism
Stress is a complex phenomenon and the body’s response to stressors that may be internal or external. It is an attempt to maintain homeostatic equilibrium. This is achieved through complex interactions of cell metabolism and the neuroendocrine and immune axes (Husband 1995). It is in this way that nutrients are partitioned in the body to meet differing metabolic demands (Husband and Bryden 1996; Klasing 2007). Extra energy and micronutrients may be required to meet the biological costs of an immune or neuroendocrine response to stressors (such as non-infectious disease or metabolic disorders, exposure to feed contaminants, temperature fluctuations and social interactions). If these nutritional needs are not satisfied, hen welfare will be compromised.
Commercial layers often encounter social and environmental stressors. Layers kept in different housing systems have similar nutrient needs for egg production; however, exposure to stressors and disease will vary among housing systems and influence nutrition requirements (Lay et al. 2011). Determination of stress is difficult, but there are physiological and behavioural indices of stress in laying hens that can be quantified and used to assess hen well being (Scanes 2016; Tilbrook and Fisher 2020). Nutritional stressors including fasting, feed restriction, and dietary nutrient deficiency, can influence plasma concentrations of corticosterone, other glucocorticoids, corticosteroid binding protein, epinephrine, and norepinephrine, and together with the blood heterophil to leukocyte (H : L) ratio, can be helpful to assess hen welfare (Shini et al. 2010). In this section, some of the stressors experienced by laying hens will be discussed in relation to changes in nutrient responses by the hen.
Immunity and inflammation
The acute phase response is a key defence mechanism of the immune system and involves increased production of pro-inflammatory cytokines, eicosanoids and other molecules that mediate a variety of physiological and metabolic changes, to create unfavourable conditions for pathogens (Kogut 2009). Inflammation is one of the body’s first non-specific responses to infection, and overactive immune responses can cause chronic inflammation, which promotes co-infection with other pathogens. In laying hens, the acute phase response can result in unspecific clinical signs such as decreased feed intake and activity, thermogenic shivering, unphysiologic feather lifting, and increased resting times. Adaptive immunity, in contrast, is mediated by lymphocytes, and responses are specific to the pathogen. These include the production and activation of antibodies, as well as cytotoxic killing through the release of antimicrobial substances.
The diet of a laying hen can affect the development, maintenance and response of the immune system. While nutritional deficiencies of linoleic acid, vitamin A, iron, selenium, and several of the B vitamins impair immune function and increase susceptibility to infectious diseases, the requirements of these nutrients are elevated during the acute or chronic stages of disease and should be increased as required (Klasing 1998). Importantly, nutrients or compounds that modulate the immune response are active at concentrations well above the established NRC requirements for growth and productivity (Koutsos and Klasing 2014). Some nutrients and compounds, including fatty acids, vitamin E, vitamin A, and vitamin D, have anti-inflammatory properties, while others have immunomodulatory properties, such as carotenoids, flavonoids, vitamin C and various long-chain polyunsaturated fatty acids (Meriwether et al. 2010; Korver 2012; Wils-Plotz and Klasing 2017).
Immune responses are not only energy demanding but require an investment of various nutrient resources. This is achieved by diverting or partitioning nutrients away from other productive activities such as ovulation and growth (Klasing 2007). The costs of different modes of immune competence (innate and adaptive immunity) can be estimated by examining the amount of protein needed for the cellular and secretory processes involved in each component of immunity relative to all other processes in an adult bird at maintenance (Koutsos and Klasing 2014). Diets with inadequate levels of protein or amino acids can compromise immune function and increase infection risk.
Heat stress
Heat stress can be chronic or acute and is a complex phenomenon involving interactions among bird genetics, ambient temperature, relative humidity, feather cover, water and feed intake and acclimatisation; it has been the subject of Australian research for many years (Yeates et al. 1941; Lee et al. 1945; Balnave 2004). Together, these factors demonstrate the difficulty of predicting responses to heat load with different nutritional mitigation strategies. Heat stress can be associated with reduced egg production and egg quality due to decreased feed and water intake and, subsequently, reduced nutrient utilisation as well as increased energy demands for metabolic (cooling) activity (Lin et al. 2006; Lara and Rostagno 2013). Moreover, heat-wave conditions or abrupt changes in heat load increase hen mortality (Daniel and Balnave 1981). Obviously, heat stress has welfare implications for laying hens, but these can be aggravated or mitigated by diet.
Many strategies are available to alleviate heat stress (Lin et al. 2006) and nutrition is always an important consideration. The ability of a nutritionist to provide a single set of nutrient specifications to satisfy all possible interacting factors is challenging and expensive (Balnave 2004). Obviously, the immediate need is to overcome the impact of reduced feed intake and adjust the energy and protein density. However, there is much conjecture in the literature on which is the appropriate approach (Balnave and Brake 2005). It is advisable that hens subject to heat stress are fed a diet where energy density is primarily achieved by adding fat rather than protein to the diet, as the metabolic heat produced to digest protein exceeds the energy required to digest fat. Furthermore, offering a pelleted diet allows for a higher energy density and can also increase the feed intake significantly. However, sudden changes of the physical form of the diet should be avoided to prevent diet refusal. It is, therefore, crucial that the farm management is well prepared and hens are adapted to heat conditions in advance. Specific diet recommendations may also include a higher dietary arginine : lysine ratio (Balnave and Brake 2002). Applying additives to cooled drinking water may be the best approach to ensure additive intake.
Glatz (2001) reported that 59–66-week-old layers housed at 30°C consumed more feed and produced eggs with better shell quality when provided with drinking water at 5°C, 10°C or 15°C, compared with uncooled water at 30°C. An improvement in feed consumption and egg production was also observed due to the supplementation of vitamin C in the drinking water (Khan and Sardar 2005). Betaine has been also suggested to alleviate the adverse effects of heat stress on laying hens. Betaine acts as an osmolyte (Saeed et al. 2017) and has been shown to improve egg production, egg quality traits and immune indices (Attia et al. 2016). Acute heat exposure causes oxidative damage and dietary supplementation with selenium for laying hens improved feed intake, feed efficiency, egg production and quality, and antioxidant status (Habibian et al. 2015). Most importantly, the hen’s panting activity results in repiratory alkalosis that impairs Ca carbonate deposition in the eggshell. Therefore, maintaining the electrolyte balance of those hens is the key factor to keep eggshell quality at an acceptable level and several commercial products are available for drinking water modification (Roberts 2004).
Moulting
Moulting is a normal physiological event in the life cycle of laying hens, during which plumage is replaced with new feathers, and is accompanied by reduced feed intake, BW loss and suspended reproduction (Yousaf and Chaudhry 2008). In contrast, induced moulting, a practice that can have significant hen welfare concerns as discussed by Glatz and Tilbrook (2020), is determined by economic considerations. Feed and water withdrawal was the classical method to induce moulting, but has serious welfare implications and is no longer permitted. Moult is now induced by feeding low energy diets such as barley or oats at least 40–60 g/bird.day with 1% Ca for up to 3 weeks (Glatz and Tilbrook 2020). Other practices that have successfully induced moult include feeding high concentrations of zinc, aluminium or potassium or the oral administration of thyroxine (Teixeira et al. 2006; Bass et al. 2007). Induced moulting is a reaction to stress and it demonstrates that perturbations in nutrient supply or balance can modify metabolism and impair bird welfare.
Metabolic disorders and feed contaminants
Metabolic disorders, often of complex aetiology, are any condition that reduces wellbeing and productivity and is associated with some abnormality in metabolism. Exceptions are that the condition is not of infective origin, does not arise from a specific genetic defect, or is not simply a dietary deficiency. In other instances, poor bird performance may result from feed containing anti-nutritive factors or toxin contamination.
Fatty liver haemorrhagic syndrome
Fatty liver haemorrhagic syndrome (FLHS) is a metabolic disease of laying hens that is characterised by excessive accumulation of fat in the liver and abdominal cavity, liver rupture and haemorrhage, and was first described by Couch (1956). FLHS occurs in commercial layers in high production and is frequently the major cause of death in otherwise healthy flocks and has been described in Australia on several occasions (Grimes 1975; Neill et al. 1975; Shini et al. 2019). In a recent Queensland study (Shini et al. 2019), it was obseved that the condition was a major cause of mortality in caged hens but not in free-range hens. The difficulty with the condition is that it can be diagnosed only at necropsy. These cases are acute and it is not possible to gauge the welfare implications of chronic FLHS where a bird has an episode but recovers.
The condition has been reviewed by several authors (Butler 1976; Annison 1983; Squires and Leeson 1988; Julian 2005; Crespo and Shivaprasad 2003) who have agreed that nutrition plays a role in the aetiology of FLHS. Excessive liver fat infiltration appears to occur in laying hens under conditions of excess energy intake due to increased feed intake coupled with little exercise and high circulating oestrogen concentrations. Husbandry factors that appear to predispose hens to FLHS include dietary ingredients that stimulate lipogenesis, genetics, environmental conditions, and stress in general. Dietary factors other than excessive caloric intake, including rapeseed meal (Pearson et al. 1978), aflatoxin (Hamilton and Garlic 1971; Bryden et al. 1979) and chelated minerals (Branton et al.1995), have been suggested as possible factors in the aetiology of FLHS.
A fatty liver is normal in laying hens, as increased hepatic lipogenesis and deposition is essential for lipid transfer to the egg during yolk formation (Annison 1983). Most previous research on FLHS has concentrated on liver lipid accumulation rather than the factors that precipitate hepatic haemorrhage. We have recently shown, using an oestrogen model, that an inflammatory response due to disturbed lipid metabolism is involved in the pathogenesis of FLHS (Shini et al. 2020a, 2020b). It appears that, under field conditions, high-producing laying hens with elevated circulating oestrogen concentrations, when exposed to an inflammatory insult, are more susceptible to the development of FLHS.
Osteoporosis
Osteoporosis is a metabolic condition of laying hens associated with the progressive loss of structural (trabecular and cortical) bone, resulting in increased susceptibility to fractures (Campbell 2020). There is no single cause of osteoporosis, and multiple factors are involved in the pathogenesis of osteopenia (Whitehead 2004). It is caused by a combination of genetic, environmental and nutritional factors affecting bone health. Birds are unique and produce medullary bone, which serves as a labile reserve of Ca for eggshell formation (Nys Le Roy 2018). Hence, bones of laying hens undergo continuous and rapid remodelling (Johnsson et al. 2015). The amount of medullary bone builds up shortly before first oviposition, and the structural bone content of the hen declines over the remainder of the laying period, increasing the risk of fracture, which is a significant welfare issue.
Osteoporosis is not a Ca deficiency per se, but may involve the inability of hens to metabolise sufficient Ca because of inadequate dietary Ca, vitamin D3 or P supply (Whitehead and Fleming 2000; de Matos 2008). Structural bone loss is also accelerated by the relative inactivity of conventionally caged birds (Newman and Leeson 1998). Good nutrition can help minimise osteoporosis, but is unable to prevent it. The timing of nutritional intervention is critical; if dietary changes are left until sexual maturity, the effect will be seen in medullary bone. Adequate dietary amounts of Ca, P and vitamins D and K are particularly important during the rearing period, before the excess resorption encountered during the laying period (Fleming et al. 1998). Feeding limestone in particulate form benefits skeletal and eggshell quality (Fleming 2008). Since both Ca and P are withdrawn and replaced during the shelling–non-shelling cycle, P is also required to rebuild medullary bone. Phosphorus deficiency-induced osteoporosis in high producing cage layers is called cage-layer fatigue (Riddell et al. 1969). Vitamin D3 deficiency will contribute to the development of osteoporosis in laying hens because it affects Ca and P metabolism (Whitehead and Fleming 2000).
Biogenic amines
Feed-borne biogenic amines are formed in animal protein meals by microbial decarboxylation of amino acids, and have important physiological roles, but are also potential toxins. The most common biogenic amines include histamine, cadaverine, tryptamine, putrescine, spermidine, tyramine and phenylalanine (Feddern et al. 2019). An extensive survey of the concentrations of putrescine, cadaverine and histamine in Australian fish and animal by-product meals was undertaken by den Brinker et al. (2003), who found the highest concentrations in fish meals. Most published work regarding the effects of biogenic amines has focussed on broilers where poor performance was often associated with gizzard erosion and proventriculus ulcers due to gizzerosine (Sugahara 1995; Barnes et al. 2001).
There is a lack of published data regarding the physiological role or toxicity of biogenic amines in laying hens. The supplementation of laying hen diets with putrescine significantly improved eggshell quality. It was recommended that, when eggshell quality is a concern, there may be economic benefits from adding small amounts of dietary putrescine (Smith et al. 2000). Interestingly, putrescine has been shown to be a key factor in the mode of action of vitamin D3 in the chick intestine, which may explain the effects observed (Shinki et al. 1991).
Mycotoxins, anti-nutritive factors and other feed toxins
Poultry diets can be contaminated with various toxic compounds (see Cheeke 1998), including mycotoxins, pesticides, organochlorine compounds, petroleum hydrocarbons, minerals, and anti-nutritive compounds found in some feed ingredients, all of which can affect bird performance and welfare, but most toxins are rarely encountered. Plants may contain secondary plant metabolites and anti-nutritive factors such as protease inhibitors (soy, lupins, peas), lectins (soy, lupins, peas), tannins (faba beans, peas, cotton seed), phytate (cereal grains, legume seeds, soy, canola), glucosinolates (cruciferes especially brassica spp), saponins (lucerne, clovers, legume seeds), alkaloids (lupins), cyanogenic glycosides (cassava, linseed) and phytoestrogens (soy, legumes). The effects of these compounds are mitigated by heat during processing and accounted for in diet formulation by restricting the inclusion level of the responsible feed ingredient or supplementing the diet with an appropriate feed enzyme.
Mycotoxins, or fungal secondary metabolites, are of concern because they can contaminate feed throughout the feed supply chain and often a feed commodity is contaminated with more than one toxin of the major toxins, namely, aflatoxin, fumonisins, deoxynivalenol, ochratoxin, zearalenone (Bryden 2009). The major risk to poultry from mycotoxin exposure is chronic mycotoxicoses that result in impaired productivity, with hens experiencing reduced egg production, poor egg and shell quality, and increased morbidity and mortality, and can be difficult to diagnose (Bryden 2012a). Importantly, immunity is often suppressed before other impacts of the toxic insult are apparent. This can reduce the effectiveness of vaccination and has obvious welfare implications. The mycotoxicoses that are likely to be encountered in Australia have been described (Bryden 2012b) and because of Australia’s dry climate, mycotoxin related problems occur much less frequently than they do in North America. The reliance on corn in North American poultry diets, as opposed to wheat and sorghum in Australia poultry diets, is also a contributing factor.
Feeding management
Birds are maintained in husdandry systems where much of the opportunity to determine what they eat and how much has been replaced by scientifically formuated diets. This ensures freedom from hunger and thirst, but eliminates the pleasure of seeking and anticipating food. Moreover, hunger and thirst are basic motivating forces and central to maintaining life. It is, therefore, not surprising that the body maintains very complex, multilayered control of both feed and water intake. Some of the factors modulating feed and water intake are discussed in this section, along with their management.
Feeding and behaviour
Birds select their feed on the basis of particle size, colour and form, with a preference for coarse, yellow to orange coloured structures (Amerah et al. 2007). If the feed particles are too fine (e.g. <500 μm), feed uptake is reduced, respiratory signs due to dust exposure may appear, and feed wastage increases, resulting in suboptimal nutrition and reduced bird welfare (Safaa et al. 2009). Care needs to be taken that the ability of hens to select and favour certain feed particles does not result in an unbalanced nutrient intake. This may occur with hens that are not beak trimmed, as their sharp tips are more capable of picking individual grain ingredients, compared with the more scooping feeding behaviour of hens with shorter beaks (Portella et al. 1988; Glatz 2003; Persyn et al. 2004; Iqbal et al. 2019b). While pellets prevent feed selection and unintentional ingredient seggregation, they result in a high-density nutrient intake. In contrast, mash diets require hens to spend more time feeding. Hens allocating a greater portion of their time budget to feeding are less aggressive and express significantly fewer unwanted behaviours such as severe feather pecking and stereotype behaviour (Vilariño et al. 1996; Huber-Eicher and Wechsler 1998; Hartini et al. 2002; van Krimpen et al. 2008). A decreased incidence of severe feather pecking can also be achieved using low energy diets and feed with higher levels of non-starch polysaccharides (van Krimpen et al. 2008). It appears that the time spent feeding is crucial to satisfy the hens’ pecking and foraging behaviour that would otherwise be redirected into inter-hen aggression. Beak trimming is used extensively to reduce the incidence of aggressive pecking and can affect feed and water intake if poorly executed (Glatz and Underwood 2020).
Inter-hen aggression depends also on the availability of resources such as feeder space. At least 14-cm trough length per hen is required to ensure adequate feed intake, without any negative impact on flock behaviour (Sirovnik et al. 2018). However, hen behaviour in non-cage systems can result in the development of flock subpopulations that demonstrate individual preferences regarding physical location and access of resources (Ruhnke et al. 2019). For example, while 10-cm feed chain per hen was provided in a three-tier aviary system, the majority of hens accessed the feeder area located at the tier close to the ground for an average of 7.29 h/hen.day. In comparison, the feed resources on top of the aviary system were utilised on average 4.29 h/hen.day (Sibanda et al. 2020a). These observations question current feed strategies where all feeder chains are run for the same amount of time and offer the same diet quantity. Precision feeding that considers the outcomes of hen behaviour such as body condition, feather cover and metabolic needs of hen subpopulations might allow a more efficient use of feed resources while providing more optimised support to under or high performing hens (Fig. 2). As indicated above, not only hen location but also physical appearance including feather cover can be an important determinant of energy requirements. Moreover, diet composition can influence plumage cover (Hetland et al. 2004).
Associations between hen behaviour and nutrient utilisation are observed. Low efficiency hens demonstrated more walking, pacing, escape and aggressive behaviours and had reduced feather cover compared with high-efficiency hens, which slept and rested more frequently (Braastad and Katle 1989). In conclusion, feed can provide a powerful tool to encourage desired hen behaviour critical for laying hen welfare. This is especially relevant when flock longevity is of increasing concern, where layers perform until 100 weeks of age or for the duration of a second laying cycle.
Housing system and feed intake
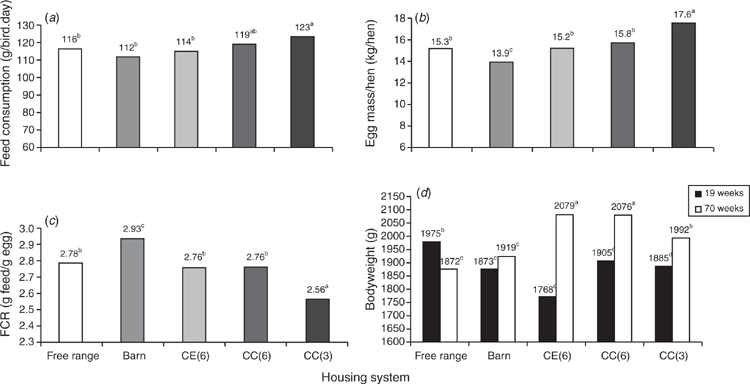
In an Australian study, laying hens, all of the same strain and hatch, were maintained in five different housing systems during egg production (Stewart et al. 2006) and data from the study are shown in Fig. 3. All birds received the same diet during lay and the nutrient density was calculated for an intake of 110 g/bird.day. Data on feed intake showed that the more productive hens had the highest consumption, such as, for example, hens in conventional cages (3 hens/cage); in contrast, the more active the hens (hens in free-range and barn systems), the higher was the feed conversion ratio (Stewart et al. 2006). Hens kept in cages in a controlled environment had the highest feed intake and a moderate FCR, resulting in the highest BW at 70 weeks. There were significant differences in feed consumption among birds in different housing systems, with hens in barn having the lowest feed intake and hens in three-bird cages consuming more feed. Average FCR demonstrated that birds in all other systems utilised feed significantly better than birds in the barn, and this may reflect the higher incidence of cannibalism in this system (Stewart et al. 2006). Birds in free-range housing utilised feed at the same level as hens caged in a controlled environment (6 hens/cage) or in conventional cages without environmental control (3 hens/cage). Presumably, the way that hens were fed affected the amount of feed consumed or wasted. Hens in free-range and barn environments were fed from automatic chain feeders (operating 6 times per day); hens in cages were all fed ad libitum, with feed troughs being topped up manually as required (Stewart et al. 2006). These factors may have affected the outcomes observed.
|
Feed form
Most of laying hens are fed complete diets formulated commercially that can be offered as mash, crumbles or pellets. Reasons to choose one above the other may include the feed production cost, availability, and physical factors such as feed particle size, number and shape that influence the uniformity and, subsequently, the segregation of feed ingredients (Axe 1995). The physical form of feed can result in unbalanced nutrient uptake and poor performance if not managed adequately (Hetland et al. 2003a). However, offering mash to layers is most common due to its beneficial effects on feeding time and, subsequently, hen behaviour (as outlined above) and lower production costs. Heavy BW is a concern for laying hens, due to its direct impact on egg size, eggshell quality and health. The benefit of feeding mash on lowering the energy density of the diet is, therefore, of advantage to producers who are able to deliver the feed via automated feeding systems that prevent particle selection. However, egg producers whose hens are exposed to uncontrolled pasture intake, and who do not have the technical advantage of automated feed delivery, may benefit from offering pelleted feed. The advantages of feeding pellets include reduced dust and microbial exposure, reduced feed-ingredient segregation, minimised feed wastage and a higher nutrient density (Behnke 2001), and prevention of hens from feed selection.
While pelleting or crumbling is standard for meat birds, the increased energy intake associated with a nutrient dense pellet is a disadvantage for most commercial laying hens, resulting in over-consumption and unfavourable BW gain (Safaa et al. 2009). However, in a recent Australian survey, 41 free-range egg farmers indicated that the majority fed mash (30.0%), followed by pellets (17.5%; Singh et al. 2017). Other feed practices included feeding whole grains (15.0%), coarse ground (25.0%), and fine ground feed (17.5%). Interestingly, 80% of farmers offered a complete diet, 12.5% used combined feeding, and 7.5% choice feeding. Regardless of the physical form provided, it is important to ensure that the diet contains adequate insoluble fibre to promote gizzard activity (Hetland et al. 2005).
Hens are able to select feed to meet their nutrient requirements, but need to be offered a reasonable choice (Emmans 1977; Summers and Leeson 1978). It is crucial to consider specific feed characteristics such as feed colour, taste and feed location, and to provide only a limited number of choices (Pousga et al. 2005). While successful implementation of choice-feeding has been demonstrated under research conditions, it has not been validated in large commercial flocks. So, while the feed choices offered need to be carefully considered, it cannot be assumed that hens will always select the feed ingredients required for optimum egg performance. For example, the curiosity of hens and the ability to choose their feed intake can result in over consumption of pasture and lead to grass impaction (Ruhnke et al. 2015b). This can severely affect calculated diet intake and, in such instances, offering pellets with a high nutrient density is recommended.
The structure of a diet affects gut health and hen performance (Yegani and Korver 2008). The grinding action of the gizzard is mostly responsible for the positive impact of large feed particles on improved gastrointestinal epithelial barrier function, increased ileal nutrient digestibility, and an overall better hen performance (Svihus and Hetland 2001; Svihus 2006). A well developed gastrointestinal barrier function is crucial to minimise exposure, colonisation and damage from pathogens and decrease the likelihood of concurrent diseases, thus ensuring optimum gut function and absorption (Röhe et al. 2014). Particle size, therefore, affects food and product safety. Large feed particles promote gastrointestinal development and, as a consequence, reduce concentrations of Salmonella spp. and Clostridium spp. in the avian intestinal tract (Bjerrum et al. 2005; Huang et al. 2006; Santos et al. 2008; Yegani and Korver 2008; Moen et al. 2012).
Particle size research has been predominantly conducted with broilers and recommendations exist for these birds, but not layers (see Amerah et al. 2007). However, it is known that physical feed characteristics such as feed particle size affect egg quality. Eggshell density was found to be higher in hens that were fed finely ground particles than in hens that were fed a coarse diet (Hafeez et al. 2015a, 2015b). With broilers, ensuring that at least 20% of the feed particles are larger than 1.5–2.0 mm is considered to be beneficial for gut health, but with layers, inclusion of whole cereals is also practised (Wu et al. 2004; Amerah et al. 2009; Svihus et al. 2010). The use of whole grain reduces feed processing and feed transportation costs. Other structural components commonly included in diets or available to the bird (especially free-range hens) are shell grit, hay, silage, vegetables, pasture, insects, and harvested grass (Steenfeldt et al. 2007; Singh et al. 2017). So as to prevent nutritional imbalances, these substituted feed components need to be analysed for their nutritional value and the quantities ingested need to be taken into account for diet formulation (Bennett and Classen 2003). With wheat or corn being a common diet component, whole-grain inclusion can provide a valuable structural component and improve hen health, nutrient digestibility and hen performance (Hetland et al. 2003b; Wu et al. 2004).
Feed and water delivery
Feed- and water-delivery systems must meet the physiologic feeding and drinking behaviour of the hen. The structure of the feed trough and drinker should prevent any contamination with excreta, bedding material, dirt or other foreign bodies. Devices that minimise feed waste are increasingly being used and include automated feeding chains, automated feeding pans, but also manually gravity-filled troughs in smaller egg-production systems (Singh et al. 2017). Automated feeding systems are commonly run five to eight times a day, providing an audio stimulus encouraging hens to feed. This is especially important in non-cage systems where flock subpopulations may prefer to spend their time on the range, in a winter garden or in other areas of the production system (Sibanda et al. 2020a). The restriction of feed intake or access can result in hen frustration and increase the likelihood of aggression and feather pecking, but also limit nutrient intake of top egg producing hens. Therefore, feeding ad libitum and formulating diets according to an estimated feed intake are recommended (Singh et al. 2017). In a survey of Australian egg producers, 77.5% of farmers fed hens ad libitum (Singh et al. 2017).
For biosecurity, feeding on the range is not legally allowed in many countries (see Grimes and Jackson 2015; Scott 2015). Feed and water sources on the range may attract migrating wild birds, and if these birds are carrying highly infectious diseases such as Avian Influenza, this is a serious risk to the industry (Feare 2010; Grillo et al. 2015; Hoque et al. 2015; Singh et al. 2017). Furthermore, open water sources such as troughs and dams used to water hens or co-grazing livestock or as water reservoirs to comply with fire regulations are potential threats to hen health and welfare as they attract wild birds (Kim et al. 2009; Hernández-Jover et al. 2015).
Pastures
During the past 20 years, there has been a renewed interest in pastures for poultry, with the rapid acceptance by industry of free-range production systems. Prior to the intensification of egg production in the decade after World War ll, hens had been largely managed in free-range conditions. For those interested in the literature that describes the nutritional management of hens at pasture pre-war, Heuser (1955) and Eyles (1963) have provided detailed accounts. Pasture species commonly grown for laying hens include rye grass (Lolium spp), kikuyu (Pennisetum clandestinum), fat hen (Chenopodium album), marshmallow (Malva parviflora), amaranth (Amaranthus spp), poke weed (Phytolacca spp), clover (Trifolium spps), cocksfoot (Dactylis glomerata), rhodes grass (Chloris gayana), buffalo grass (Stenotaphrum secundatum), couch grass (Cynodon dactylon) and African love grass (Eragrostis curvula; Singh et al. 2017). The nutritional value of pasture depends on its vegetative state, with young shoots containing high sugar and protein concentrations, in constast to senesencing grasses that are dominated by fibre and lignin content (Barnes et al. 1995; Brockman 1995; Knudsen 1997). The large variety of pasture species grown and the various factors that influence grass growth and composition, including temperature, rainfall, soil fertility and season, make it challenging for nutritionists to calculate and integrate the nutrients obtained from pasture into diet formulations for hens.
Pasture intake can imbalance nutrient intake and reduce nutrient digestibility, but is associated with additional fibre intake and this can be beneficial for gastrointestinal development and BW (Montagne et al. 2003). Some studies have reported that hens spending more time on the range have higher BWs (Singh et al. 2016; Iqbal et al. 2018). Foraging is one of the most common behaviours observed on the range, with 36–60% of hens exhibiting this activity (Campbell et al. 2017; Larsen et al. 2017). However, the quantity of nutrient uptake from the range in large commercial flocks housed in static sheds is usually considered to be negligible (0–5%), unless an effort is taken to promote pasture regrowth and allow for substantial recovery periods with rotational grazing or provision of harvested fodder (Walker and Gordon 2003). Some 82% of Australian free-range farms are never stripped of vegetation, indicating the importance of appropriate agronomic practices for range and pasture management (Singh et al. 2017).
The quantity and quality of pasture ingestion by individual hens can vary significantly and result in reduced flock uniformity (Walker and Gordon 2003; Hegelund et al. 2005). With average pasture intake of birds allowed unlimited grass access being as high as 30–60 g grass/hen.day, current trends promoting pasture-raised poultry ignore the fact that chickens are not grazers and have a very limited ability to utilise fibre (Fengler and Marquardt 1988; Singh and Cowieson 2013; Ruhnke et al. 2015b). Excessive availability of pasture can severely affect the nutrient intake of a formulated diet, resulting in reduced body condition and egg production, impaired welfare, and increased flock mortality. Importantly, grass impaction, a physical blockage of the gastrointestinal tract, can result from uncontrolled pasture intake (Singh and Cowieson 2013; Ruhnke et al. 2015b). Options to reduce unwanted effects associated with pasture intake may include mowing the range, so as to restrict the physical length of the grass ingested. Traditionally, the administration of apple cider vinegar via the drinking water has been reported to reduce clinical signs of grass constipation (Ruhnke et al. 2015b). However, high concentrations or long-term administration of these organic acids may negatively affect the hen’s acid–base balance, impairing eggshell quality and a successful vaccination response.
Feeding high density pellets has been shown to be beneficial, compensating for the reduced feed intake and associated nutrient dilution of pasture intake. Feed enzyme supplementation increases nutrient digestibility and improves performance of layers and broilers consumming pastures (Buchanan et al. 2007; Iqbal et al. 2018, 2019a). Benzoic acid and essential oils also reduce the negative impact of pasture intake and increase ileal protein and fibre digestibility in free-range layers (Weber et al. 2012; Iqbal et al. 2019a). Another strategy to reduce the negative impact of traditional range grasses is to plant lucerne (Medicago sativa), oats (Avena sativa), sorghum (Sorghum bicolor) and faba beans (Vicia faba) (Singh et al. 2017). This minimises the consequences of nutrient dilution due to pasture intake, by providing a higher nutrient feed source on the range. While this may also minimise the abrasive damage that the long structural grass species can have on the intestinal villi, or reduce the likelihood for gastrointestinal ileus and intusseption (Cassidy et al. 1981; Tasman-Jones et al. 1982), the overall diet intake may still be imbalanced and affect individual hen performance and welfare.
Forage and shade availability for free-range hens can be a requirement of various accreditation bodies (ACO 2013; FREPA 2015). When deciding which plants should be included in a pasture mix, the possibility of plant secondary metabolites and anti-nutritive factors, as mentioned above, should be considered. These compounds may reduce voluntary intake, egg production and egg quality. Meeting the need for shelter on the range can be accomplished by pasture, but it is best achieved by shrubs and bushes that are less likely to be ingested (de Koning et al. 2019). For example, when free-range hens were provided with old man saltbush (Atriplex nummularia), 5% of their dry-matter intake was from the bush (de Koning et al. 2019). Incorporation of river saltbush (Atriplex amnicola) into layer diets at up to 20% resulted in hens producing significantly darker egg yolks and thicker eggshells, while the taste of the eggs was considered superior by consumers (de Koning et al. 2019). However, excreta moisture content increased significantly with increased saltbush concentration in the diet. Nevertheless, alternative range vegetation such as trees, shrubs and bushes may provide enrichment while limiting the negative consequences that can be observed when exposing hens to uncontrolled pasture access.
Conclusions
Modern laying hens produce significantly more eggs than did hens 50 years ago. The application of scientific knowledge to layer breeding, nutrition, physiology and environmental control, together with improved husbandry and biosecurity, has resulted in outstanding advances in liveability, egg production, improved feed efficiency and, generally, low cost production of high quality eggs for consumers.
These advances have been accompanied by improvements in hen welfare, but little attention has been given to the relationship between the diet supplied and hen welfare. This has occurred because most laying hens are fed a complete diet formulated commercially to provide the required nutrients for optimal health and egg production. In other words, the laying hen, irrespective of her housing, does not experience hunger or thirst. Nevertheless, there are indications that hens do experience different degrees of stress in different husbandry systems. Although diet can help laying hens cope with stress and maintain wellbeing, it is not always obvious what nutritional considerations need to be made to achieve this. Further research is required on responses to nutrition, feeding and housing systems, high temperatures, feather cover, disease challenge, inflammation, nutrient imbalances, gut microbiota, toxic insult, pastures and variations in water quality.
Conflicts of interest
Australian Eggs Ltd provided funding support for the review but had no role in the content. WLB and SS are members of the Editorial Board of Animal Production Science. Neither were involved in the review and editorial process for this paper. The authors have no further conflicts of interest to declare.
Acknowledgements
Funding support for the review was received from the Australian Eggs Ltd. This paper was developed from a review by the authors prepared as one of a series of reviews on Laying Hen Welfare commissioned by Australian Eggs Ltd through an agreement with the University of Melbourne.
References
ACO (2013) ‘Australian certified organic standard: the requirements for organic certification.’ (Australian Organics: Benalla, Vic., Australia)Aerni V, Brinkhof MWG, Wechsler B, Oester H, Fröhlich E (2005) Productivity and mortality of laying hens in aviaries: a systematic review World’s Poultry Science Journal 61, 130–142.
| Productivity and mortality of laying hens in aviaries: a systematic reviewCrossref | GoogleScholarGoogle Scholar |
Ajakaiye JJ, Perez-Bello A, Mollineda-Trujillo A (2011) Impact of heat stress on egg quality in layer hens supplemented with l-ascorbic acid and dl-tocopherol acetate. Veterinarski Arhiv 81, 119–132.
Amerah AM, Ravindran V, Lentle RG, Thomas DG (2007) Feed particle size: implications on the digestion and performance of poultry World’s Poultry Science Journal 63, 439–455.
| Feed particle size: implications on the digestion and performance of poultryCrossref | GoogleScholarGoogle Scholar |
Amerah AM, Ravindran V, Lentle RG (2009) Influence of insoluble fiber and whole wheat inclusion on the performance, digestive tract development and ileal microbiota profile of broiler chickens British Poultry Science 50, 366–375.
| Influence of insoluble fiber and whole wheat inclusion on the performance, digestive tract development and ileal microbiota profile of broiler chickensCrossref | GoogleScholarGoogle Scholar | 19637037PubMed |
Annison EF (1983) Lipid metabolism. In ‘Physiology and biochemistry of the domestic fowl, vol. 3’. (Ed. BM Freeman) pp. 165–174. (Elsevier: Amsterdam, The Netherlands)
Annison EF, Hill KJ, Kenworthy R (1968) Volatile fatty acids in the digestive tract of the fowl. British Journal of Nutrition 22, 207–216.
| Volatile fatty acids in the digestive tract of the fowl.Crossref | GoogleScholarGoogle Scholar |
Attia YA, Aelh EH, Abedalla AA, Berika MA, Alharthi MA, Kucuk O, Sahin K, Aboushehema BM (2016) Laying performance, digestibility and plasma hormones in laying hens exposed to chronic heat stress as affected by betaine, vitamin C, and/or vitamin E supplementation SpringerPlus 5, 1619
| Laying performance, digestibility and plasma hormones in laying hens exposed to chronic heat stress as affected by betaine, vitamin C, and/or vitamin E supplementationCrossref | GoogleScholarGoogle Scholar | 27652192PubMed |
Axe DE (1995) Factors affecting uniformity of a mix. Animal Feed Science and Technology 53, 211–220.
| Factors affecting uniformity of a mix.Crossref | GoogleScholarGoogle Scholar |
Bajagal YS, Klieve AV, Dart PJ, Bryden, WL (2016) ‘Probiotics in animal nutrition: production, impact and regulation. FAO animal production and health paper no. 179.’ (Ed. HPS Makkar) (FAO: Rome, Italy)
Balnave D (2004) Challenges of accurately defining the nutrient requirements of heat-stressed poultry. Poultry Science 83, 5–14.
| Challenges of accurately defining the nutrient requirements of heat-stressed poultry.Crossref | GoogleScholarGoogle Scholar | 14761078PubMed |
Balnave D, Brake J (2002) Re-evaluation of the classical dietary arginine:lysine interaction for modern poultry diets: a review. World’s Poultry Science Journal 58, 275–289.
| Re-evaluation of the classical dietary arginine:lysine interaction for modern poultry diets: a review.Crossref | GoogleScholarGoogle Scholar |
Balnave D, Brake J (2005) Nutrition and management of heat-stressed pullets and laying hens. World’s Poultry Science Journal 61, 399–406.
| Nutrition and management of heat-stressed pullets and laying hens.Crossref | GoogleScholarGoogle Scholar |
Balnave D, Robinson D (2000) ‘Amino acid and energy requirements of imported Brown layer strains.’ (Rural Industries Research and Development Corporation: Canberra, ACT, Australia)
Balnave D, Scott T (1986) The influence of minerals in egg shell defects. Nutrition Reports International 34, 29–34.
Balnave D, Yoselewitz I, Dixon RJ (1989) Physiological changes associated with the production of defective egg shells by hens receiving sodium chloride in the drinking water. British Journal of Nutrition 61, 35–43.
| Physiological changes associated with the production of defective egg shells by hens receiving sodium chloride in the drinking water.Crossref | GoogleScholarGoogle Scholar |
Bar A (2009) Calcium transport in strongly calcifying laying birds: mechanisms and regulation. Comparative Biochemistry and Physiology. Part A, Molecular & Integrative Physiology 152, 447–469.
| Calcium transport in strongly calcifying laying birds: mechanisms and regulation.Crossref | GoogleScholarGoogle Scholar |
Barnes RF, Miller DF, Nelson JC (1995) ‘Forages: an introduction to grassland agriculture, vol. 1.’ (Iowa State University Press: Ames, IA, USA)
Barnes DM, Kirby YK, Oliver KG (2001) Effects of biogenic amines on growth and the incidence of proventricular lesions in broiler chickens Poultry Science 80, 906–911.
| Effects of biogenic amines on growth and the incidence of proventricular lesions in broiler chickensCrossref | GoogleScholarGoogle Scholar | 11469653PubMed |
Bass PD, Hooge DM, Koutsos EA (2007) Dietary thyroxine induces moult in chickens (Gallus gallus domesticus) Comparative Biochemistry and Physiology. Part A. Molecular & Integrative Physiology 146, 335–341.
| Dietary thyroxine induces moult in chickens (Gallus gallus domesticus)Crossref | GoogleScholarGoogle Scholar |
Behnke KC (2001) Factors influencing pellet quality. Feed Technology 5, 19–22.
Benabdeljelil K, Ryadi A, Jensen LS (1990) Effect of dietary ascorbic acid supplementation on the performance of brown-egg layers and egg quality Animal Feed Science and Technology 30, 301–311.
| Effect of dietary ascorbic acid supplementation on the performance of brown-egg layers and egg qualityCrossref | GoogleScholarGoogle Scholar |
Bennett CD, Classen HL (2003) Performance of two strains of laying hens fed ground and whole barley with and without access to insoluble grit Poultry Science 82, 147–149.
| Performance of two strains of laying hens fed ground and whole barley with and without access to insoluble gritCrossref | GoogleScholarGoogle Scholar | 12580257PubMed |
Bjerrum L, Pedersen K, Engberg RM (2005) The influence of whole wheat feeding on Salmonella infection and gut flora composition in broilers Avian Diseases 49, 9–15.
| The influence of whole wheat feeding on Salmonella infection and gut flora composition in broilersCrossref | GoogleScholarGoogle Scholar | 15839406PubMed |
Bouvarel I, Nys Y, Lescoat P (2011) Hen nutrition for sustained egg quality. In ‘Improving the safety and quality of eggs and egg products, vol. 1: egg chemistry, production and consumption’. (Eds Y Nys, M Bain, F Van Immerseel) pp. 261–299. (Woodhead Publishing: Cambridge, UK)
Braastad BO, Katle J (1989) Behavioural differences between laying hen populations selected for high and low efficiency of food utilisation. British Poultry Science 30, 533–544.
| Behavioural differences between laying hen populations selected for high and low efficiency of food utilisation.Crossref | GoogleScholarGoogle Scholar | 2819497PubMed |
Brackpool CE, Roberts JR, Balnave D (1996) Blood electrolyte status over the daily laying cycle and the effect of saline drinking water on the availability of calcium in the blood for egg-shell formation in the laying hen. Journal of Animal Physiology and Animal Nutrition 75, 214–225.
| Blood electrolyte status over the daily laying cycle and the effect of saline drinking water on the availability of calcium in the blood for egg-shell formation in the laying hen.Crossref | GoogleScholarGoogle Scholar |
Branton SL, Lott DB, Maslin WL, Day EJ (1995) Fatty liver hemorrhagic syndrome observed in commercial layers fed diets containing chelated minerals. Avian Diseases 39, 631–635.
| Fatty liver hemorrhagic syndrome observed in commercial layers fed diets containing chelated minerals.Crossref | GoogleScholarGoogle Scholar | 8561751PubMed |
Bregendahl K, Roberts SA, Kerr B, Hoehler D (2008) Ideal ratios of isoleucine, methionine, methionine plus cystine, threonine, tryptophan, and valine relative to lysine for white leghorn-type laying hens of twenty-eight to thirty-four weeks of age. Poultry Science 87, 744–758.
| Ideal ratios of isoleucine, methionine, methionine plus cystine, threonine, tryptophan, and valine relative to lysine for white leghorn-type laying hens of twenty-eight to thirty-four weeks of age.Crossref | GoogleScholarGoogle Scholar | 18339997PubMed |
Britton G (1996) Carotenoids. In ‘Natural food colorant’. 2nd edn. (Eds GAF Hendry, JD Houghton) pp. 197–243. (Springer: Boston, MA, USA)
Brockman JS (1995) Grassland. In ‘The agricultural notebook’, 19th edn. (Ed. RJ Soffe) pp. 194–226. (Blackwell Science: Oxford, UK)
Bryden WL (2009). Mycotoxins and mycotoxicoses: significance, occurrence and mitigation in the food chain. In ‘General and applied toxicology’. 3rd edn. (Eds B Ballantyne, T Marrs, T Syversen) pp. 3529–3553. (John Wiley & Sons Ltd: Chichester, UK)
Bryden WL (2012a) Mycotoxin contamination of the feed supply chain: implications for animal productivity and feed security Animal Feed Science and Technology 173, 134–158.
| Mycotoxin contamination of the feed supply chain: implications for animal productivity and feed securityCrossref | GoogleScholarGoogle Scholar |
Bryden WL (2012b) Food and feed, mycotoxins and the perpetual pentagram in a changing animal production environment Animal Production Science 52, 383–397.
| Food and feed, mycotoxins and the perpetual pentagram in a changing animal production environmentCrossref | GoogleScholarGoogle Scholar |
Bryden WL, Cumming RB, Balnave D (1979) The influence of vitamin A status on the response of chickens to aflatoxin B1 and changes in liver lipid metabolism associated with aflatoxicosis. British Journal of Nutrition 41, 529–540.
| The influence of vitamin A status on the response of chickens to aflatoxin B1 and changes in liver lipid metabolism associated with aflatoxicosis.Crossref | GoogleScholarGoogle Scholar |
Bryden WL, Selle PH, Ravindran V, Acamovic T (2007) Phytate: an antinutrient factor in animal diets. In ‘Poisonous plants: global research and solutions’. (Eds KE Panter, TL Wierenga, JA Pfister) pp. 279–284. (CABI Publishing: Wallingford, UK)
Buchanan N, Kimbler L, Parsons A, Seidel G, Bryan W, Felton E, Moritz J (2007) The effects of nonstarch polysaccharide enzyme addition and dietary energy restriction on performance and carcass quality of organic broiler chickens Journal of Applied Poultry Research 16, 1–12.
| The effects of nonstarch polysaccharide enzyme addition and dietary energy restriction on performance and carcass quality of organic broiler chickensCrossref | GoogleScholarGoogle Scholar |
Burley HK, Anderson KE, Patterson PH, Tillman PB (2016) Formulation challenges of organic poultry diets with readily available ingredients and limited synthetic methionine Journal of Applied Poultry Research 25, 443–454.
| Formulation challenges of organic poultry diets with readily available ingredients and limited synthetic methionineCrossref | GoogleScholarGoogle Scholar |
Butler EJ (1976) Fatty liver diseases in the domestic fowl: a review. Avian Pathology 5, 1–14.
| Fatty liver diseases in the domestic fowl: a review.Crossref | GoogleScholarGoogle Scholar | 18777323PubMed |
Calderon VM, Jensen LS (1990) The requirement for sulfur amino acid by laying hens as influenced by the protein concentration Poultry Science 69, 934–944.
| The requirement for sulfur amino acid by laying hens as influenced by the protein concentrationCrossref | GoogleScholarGoogle Scholar | 2395789PubMed |
Campbell DLM (2020) Skeletal health of layers across all housing systems and future research directions for Australia. Animal Production Science.
| Skeletal health of layers across all housing systems and future research directions for Australia.Crossref | GoogleScholarGoogle Scholar |
Campbell DLM, Hinch GN, Downing JA, Lee C (2017) Outdoor stocking density in free-range laying hens: effects on behaviour and welfare Animal 11, 1036–1045.
| Outdoor stocking density in free-range laying hens: effects on behaviour and welfareCrossref | GoogleScholarGoogle Scholar | 27821220PubMed |
Carter TA, Sneed RE (1987) ‘Drinking water quality for poultry. PS&T guide no. 42. Extension poultry science.’ (North Carolina State University: Raleigh, NC, USA)
Cassidy MM, Lightfoot FG, Grau LE, Story JA, Kritchevsky D, Vahouny GC (1981) Effect of chronic intake of dietary fibers on the ultrastructural topography of rat jejunum and colon: a scanning electron microscopy study The American Journal of Clinical Nutrition 34, 218–228.
| Effect of chronic intake of dietary fibers on the ultrastructural topography of rat jejunum and colon: a scanning electron microscopy studyCrossref | GoogleScholarGoogle Scholar | 6259920PubMed |
Caston L, Leeson S (1990) Research note: dietary flax and egg composition Poultry Science 69, 1617–1620.
| Research note: dietary flax and egg compositionCrossref | GoogleScholarGoogle Scholar |
Centraal Veevoederbureau (CVB) (1996) Aminozurenbehoefte van Leghennen en Vleeskuikens [Amino acid requirements for laying hens and broiler chickens]. Documentation report nr. 18. CVB, Lelystad, The Netherlands. [In Dutch]
Cheeke PR (1998) ‘Natural toxicants in feeds, forages and poisonous plants.’ (Interstate Publishers, Inc.: Danville, VI, USA)
Coelho M (2001) Involvement of calcium and phosphorus in bone and shell quality of early maturing commercials layers World Poultry 17, 16–19.
Coon C, Zhang B (1999) Ideal amino acid profile for layers examined. Feedstuffs 71, 13–15.
Couch JR (1956) Fatty livers in laying hens: a condition which may occur as a result of increased strain. Feedstuffs 28, 46–54.
Crespo R, Shivaprasad HL (2003) Developmental, metabolic, and other non-infectious disorders. In ‘Diseases of poultry’. 11th edn (Eds YM Saif, HJ Barnes JR Glisson AM Fadley LR McDougald, DE Swayne) pp. 1055–1102 (Iowa State Press: Ames, IA, USA)
Dalibard P, Hess V, Tutour LLE, Peisker M, Peris S, Perojo Gutierrez A, Redshow M (2014) ‘Amino acids in animal nutrition.’ (Fefana Publication: Brussels, Belgium)
Damron BL (1998) Sodium chloride concentration in drinking water and egg shell quality. Poultry Science 77, 1488–1491.
| Sodium chloride concentration in drinking water and egg shell quality.Crossref | GoogleScholarGoogle Scholar | 9776055PubMed |
Daniel M, Balnave D (1981) Responses of laying hens to gradual and abrupt increases in ambient temperature and humidity. Australian Journal of Experimental Agriculture and Animal Husbandry 21, 189–195.
| Responses of laying hens to gradual and abrupt increases in ambient temperature and humidity.Crossref | GoogleScholarGoogle Scholar |
de Almeida Brainer MM, Bôa-Viagem Rabello C, Batista dos Santos MJ, Ludkec JV, da Costa Lopes C, de Medeiros WRL, Costa FGP (2016) Crude protein requirements of free-range laying hens. Animal Production Science 56, 1622–1628.
de Matos R (2008) Calcium metabolism in birds The Veterinary Clinics of North America. Exotic Animal Practice 11, 59–82.
| Calcium metabolism in birdsCrossref | GoogleScholarGoogle Scholar | 18165138PubMed |
de Koning C, Barekatain R, Singh M, Drake K (2019) Saltbush (Atriplex nummularia and A. amnicola) as potential plants for free-range layer farms: consequences for layer performance, egg sensory qualities and excreta moisture Poultry Science 98, 4555–4564.
| Saltbush (Atriplex nummularia and A. amnicola) as potential plants for free-range layer farms: consequences for layer performance, egg sensory qualities and excreta moistureCrossref | GoogleScholarGoogle Scholar | 31135917PubMed |
den Brinker CA, Rayner CJ, Kerr MG, Bryden WL (2003) Biogenic amines in Australian animal by-product meals. Australian Journal of Experimental Agriculture 43, 113–119.
| Biogenic amines in Australian animal by-product meals.Crossref | GoogleScholarGoogle Scholar |
Department of Agriculture, Fisheries and Forestry (DAFF) (2009) ‘National water biosecurity manual: poultry production.’ (Department of Agriculture, Fisheries and Forestry: Canberra, ACT, Australia)
Edwards LE, Hemsworth PH (2021) The impact of management, husbandry and stockperson decisions on the welfare of laying hens in Australia. Animal Production Science.
| The impact of management, husbandry and stockperson decisions on the welfare of laying hens in Australia.Crossref | GoogleScholarGoogle Scholar |
Elaroussi MA, Forte LR, Eber SL, Biellier HV (1994) Calcium homeostasis in the laying hen. 1. Age and dietary calcium effects Poultry Science 73, 1581–1589.
| Calcium homeostasis in the laying hen. 1. Age and dietary calcium effectsCrossref | GoogleScholarGoogle Scholar | 7816733PubMed |
Emmans GC (1977) The nutrient intake of laying hens given a choice of diets, in relation to their production requirements British Poultry Science 18, 227–236.
| The nutrient intake of laying hens given a choice of diets, in relation to their production requirementsCrossref | GoogleScholarGoogle Scholar |
Engberg RM, Hedemann MS, Jensen BB (2002) The influence of grinding and pelleting of feed on the microbial composition and activity in the digestive tract of broiler chickens British Poultry Science 43, 569–579.
| The influence of grinding and pelleting of feed on the microbial composition and activity in the digestive tract of broiler chickensCrossref | GoogleScholarGoogle Scholar | 12365514PubMed |
Engberg RM, Hedemann MS, Steenfeldt S, Jensen BB (2004) Influence of whole wheat and xylanase on broiler performance and microbial composition and activity in the digestive tract Poultry Science 83, 925–938.
| Influence of whole wheat and xylanase on broiler performance and microbial composition and activity in the digestive tractCrossref | GoogleScholarGoogle Scholar | 15206619PubMed |
Etches RJ (1987) Calcium logistics in the laying hen. The Journal of Nutrition 117, 619–628.
| Calcium logistics in the laying hen.Crossref | GoogleScholarGoogle Scholar | 3572575PubMed |
Eyles DE (1963) Non-ruminants, In ‘Animal health, production and pasture’. (Eds AN Worden, KC Sellers, DE Tribe) pp. 359–383. (Longmans: London, UK)
Feare CJ (2010) Role of wild birds in the spread of highly pathogenic avian influenza virus H5N1 and implications for global surveillance Avian Diseases 54, 201–212.
| Role of wild birds in the spread of highly pathogenic avian influenza virus H5N1 and implications for global surveillanceCrossref | GoogleScholarGoogle Scholar | 20521633PubMed |
Feddern V, Mazzuco H, Fonseca FN, de Lima GJMM (2019) A review on biogenic amines in food and feed: toxicological aspects, impact on health and control measures Animal Production Science 59, 608–618.
| A review on biogenic amines in food and feed: toxicological aspects, impact on health and control measuresCrossref | GoogleScholarGoogle Scholar |
Fengler AI, Marquardt RR (1988) Water-soluble pentosans from rye: II. Effects on rate of dialysis and on the retention of nutrients by the chick. Cereal Chemistry 65, 298–302.
Fleming RH (2008) Nutritional factors affecting poultry bone health. The Proceedings of the Nutrition Society 67, 177–183.
| Nutritional factors affecting poultry bone health.Crossref | GoogleScholarGoogle Scholar | 18412991PubMed |
Fleming RH, McCormack HA, Mcteir L, Whitehead CC (1998) Bone structure and strength at different ages in laying hens and effects of dietary particulate limestone, vitamin K and ascorbic acid. British Poultry Science 39, 434–440.
| Bone structure and strength at different ages in laying hens and effects of dietary particulate limestone, vitamin K and ascorbic acid.Crossref | GoogleScholarGoogle Scholar | 9693828PubMed |
FREPA (2015) ‘Free range egg standards.’ (Free Range Egg and Poultry Australia: Benalla, Vic., Australia)
Frikha M, Safaa HM, Serrano MP, Arbe X, Mateos GG (2009) Influence of the main cereal and feed form of the diet on performance and digestive tract traits of brown-egg laying pullets Poultry Science 88, 994–1002.
| Influence of the main cereal and feed form of the diet on performance and digestive tract traits of brown-egg laying pulletsCrossref | GoogleScholarGoogle Scholar | 19359688PubMed |
Frikha M, Safaa HM, Serrano MP, Jiménez-Moreno E, Lázaro R, Mateos GG (2011) Influence of the main cereal in the diet and particle size of the cereal on productive performance and digestive traits of brown-egg laying pullets Animal Feed Science and Technology 164, 106–115.
| Influence of the main cereal in the diet and particle size of the cereal on productive performance and digestive traits of brown-egg laying pulletsCrossref | GoogleScholarGoogle Scholar |
GfE (1999) ‘Empfehlungen zur Energie- und Nährstoffversorgung der Legehennen und Masthühner (Broiler).’ (DLG-Verlag: Frankfurt/Main, Germany) [In German]
Glatz PC (2001) Effect of cool drinking water on production and shell quality of laying hens in summer. Asian-Australasian Journal of Animal Sciences 14, 850–854.
| Effect of cool drinking water on production and shell quality of laying hens in summer.Crossref | GoogleScholarGoogle Scholar |
Glatz PC (2003) The effect of beak length and condition on food intake and feeding behaviour of hens International Journal of Poultry Science 2, 53–57.
| The effect of beak length and condition on food intake and feeding behaviour of hensCrossref | GoogleScholarGoogle Scholar |
Glatz PC, Tilbrook AJ (2020) Welfare issues associated with moulting of laying hens. Animal Production Science.
| Welfare issues associated with moulting of laying hens.Crossref | GoogleScholarGoogle Scholar |
Glatz PC, Underwood G (2020) Current methods and techniques of beak trimming laying hens, welfare issues and alternative approaches. Animal Production Science.
| Current methods and techniques of beak trimming laying hens, welfare issues and alternative approaches.Crossref | GoogleScholarGoogle Scholar |
Gous RM, Morris TR (2001) The influence of pelleted feed on the response of growing pullets to photoperiods of less than ten hours British Poultry Science 42, 203–206.
| The influence of pelleted feed on the response of growing pullets to photoperiods of less than ten hoursCrossref | GoogleScholarGoogle Scholar | 11421329PubMed |
Grillo VL, Arzey KE, Hansbro PM, Hurt AC, Warner S, Bergfeld J, Burgess GW, Cookson B, Dickason CJ, Ferenczi M, Hollingsworth T (2015) Avian influenza in Australia: a summary of 5 years of wild bird surveillance. Australian Veterinary Journal 93, 387–393.
| Avian influenza in Australia: a summary of 5 years of wild bird surveillance.Crossref | GoogleScholarGoogle Scholar | 26503532PubMed |
Grimes TM (1975) Causes of disease in two commercial flocks of laying hens. Australian Veterinary Journal 51, 337–343.
| Causes of disease in two commercial flocks of laying hens.Crossref | GoogleScholarGoogle Scholar | 1180772PubMed |
Grimes T, Jackson C (2015) ‘Code of practice for biosecurity in the egg industry.’ (Australian Egg Corporation: Sydney, NSW, Australia)
Grobas S, Mendez J, De Blas C, Mateos GG (1999a) Laying hen productivity as affected by energy, supplemental fat, and linoleic acid concentration of the diet Poultry Science 78, 1542–1551.
| Laying hen productivity as affected by energy, supplemental fat, and linoleic acid concentration of the dietCrossref | GoogleScholarGoogle Scholar | 10560827PubMed |
Grobas S, Mendez J, De Blas C, Mateos GG (1999b) Influence of dietary energy, supplemental fat and linoleic acid concentration on performance of laying hens at two ages British Poultry Science 40, 681–687.
| Influence of dietary energy, supplemental fat and linoleic acid concentration on performance of laying hens at two agesCrossref | GoogleScholarGoogle Scholar | 10670682PubMed |
Grobas S, Mendez J, Lazaro R, De Blas C, Mateo GG (2001) Influence of source and percentage of fat added to diet on performance and fatty acid composition of egg yolks of two strains of laying hens Poultry Science 80, 1171–1179.
| Influence of source and percentage of fat added to diet on performance and fatty acid composition of egg yolks of two strains of laying hensCrossref | GoogleScholarGoogle Scholar | 11495470PubMed |
Groves P (2021) Impact of parasites on Australian laying hen welfare. Animal Production Science
| Impact of parasites on Australian laying hen welfare.Crossref | GoogleScholarGoogle Scholar | (In press).
Habibian M, Sadeghi G, Ghazi S, Moeini MM (2015) Selenium as a feed supplement for heat-stressed poultry: a review. Biological Trace Element Research 165, 183–193.
| Selenium as a feed supplement for heat-stressed poultry: a review.Crossref | GoogleScholarGoogle Scholar | 25721721PubMed |
Hafeez A, Mader A, Ruhnke I, Röhe I, Goodarzi Boroojeni F, Yousaf MS, Männer K, Zentek J (2015) Implication of milling methods, thermal treatment, and particle size of feed in layers on mineral digestibility and retention of minerals in egg contents Poultry Science 94, 240–248.
| Implication of milling methods, thermal treatment, and particle size of feed in layers on mineral digestibility and retention of minerals in egg contentsCrossref | GoogleScholarGoogle Scholar | 25630675PubMed |
Hafeez A, Mader A, Röhe I, Ruhnke I, Goodarzi Boroojeni F, Yousaf MS, Männer K, Zentek J (2015) The effect of milling methods, thermal treatment, and particle size of feed on exterior and interior egg quality in laying hens European Journal of Poultry Science 79,
Hamilton PB, Garlich JD (1971) Aflatoxin as a possible cause of fatty liver syndrome in laying hens. Poultry Science 50, 800–804.
Hamilton RMG (1982) Methods and factors that affect the measurement of egg shell quality. Poultry Science 61, 2022–2039.
| Methods and factors that affect the measurement of egg shell quality.Crossref | GoogleScholarGoogle Scholar |
Harms RH, Costa PT, Miles RD (1982) Daily feed intake and performance of laying hens grouped according to their body weight Poultry Science 61, 1021–1024.
| Daily feed intake and performance of laying hens grouped according to their body weightCrossref | GoogleScholarGoogle Scholar |
Hartini S, Choct M, Hinch G, Nolan JV (2002) Effect of diet composition and feed form on the behavior of ISA brown laying hens. Proceedings of the Australian Poultry Science Symposium 4, 104–110.
Hegelund L, Sørensen JT, Kjaer JB, Kristensen IS (2005) Use of the range area in organic egg production systems: effect of climatic factors, flock size, age and artificial cover British Poultry Science 46, 1–8.
| Use of the range area in organic egg production systems: effect of climatic factors, flock size, age and artificial coverCrossref | GoogleScholarGoogle Scholar | 15835245PubMed |
Hencken H (1992) Chemical and physiological behavior of feed carotenoids and their effects on pigmentation Poultry Science 71, 711–717.
| Chemical and physiological behavior of feed carotenoids and their effects on pigmentationCrossref | GoogleScholarGoogle Scholar | 1594522PubMed |
Hernández-Jover M, Schemann K, East IJ, Toribio JA (2015) Evaluating the risk of avian influenza introduction and spread among poultry exhibition flocks in Australia Preventive Veterinary Medicine 118, 128–141.
| Evaluating the risk of avian influenza introduction and spread among poultry exhibition flocks in AustraliaCrossref | GoogleScholarGoogle Scholar | 25496909PubMed |
Hetland H, Svihus B, Moe R (2003a) Effect of feed structure on performance and welfare in laying hens housed in conventional and furnished cages. Acta Agriculturæ Scandinavica. Section A. Animal Science 53, 92–100.
| Effect of feed structure on performance and welfare in laying hens housed in conventional and furnished cages.Crossref | GoogleScholarGoogle Scholar |
Hetland H, Svihus B, Krogdahl A (2003b) Effects of oat hulls and wood shavings on digestion in broilers and layers fed diets based on whole or ground wheat British Poultry Science 44, 275–282.
| Effects of oat hulls and wood shavings on digestion in broilers and layers fed diets based on whole or ground wheatCrossref | GoogleScholarGoogle Scholar | 12828213PubMed |
Hetland H, Moe RO, Tauson R, Lervik S, Svihus B (2004) Effect of including whole oats into pellets on performance and plumage condition in laying hens housed in conventional and furnished cages. Acta Agriculturæ Scandinavica. Section A. Animal Science 54, 206–212.
| Effect of including whole oats into pellets on performance and plumage condition in laying hens housed in conventional and furnished cages.Crossref | GoogleScholarGoogle Scholar |
Hetland H, Svihus B, Choct M (2005) Role of insoluble fiber on gizzard activity in layers Journal of Applied Poultry Science Research 14, 38–46.
| Role of insoluble fiber on gizzard activity in layersCrossref | GoogleScholarGoogle Scholar |
Heuser GF (1955) ‘Feeding poultry: the classic guide to poultry nutrition.’ (John Wiley & Sons: New York, NY, USA)
Hoque MA, Burgess GW, Cheam AL, Skerratt LF (2015) Epidemiology of avian influenza in wild aquatic birds in a biosecurity hotspot, north Queensland, Australia. Preventive Veterinary Medicine 118, 169–181.
| Epidemiology of avian influenza in wild aquatic birds in a biosecurity hotspot, north Queensland, Australia.Crossref | GoogleScholarGoogle Scholar | 25481624PubMed |
Huang DS, Li DF, Xing JJ, Ma YX, Li ZJ, Lv SQ (2006) Effects of feed particle size and feed form on survival of Salmonella typhimurium in the alimentary tract and cecal S. typhimurium reduction in growing broilers Poultry Science 85, 831–836.
| Effects of feed particle size and feed form on survival of Salmonella typhimurium in the alimentary tract and cecal S. typhimurium reduction in growing broilersCrossref | GoogleScholarGoogle Scholar | 16673759PubMed |
Huber-Eicher B, Wechsler B (1998) The effect of quality and availability of foraging materials on feather pecking in laying hen chicks Applied Animal Behaviour Science 55, 861–873.
| The effect of quality and availability of foraging materials on feather pecking in laying hen chicksCrossref | GoogleScholarGoogle Scholar |
Husband AJ (1995) The immune system and integrated homeostasis. Immunology and Cell Biology 73, 377–382.
| The immune system and integrated homeostasis.Crossref | GoogleScholarGoogle Scholar | 7493777PubMed |
Husband AJ, Bryden WL (1996) Nutrition, stress and immune activation. Proceedings of the Nutrition Society of Australia 20, 60–70.
Iqbal Z, Roberts J, Perez-Maldonado RA, Goodarzi Boroojeni F, Swick RA, Ruhnke I (2018) Pasture, multi-enzymes, benzoic acid and essential oils positively influence performance, intestinal organ weight and egg quality in free-range laying hens British Poultry Science 59, 180–189.
| Pasture, multi-enzymes, benzoic acid and essential oils positively influence performance, intestinal organ weight and egg quality in free-range laying hensCrossref | GoogleScholarGoogle Scholar | 29130331PubMed |
Iqbal Z, Metzger F, Singh M, Morgan N, Swick RA, Perez-Maldonado RA, M’Sadeq S, Zentek J, Ruhnke I (2019a) Enzymes and/or combination of organic acid and essential oils supplementation in pasture-fed free-range laying hens increased the digestibility of nutrients and non-starch polysaccharides Poultry Science 98, 1410–1424.
| Enzymes and/or combination of organic acid and essential oils supplementation in pasture-fed free-range laying hens increased the digestibility of nutrients and non-starch polysaccharidesCrossref | GoogleScholarGoogle Scholar | 30307570PubMed |
Iqbal Z, Drake K, Swick RA, Perez-Maldonado RA, Ruhnke I (2019b) Feed particle selection and nutrient intake altered by pecking stone consumption and beak length in free-range laying hens Animal Nutrition 5, 140–147.
| Feed particle selection and nutrient intake altered by pecking stone consumption and beak length in free-range laying hensCrossref | GoogleScholarGoogle Scholar | 31193914PubMed |
Jais C, Roth FX, Kirchgessner M (1995) The determination of the optimum ratio between the essential amino acids in laying hen diets. Archiv für Geflügelkunde 59, 292–302.
Jalal MA, Scheideler SE, Marx D (2006) Effect of bird cage space and dietary metabolizable energy level on production parameters in laying hens Poultry Science 85, 306–311.
| Effect of bird cage space and dietary metabolizable energy level on production parameters in laying hensCrossref | GoogleScholarGoogle Scholar | 16523631PubMed |
Janczak AM, Riber AB (2015) Review of rearing-related factors affecting the welfare of laying hens. Poultry Science 94, 1454–1469.
| Review of rearing-related factors affecting the welfare of laying hens.Crossref | GoogleScholarGoogle Scholar | 26009752PubMed |
Johnsson M, Jonsson KB, Andersson L, Jensen P, Wright D (2015) Genetic regulation of bone metabolism in the chicken: similarities and differences to mammalian systems. PLOS Genetics 11, e1005250
| Genetic regulation of bone metabolism in the chicken: similarities and differences to mammalian systems.Crossref | GoogleScholarGoogle Scholar | 26023928PubMed |
Jongman EC (2021) Rearing conditions of laying hens and welfare during the laying phase. Animal Production Science
| Rearing conditions of laying hens and welfare during the laying phase.Crossref | GoogleScholarGoogle Scholar | (In press).
Julian RJ (2005) Production and growth related disorders and other metabolic diseases of poultry: a review. Veterinary Journal 169, 350–369.
| Production and growth related disorders and other metabolic diseases of poultry: a review.Crossref | GoogleScholarGoogle Scholar |
Karunajeewa H (1978) The performance of cross‐bred hens given free choice feeding of whole grains and a concentrate mixture and the influence of source of xanthophylls on yolk colour British Poultry Science 19, 699–708.
| The performance of cross‐bred hens given free choice feeding of whole grains and a concentrate mixture and the influence of source of xanthophylls on yolk colourCrossref | GoogleScholarGoogle Scholar |
Karunajeewa H, Hughes R, McDonald M, Shenstone F (1984) A review of factors influencing pigmentation of egg yolks. World’s Poultry Science Journal 40, 52–65.
| A review of factors influencing pigmentation of egg yolks.Crossref | GoogleScholarGoogle Scholar |
Khalafalla MK, Bessei W (1997) Effect of ascorbic acid supplementation on eggshell quality of laying hens receiving saline drinking water. Archiv für Geflügelkunde 61, 172–175.
Khan SH, Sardar R (2005) Effect of vitamin C supplementation on the performance of desi, Fayoumi and commercial white leghorn chicken exposed to heat stress. Pakistan Veterinary Journal 25, 136–166.
Khattak F, Helmbrecht A (2019) Effect of different levels of tryptophan on productive performance, egg quality, blood biochemistry, and caecal microbiota of hens housed in enriched colony cages under commercial stocking density Poultry Science 98, 2094–2104.
| Effect of different levels of tryptophan on productive performance, egg quality, blood biochemistry, and caecal microbiota of hens housed in enriched colony cages under commercial stocking densityCrossref | GoogleScholarGoogle Scholar | 30590740PubMed |
Kim WK, Donalson LM, Herrera P, Kubena LF, Nisbet DJ, Ricke SC (2005) Comparisons of moulting diets on skeletal quality and eggshell parameters in hens at the end of the second egg-laying cycle Poultry Science 84, 522–527.
| Comparisons of moulting diets on skeletal quality and eggshell parameters in hens at the end of the second egg-laying cycleCrossref | GoogleScholarGoogle Scholar | 15844806PubMed |
Kim JK, Negovetich NJ, Forrest HL, Webster RG (2009) Ducks: the ‘Trojan horses’ of H5N1 influenza Influenza and Other Respiratory Viruses 3, 121–128.
| Ducks: the ‘Trojan horses’ of H5N1 influenzaCrossref | GoogleScholarGoogle Scholar | 19627369PubMed |
Klasing KC (1998) Avian macrophages: regulators of local and systemic immune responses. Poultry Science 77, 983–989.
| Avian macrophages: regulators of local and systemic immune responses.Crossref | GoogleScholarGoogle Scholar | 9657608PubMed |
Klasing KC (2007) Nutrition and the immune system British Poultry Science 48, 525–537.
| Nutrition and the immune systemCrossref | GoogleScholarGoogle Scholar | 17952723PubMed |
Knudsen KEB (1997) Carbohydrate and lignin contents of plant materials used in animal feeding Animal Feed Science and Technology 67, 319–338.
| Carbohydrate and lignin contents of plant materials used in animal feedingCrossref | GoogleScholarGoogle Scholar |
Kogut MH (2009) Impact of nutrition on the innate immune response to infection in poultry Journal of Applied Poultry Research 18, 111–124.
| Impact of nutrition on the innate immune response to infection in poultryCrossref | GoogleScholarGoogle Scholar |
Kolakshyapati M, Flavel R, Sibanda TZ, Ruhnke I (2019) Range access has no beneficial effect on tibia health of free-range laying hens while body weight is positively correlated with different bone parameters Poultry Science 98, 6241–6250.
| Range access has no beneficial effect on tibia health of free-range laying hens while body weight is positively correlated with different bone parametersCrossref | GoogleScholarGoogle Scholar | 31504903PubMed |
Korver DR (2012) Implications of changing immune function through nutrition in poultry Animal Feed Science and Technology 173, 54–64.
| Implications of changing immune function through nutrition in poultryCrossref | GoogleScholarGoogle Scholar |
Koutsos E, Arias V (2006) Intestinal ecology: interactions among the gastrointestinal tract, nutrition, and the microflora Journal of Applied Poultry Research 15, 161–173.
| Intestinal ecology: interactions among the gastrointestinal tract, nutrition, and the microfloraCrossref | GoogleScholarGoogle Scholar |
Koutsos EA, Klasing KC (2014) Factors modulating the avian immune system. In ‘Avian immunology’. 2nd edn. (Eds KA Schat, B Kaspers, P Kaiser) pp. 299–313 (Academic Press: London, UK)
Kraimi N, Dawkins M, Gebhardt-Henrich SG, Velge P, Rychlik I, Volf J, Creach P, Smith A, Colles F, Leterrier C (2019) Influence of the microbiota–gut–brain axis on behavior and welfare in farm animals: a review. Physiology & Behavior 210, 112658
| Influence of the microbiota–gut–brain axis on behavior and welfare in farm animals: a review.Crossref | GoogleScholarGoogle Scholar |
Lara LJ, Rostagno MH (2013) Impact of heat stress on poultry production. Animals 3, 356–369.
| Impact of heat stress on poultry production.Crossref | GoogleScholarGoogle Scholar | 26487407PubMed |
Larsen H, Cronin G, Smith CL, Hemsworth P, Rault JL (2017) Behaviour of free-range laying hens in distinct outdoor environments Animal Welfare 26, 255–264.
| Behaviour of free-range laying hens in distinct outdoor environmentsCrossref | GoogleScholarGoogle Scholar |
Lay DC, Fulton RM, Hester PY, Karcher DM, Kjaer JB, Mench JA, Mullens BA, Newberry RC, Nicol CJ, O’Sullivan NP, Porter RE (2011) Hen welfare in different housing systems. Poultry Science 90, 278–294.
| Hen welfare in different housing systems.Crossref | GoogleScholarGoogle Scholar | 21177469PubMed |
Laycock SR, Ball RO (1990) Alleviation of hysteria in laying hens with dietary tryptophan. Canadian Journal of Veterinary Research 54, 291–295.
Lee DHK, Robinson KW, Yeates NTM, Scott MIR (1945) Poultry husbandry in hot climates: experimental enquiries. Poultry Science 24, 195–207.
| Poultry husbandry in hot climates: experimental enquiries.Crossref | GoogleScholarGoogle Scholar |
Leenstra F, Maurer V, Bestman M, van Sambeek F, Zeltner E, Reuvekamp B, Galea F, van Niekerk T (2012) Performance of commercial laying hen genotypes on free range and organic farms in Switzerland, France and the Netherlands British Poultry Science 53, 282–290.
| Performance of commercial laying hen genotypes on free range and organic farms in Switzerland, France and the NetherlandsCrossref | GoogleScholarGoogle Scholar | 22978584PubMed |
Leenstra F, Maurer V, Galea F, Bestman M, Amsler-Kepalaite Z, Visscher J, Vermeij I, van Krimpen M (2014) Laying hen performance in different production systems; why do they differ and how to close the gap? Results of discussions with groups of farmers in the Netherlands, Switzerland and France, benchmarking and model calculations. Archiv für Geflügelkunde 78, 1–10.
Leeson S, Summers JD (2005) ‘Commercial poultry production.’ 3rd edn. (University Books: Guelph, Canada)
Leeson S, Summers JD (2009) ‘Commercial poultry nutrition.’ 3rd edn. (Nottingham University Press: Nottingham, UK)
Leeson S, Summers JD, Moran ET (1976) Avian water metabolism: a review. World’s Poultry Science Journal 32, 185–195.
| Avian water metabolism: a review.Crossref | GoogleScholarGoogle Scholar |
Leeson S, Diaz GJ, Summers JD (1995) ‘Poultry metabolic disorders and mycotoxins.’ (University Books: Guelph, Canada)
Leinonen I, Williams AG, Wiseman J, Guy J, Kyriazakis I (2012) Predicting the environmental impacts of chicken systems in the United Kingdom through a life cycle assessment: egg production systems Poultry Science 91, 26–40.
| Predicting the environmental impacts of chicken systems in the United Kingdom through a life cycle assessment: egg production systemsCrossref | GoogleScholarGoogle Scholar | 22184425PubMed |
Lemme A (2009) Amino acid recommendations for laying hens. Lohmann Information 44, 21–32.
Li X, Bryden WL, Zhang D (2016a) ‘Available phosphorus requirement of laying hens. 1UQ101.’ Available at https://www.aecl.org/assets/RD-files/Outputs-2/160115-1UQ101-Final-Report-.pdf [Verified 22 September 2019]
Li X, Zhang D, Yang TY, Bryden WL (2016b) Phosphorus bioavailability: a key aspect for conserving this critical animal feed resource with reference to broiler nutrition. Agriculture 6, 25
| Phosphorus bioavailability: a key aspect for conserving this critical animal feed resource with reference to broiler nutrition.Crossref | GoogleScholarGoogle Scholar |
Li X, Zhang D, Bryden WL (2017) Calcium and phosphorus nutrition of poultry: are modern diets formulated in excess? Animal Production Science 57, 2304–2310.
| Calcium and phosphorus nutrition of poultry: are modern diets formulated in excess?Crossref | GoogleScholarGoogle Scholar |
Lin H, Jiao HC, Buyse J, Decuypere E (2006) Strategies for preventing heat stress in poultry. World’s Poultry Science Journal 62, 71–85.
| Strategies for preventing heat stress in poultry.Crossref | GoogleScholarGoogle Scholar |
Lootens P, Van Waes J, Carlier L (2004) Effect of a short photoinhibition stress on photosynthesis, chlorophyll a fluorescence, and pigment contents of different maize cultivars. Can a rapid and objective stress indicator be found? Photosynthetica 42, 187–192.
| Effect of a short photoinhibition stress on photosynthesis, chlorophyll a fluorescence, and pigment contents of different maize cultivars. Can a rapid and objective stress indicator be found?Crossref | GoogleScholarGoogle Scholar |
MacLeod M (2013) Nutrition-related opportunities and challenges of alternative poultry production systems. Lohmann Information 48, 23–28.
Mahdavi AH, Rahmani HR, Pourreza J (2005) Effect of probiotic supplements on egg quality and laying hen’s performance International Journal of Poultry Science 4, 488–492.
| Effect of probiotic supplements on egg quality and laying hen’s performanceCrossref | GoogleScholarGoogle Scholar |
Mátéová S, Gaálová M, Šály J, Fialkovičová M (2009) Investigation of the effect of probiotics and potentiated probiotics on productivity of laying hens Czech Journal of Animal Science 54, 24–30.
| Investigation of the effect of probiotics and potentiated probiotics on productivity of laying hensCrossref | GoogleScholarGoogle Scholar |
Mathlouthi N, Larbier M, Mohamed MA, Lessire M (2002) Performance of laying hens fed wheat, wheat–barley or wheat–barley–wheat bran based diets supplemented with xylanase. Canadian Journal of Animal Science 82, 193–199.
| Performance of laying hens fed wheat, wheat–barley or wheat–barley–wheat bran based diets supplemented with xylanase.Crossref | GoogleScholarGoogle Scholar |
Maurice DV (1989) Salinity of drinking water and performance of chickens. In ‘Proceedings of the Georgia nutrition conference’, University of Georgia, Athens, Georgia, USA. pp. 140–144.
Meriwether LS, Humphrey BD, Peterson DG, Klasing KC, Koutsos EA (2010) Lutein exposure, in ovo or in the diet, reduces parameters of inflammation in the liver and spleen laying-type chicks (Gallus gallus domesticus). Journal of Animal Physiology and Animal Nutrition 94, e115–e122.
| Lutein exposure, in ovo or in the diet, reduces parameters of inflammation in the liver and spleen laying-type chicks (Gallus gallus domesticus).Crossref | GoogleScholarGoogle Scholar | 20546071PubMed |
Metges CC (2000) Contribution of microbial amino acids to amino acid homeostasis of the host. The Journal of Nutrition 130, 1857S–1864S.
| Contribution of microbial amino acids to amino acid homeostasis of the host.Crossref | GoogleScholarGoogle Scholar | 10867063PubMed |
Moen B, Rudi K, Svihus B, Skanseng B (2012) Reduced spread of Campylobacter jejuni in broiler chickens by stimulating the bird’s natural barriers Journal of Applied Microbiology 113, 1176–1183.
| Reduced spread of Campylobacter jejuni in broiler chickens by stimulating the bird’s natural barriersCrossref | GoogleScholarGoogle Scholar | 22817452PubMed |
Montagne L, Pluske JR, Hampson DJ (2003) A review of interactions between dietary fibre and the intestinal mucosa, and their consequences on digestive health in young non-ruminant animals Animal Feed Science and Technology 108, 95–117.
| A review of interactions between dietary fibre and the intestinal mucosa, and their consequences on digestive health in young non-ruminant animalsCrossref | GoogleScholarGoogle Scholar |
Mousavi SN, Afsar A, Khalaji S, Abbasi M (2018) Estimation of digestible tryptophan:lysine ratios for maximum performance, egg quality and welfare of white-egg-laying hens by fitting the different non-linear models Journal of Applied Animal Research 46, 411–416.
| Estimation of digestible tryptophan:lysine ratios for maximum performance, egg quality and welfare of white-egg-laying hens by fitting the different non-linear modelsCrossref | GoogleScholarGoogle Scholar |
National Research Council (NRC) (1974) ‘Nutrients and toxic substances in water for livestock and poultry.’ (The National Academies Press: Washington, DC, USA)
National Research Council (NRC) (1994) ‘Nutrient requirements of poultry.’ 9th revised edn. (The National Academies Press: Washington, DC, USA)
Neijat M, House JD, Guenter W, Kebreab E (2011) Calcium and phosphorus dynamics in commercial laying hens housed in conventional or enriched cage systems. Poultry Science 90, 2383–2396.
| Calcium and phosphorus dynamics in commercial laying hens housed in conventional or enriched cage systems.Crossref | GoogleScholarGoogle Scholar | 21934024PubMed |
Neill AR, McKenzie RA, Schultz K, Connor JK (1975) Reticulolysis and fatty liver syndrome commercial laying fowls. Australian Veterinary Journal 51, 104–105.
| Reticulolysis and fatty liver syndrome commercial laying fowls.Crossref | GoogleScholarGoogle Scholar | 1164274PubMed |
Newman S, Leeson S (1998) Effect of housing birds in cages or an aviary system on bone characteristics Poultry Science 77, 1492–1496.
| Effect of housing birds in cages or an aviary system on bone characteristicsCrossref | GoogleScholarGoogle Scholar | 9776056PubMed |
Noormohammadi AH (2021) Welfare implications of bacterial and viral infectious diseases for laying hens. Animal Production Science.
| Welfare implications of bacterial and viral infectious diseases for laying hens.Crossref | GoogleScholarGoogle Scholar |
Nys Y (2000) Dietary carotenoids and egg yolk coloration Archiv für Geflügelkunde 64, 45–54.
Nys Y, Guyot N (2011) Egg formation and chemistry. In ‘Improving the safety and quality of eggs and egg products, vol. 1: egg chemistry, production and consumption’. (Eds Y Nys, M Bain, F Van Immerseel) pp. 81–132. (Woodhead Publishing: Cambridge, UK)
Nys Y, Le Roy N (2018) Calcium homeostasis and eggshell biomineralization in female chicken. In ‘Vitamin D, vol. 1: biochemistry, physiology and diagnostics’. 4th edn. (Ed. D Feldman) pp. 361–382. (Academic Press: San Diego, CA, USA)
Pan D, Yu Z (2014) Intestinal microbiome of poultry and its interaction with host and diet Gut Microbes 5, 108–119.
| Intestinal microbiome of poultry and its interaction with host and dietCrossref | GoogleScholarGoogle Scholar | 24256702PubMed |
Pearson AW, Butler EJ, Curtis RF, Fenwick GR, Hobson-Frohock A, Land DG, Hall SA (1978) Effects of rapeseed meal on laying hens (Gallus domesticus) in relation to fatty liver-haemorrhagic syndrome and egg taint. Research in Veterinary Science 25, 307–313.
Peguri A, Coon C (1991) Effect of temperature and dietary energy on layer performance Poultry Science 70, 126–138.
| Effect of temperature and dietary energy on layer performanceCrossref | GoogleScholarGoogle Scholar | 2017407PubMed |
Peguri A, Coon C (1993) Effect of feather coverage and temperature on layer performance Poultry Science 72, 1318–1329.
| Effect of feather coverage and temperature on layer performanceCrossref | GoogleScholarGoogle Scholar |
Persyn KE, Xin H, Nettleton D, Ikeguchi A, Gates RS (2004) Feeding behaviors of laying hens with or without beak trimming Proceedings of the Transactions of the American Society of Agricultural Engineers 47, 591–596.
| Feeding behaviors of laying hens with or without beak trimmingCrossref | GoogleScholarGoogle Scholar |
Pines M, Reshef R (2015) Poultry bone development and bone disorders. In ‘Sturkie’s avian physiology’. 6th edn. (Ed. CG Scanes) pp. 367–377. (Elsevier Inc.: New York, NY, USA)
Portella F, Caston L, Leeson S (1988) Apparent feed particle size preference by laying hens Canadian Journal of Animal Science 68, 915–922.
| Apparent feed particle size preference by laying hensCrossref | GoogleScholarGoogle Scholar |
Pottgüter R (2016) Feeding laying hens to 100 weeks of age. Lohmann Information 50, 18–21.
Pourreza J, Nili N, Edriss MA (1994) Relationship of plasma calcium and phosphorus to the shell quality of laying hens receiving saline drinking water British Poultry Science 35, 755–762.
| Relationship of plasma calcium and phosphorus to the shell quality of laying hens receiving saline drinking waterCrossref | GoogleScholarGoogle Scholar | 7719739PubMed |
Pousga S, Boly H, Ogle B (2005) Choice feeding of poultry: a review. Livestock Research for Rural Development 17, 45
Rao KS, Roland DA (1990) In vivo limestone solubilization in commercial Leghorns: role of dietary calcium level, limestone particle size, in vitro limestone solubility rate, and the calcium status of the hen Poultry Science 69, 2170–2176.
| In vivo limestone solubilization in commercial Leghorns: role of dietary calcium level, limestone particle size, in vitro limestone solubility rate, and the calcium status of the henCrossref | GoogleScholarGoogle Scholar | 2084675PubMed |
Rault J-L, Shelby Cree S, Hemsworth P (2016) The effects of water deprivation on the behaviour of laying hens Poultry Science 95, 473–481.
| The effects of water deprivation on the behaviour of laying hensCrossref | GoogleScholarGoogle Scholar | 26628345PubMed |
Riczu CM, Saunders-Blades JL, Yngvesson AK, Robison FE, Korver DR (2004) End-of-cycle bone quality in white- and brown-egg laying hens Poultry Science 83, 375–383.
| End-of-cycle bone quality in white- and brown-egg laying hensCrossref | GoogleScholarGoogle Scholar | 15049489PubMed |
Riddell C, Helmboldt CF, Singsen EP (1969) A histologic study of medullary bone of laying hens under different diet and housing conditions. Avian Diseases 13, 163–170.
| A histologic study of medullary bone of laying hens under different diet and housing conditions.Crossref | GoogleScholarGoogle Scholar | 5775408PubMed |
Roberts JR (2004) Factors determining internal egg quality and egg shell quality in laying hens. Journal of Poultry Science 41, 161–177.
| Factors determining internal egg quality and egg shell quality in laying hens.Crossref | GoogleScholarGoogle Scholar |
Roberts JR, Balnave D (1992) The physiological basis of poor eggshell quality in laying hens: the effect of saline drinking water on electrolyte balance and renal function. Journal of Animal Physiology and Animal Nutrition 68, 197–204.
| The physiological basis of poor eggshell quality in laying hens: the effect of saline drinking water on electrolyte balance and renal function.Crossref | GoogleScholarGoogle Scholar |
Röhe I, Ruhnke I, Knorr F, Mader A, Goodarzi Boroojeni F, Löwe R, Zentek J (2014) Effects of grinding method, particle size, and physical form of the diet on gastrointestinal morphology and jejunal glucose transport in laying hens Poultry Science 93, 2060–2068.
| Effects of grinding method, particle size, and physical form of the diet on gastrointestinal morphology and jejunal glucose transport in laying hensCrossref | GoogleScholarGoogle Scholar | 24902702PubMed |
Rostagno HS (Ed.) (2005) ‘Brazilian tables for poultry and swine: Composition of feedstuffs and nutritional requirements.’ 2nd edn. (Departamento de Zootecnia, Universidade Federal de Vicosa: Vicosa, Brazil)
Rostagno HS, Albino LFT, Donzele JL, Gomes PC, de Oliveira RF, Lopes DC, Ferreira AS, Barreto SLT, Euclides RF (2011) ‘Brazilian tables for poultry and swine: composition of feedstuffs and nutritional requirements.’ 3rd edn. (Ed. HS Rostagno) (Departamento de Zootecnia, Universidade Federal de Vicosa: Vicosa, Brazil)
Ruhnke I, Röhe I, Krämer C, Goodarzi Boroojeni F, Knorr F, Mader A, Hafeez A, Löwe R, Zentek J (2015a) Effect of feed particle size, milling method, and heat treatment on performance, apparent ileal digestibility, and pH of the digesta in laying hens Poultry Science 94, 692–699.
| Effect of feed particle size, milling method, and heat treatment on performance, apparent ileal digestibility, and pH of the digesta in laying hensCrossref | GoogleScholarGoogle Scholar | 25717083PubMed |
Ruhnke I, Cowling G, Sommerlad M, Swick R, Choct M (2015b) Gut impaction in free-range hens. Proceedings Australian Poultry Science Symposium 26, 242–244.
Ruhnke IJ, Boshoff J, Cristiani IV, Schneider D, Welch M, Sibanda TZ, Kolakshyapati M (2019) Free-range laying hens: using technology to show the dynamics and impact of hen movement. Animal Production Science 59, 2046–2056.
| Free-range laying hens: using technology to show the dynamics and impact of hen movement.Crossref | GoogleScholarGoogle Scholar |
Saeed M, Babazadeh D, Naveed M, Arain MA, Hassan FU, Chao S (2017) Reconsidering betaine as a natural anti-heat stress agent in poultry industry: a review Tropical Animal Health and Production 49, 1329–1338.
| Reconsidering betaine as a natural anti-heat stress agent in poultry industry: a reviewCrossref | GoogleScholarGoogle Scholar | 28733762PubMed |
Safaa HM, Jiminez-Moreno E, Valencia DG, Frikha M, Serrano MP, Mateos GG (2009) Effect of main cereal of the diet and particle size of the cereal on productive performance and egg quality of brown egg-laying hens in early phase of production Poultry Science 88, 608–614.
| Effect of main cereal of the diet and particle size of the cereal on productive performance and egg quality of brown egg-laying hens in early phase of productionCrossref | GoogleScholarGoogle Scholar | 19211532PubMed |
Santos FBO, Sheldon BW, Santos AA, Ferket PR (2008) Influence of housing system, grain type, and particle size on Salmonella colonization and shedding of broilers fed triticale or corn-soybean meal diets Poultry Science 87, 405–420.
| Influence of housing system, grain type, and particle size on Salmonella colonization and shedding of broilers fed triticale or corn-soybean meal dietsCrossref | GoogleScholarGoogle Scholar |
Scanes CG (2016) Biology of stress in poultry with emphasis on glucocorticoids and the heterophil to lymphocyte ratio Poultry Science 95, 2208–2215.
| Biology of stress in poultry with emphasis on glucocorticoids and the heterophil to lymphocyte ratioCrossref | GoogleScholarGoogle Scholar | 27252367PubMed |
Scott P (2015) ‘National farm biosecurity technical manual for egg production.’ (Animal Heath Australia and Australian Egg Corporation: Sydney, NSW, Australia)
Scott ML, Nesheim MC, Young RJ (1969) ‘Nutrition of the chicken.’ (ML Scott & Associates: Ithaca, NY, USA)
Shang Y, Kumar S, Oakley B, Kim WK (2018) Chicken gut microbiota: importance and detection technology. Frontiers in Veterinary Science 5,
| Chicken gut microbiota: importance and detection technology.Crossref | GoogleScholarGoogle Scholar | 30406117PubMed |
Shini S, Huff GR, Shini A, Kaiser P (2010) Understanding stress-induced immunosuppression: exploration of cytokine and chemokine gene profiles in chicken peripheral leukocytes Poultry Science 89, 841–851.
| Understanding stress-induced immunosuppression: exploration of cytokine and chemokine gene profiles in chicken peripheral leukocytesCrossref | GoogleScholarGoogle Scholar | 20308420PubMed |
Shini S, Shini A, Blackall PJ (2013) The potential for probiotics to prevent reproductive tract lesions in free-range laying hens. Animal Production Science 53, 1298–1308.
| The potential for probiotics to prevent reproductive tract lesions in free-range laying hens.Crossref | GoogleScholarGoogle Scholar |
Shini A, Shini S, Bryden WL (2019) Fatty liver haemorrhagic syndrome occurrence in laying hens: impact of production system Avian Pathology 48, 25–34.
| Fatty liver haemorrhagic syndrome occurrence in laying hens: impact of production systemCrossref | GoogleScholarGoogle Scholar | 30345810PubMed |
Shini S, Shini A, Bryden WL (2020a) Unravelling fatty liver haemorrhagic syndrome 1. Oestrogen and inflammation Avian Pathology 49, 87–98.
| Unravelling fatty liver haemorrhagic syndrome 1. Oestrogen and inflammationCrossref | GoogleScholarGoogle Scholar | 31565961PubMed |
Shini S, Shini A, Bryden WL (2020b) Unravelling fatty liver haemorrhagic syndrome 2. Inflammation and pathophysiology. Avian Pathology 49, 131–143.
| Unravelling fatty liver haemorrhagic syndrome 2. Inflammation and pathophysiology.Crossref | GoogleScholarGoogle Scholar | 31621393PubMed |
Shinki T, Tanaka H, Takito J, Yamaguchi A, Nakamu Y, Yoshiki S (1991) Putrescine is involved in the vitamin D action in chick intestine. Gastroenterology 100, 113–122.
| Putrescine is involved in the vitamin D action in chick intestine.Crossref | GoogleScholarGoogle Scholar | 1983813PubMed |
Sibanda TZ, Walkden-Brown SW, Kolakshyapatia M, Dawson B, Schneider D, Welch M, Iqbal Z, Cohen-Barnhouse A, Morgan NK, Boshoff J, Ruhnke I (2020a) Flock use of the range is associated with the use of different components of a multi-tier aviary system in commercial free-range laying hens British Poultry Science 61, 97–106.
| Flock use of the range is associated with the use of different components of a multi-tier aviary system in commercial free-range laying hensCrossref | GoogleScholarGoogle Scholar | 31661978PubMed |
Sibanda TZ, Flavel R, Kolakshyapati M, Schneider D, Welch M, Ruhnke I (2020b) The association between range usage and tibial quality in commercial free-range laying hens. British Poultry Science
| The association between range usage and tibial quality in commercial free-range laying hens.Crossref | GoogleScholarGoogle Scholar | 32336127PubMed |
Sinanoglou VJ, Strati IF, Miniadis-Meimaroglou S (2011) Lipid, fatty acid and carotenoid content of edible egg yolks from avian species: a comparative study Food Chemistry 124, 971–977.
| Lipid, fatty acid and carotenoid content of edible egg yolks from avian species: a comparative studyCrossref | GoogleScholarGoogle Scholar |
Singh M, Cowieson AJ (2013) Range use and pasture consumption in free-range poultry production. Animal Production Science 53, 1202–1208.
| Range use and pasture consumption in free-range poultry production.Crossref | GoogleScholarGoogle Scholar |
Singh M, Hernandez CE, Lee C, Hinch G, Cowieson AJ (2016) Wanderers versus stay at home: who has the better guts? Proceedings of the Australian Poultry Science Symposium 27, 78–81.
Singh M, Ruhnke I, DeKoning C, Drake K, Skerman A, Hinch GN, Glatz PC (2017) Free-range poultry survey: demographics and practices of semi-intensive free-range farming systems in Australia with an outdoor stocking density of ≤1500 hens/hectare PLoS One 12, e0187057
| Free-range poultry survey: demographics and practices of semi-intensive free-range farming systems in Australia with an outdoor stocking density of ≤1500 hens/hectareCrossref | GoogleScholarGoogle Scholar | 29206228PubMed |
Sirovnik J, Würbel H, Toscano MJ (2018) Feeder space affects access to the feeder, aggression, and feed conversion in laying hens in an aviary system Applied Animal Behaviour Science 198, 75–82.
| Feeder space affects access to the feeder, aggression, and feed conversion in laying hens in an aviary systemCrossref | GoogleScholarGoogle Scholar |
Smith TK, Tapia-Salazar M, Cruz-Suarez LE, Ricque-Marie D (2000) Feed-borne biogenic amines: natural toxicants or growth promotors? In ‘Avances en nutrición acuícola V. Memorias del V simposium internacional de nutrición acuícola’, Mérida, Yucatán, Mexico. (Eds LE Cruz-Suarez, D Ricque-Marie, M Tapia-Salazar, MA Olvera-Novoa, R Civera-Cerecedo) pp. 24–32. (Universidad Autónoma de Nuevo León: Monterrey, NL, México)
Soares L, Sakomura NK, Dorigam JCP, Liebert F, Sunder A, Nascimento MQD, Leme BB (2019) Optimal in‐feed amino acid ratio for laying hens based on deletion method. Journal of Animal Physiology and Animal Nutrition 103, 170–181.
| Optimal in‐feed amino acid ratio for laying hens based on deletion method.Crossref | GoogleScholarGoogle Scholar | 30478953PubMed |
Sonnenburg JL, Bäckhed F (2016) Diet–microbiota interactions as moderators of human metabolism. Nature 535, 56–64.
| Diet–microbiota interactions as moderators of human metabolism.Crossref | GoogleScholarGoogle Scholar | 27383980PubMed |
Squires EJ, Leeson S (1988) Aetiology of fatty liver syndrome in laying hens. The British Veterinary Journal 144, 602–609.
| Aetiology of fatty liver syndrome in laying hens.Crossref | GoogleScholarGoogle Scholar | 3060215PubMed |
Steenfeldt S, Kjaer JB, Engberg RM (2007) Effect of feeding silages or carrots as supplements to laying hens on production performance, nutrient digestibility, gut structure, gut microflora and feather pecking behaviour British Poultry Science 48, 454–468.
| Effect of feeding silages or carrots as supplements to laying hens on production performance, nutrient digestibility, gut structure, gut microflora and feather pecking behaviourCrossref | GoogleScholarGoogle Scholar | 17701499PubMed |
Stewart GD, Rudkin C, Shini S, Bryden WL (2006) Assessment of laying hens maintained in different housing systems. Final report for the Australian Egg Corporation. Available at https://www.australianeggs.org.au/dmsdocument/602-assessment-of-laying-hens-maintained-in-different-housing-systems [29 September, 2019]
Sugahara M (1995) Black vomit, gizzard erosion and gizzerosine. World’s Poultry Science Journal 51, 293–306.
| Black vomit, gizzard erosion and gizzerosine.Crossref | GoogleScholarGoogle Scholar |
Summers JD, Leeson S (1978) Dietary selection of protein and energy by pullets and broilers British Poultry Science 19, 425–430.
| Dietary selection of protein and energy by pullets and broilersCrossref | GoogleScholarGoogle Scholar |
Summers JD, Leeson S (1994) Laying hen performance as influenced by protein intake to sixteen weeks of age and body weight at point of lay Poultry Science 73, 495–501.
| Laying hen performance as influenced by protein intake to sixteen weeks of age and body weight at point of layCrossref | GoogleScholarGoogle Scholar | 8202428PubMed |
Surai PF, Sparks NHC (2001) Designer eggs: from improvement of egg composition to functional food Trends in Food Science & Technology 12, 7–16.
| Designer eggs: from improvement of egg composition to functional foodCrossref | GoogleScholarGoogle Scholar |
Surai PF, Speake BK, Sparks NHC (2001) Carotenoids in avian nutrition and embryonic development. 1. Absorption, availability and levels in plasma and egg yolk Journal of Poultry Science 38, 1–27.
| Carotenoids in avian nutrition and embryonic development. 1. Absorption, availability and levels in plasma and egg yolkCrossref | GoogleScholarGoogle Scholar |
Svihus B (2006) The role of feed processing on gastrointestinal function and health in poultry. In ‘Avian gut function in health and disease’. (Ed. GA. Perry) pp. 183–194. (CABI Publishing: Wallingford, UK)
Svihus B (2011) The gizzard: function, influence of diet structure and effects on nutrient availability World’s Poultry Science Journal 67, 207–224.
| The gizzard: function, influence of diet structure and effects on nutrient availabilityCrossref | GoogleScholarGoogle Scholar |
Svihus B, Hetland H (2001) Ileal starch digestibility in growing broiler chickens fed on a wheat-based diet is improved by mash feeding, dilution with cellulose or whole wheat inclusion British Poultry Science 42, 633–637.
| Ileal starch digestibility in growing broiler chickens fed on a wheat-based diet is improved by mash feeding, dilution with cellulose or whole wheat inclusionCrossref | GoogleScholarGoogle Scholar | 11811915PubMed |
Svihus B, Juvik E, Hetland H, Krogdahl A (2004) Causes for improvement in nutritive value of broiler chicken diets with whole wheat instead of ground wheat British Poultry Science 45, 55–60.
| Causes for improvement in nutritive value of broiler chicken diets with whole wheat instead of ground wheatCrossref | GoogleScholarGoogle Scholar | 15115201PubMed |
Svihus B, Sacranie A, Denstadli V, Choct M (2010) Nutrient utilization and functionality of the anterior digestive tract caused by intermittent feeding and inclusion of whole wheat in diets for broiler chickens Poultry Science 89, 2617–2625.
| Nutrient utilization and functionality of the anterior digestive tract caused by intermittent feeding and inclusion of whole wheat in diets for broiler chickensCrossref | GoogleScholarGoogle Scholar | 21076099PubMed |
Tasman-Jones C, Owen RL, Jones AL (1982) Semipurified dietary fiber and small-bowel morphology in rats Digestive Diseases and Sciences 27, 519–524.
| Semipurified dietary fiber and small-bowel morphology in ratsCrossref | GoogleScholarGoogle Scholar | 6282549PubMed |
Tauson R, Svensson SA (1980) Influence of plumage condition on the hen’s feed requirement Swedish Journal of Agricultural Research 10, 35–39.
Teixeira RSC, Romão JM, Câmara SR, Oliveira WF, Niza MH, Sobral R, Siqueira AA, Cardoso WM (2006) Indução a muda forçada em galinhas d’angola (Numida meleagris) através do óxido de zinco. Brazilian Journal of Veterinary Research and Animal Science 43, 448–455.
| Indução a muda forçada em galinhas d’angola (Numida meleagris) através do óxido de zinco.Crossref | GoogleScholarGoogle Scholar |
Tellez G, Higgins S, Donoghue A, Hargis B (2006) Digestive physiology and the role of microorganisms. Journal of Applied Poultry Research 15, 136–144.
| Digestive physiology and the role of microorganisms.Crossref | GoogleScholarGoogle Scholar |
Thiele HH (2012) Management tools to influence egg weight in commercial layers. Lohmann Information 47, 21–31.
Tilbrook AJ, Fisher AD (2020) Stress, health and the welfare of laying hens. Animal Production Science.
| Stress, health and the welfare of laying hens.Crossref | GoogleScholarGoogle Scholar |
Tiller H (2001) Nutrition and animal welfare in egg production systems. In ‘13th European symposium poultry nutrition 13’. pp. 226–232. (Blankenberge, Belgium)
Underwood G, Andrews D, Phung T (2021) Advances in genetic selection and breeder practice improve commercial layer hen welfare. Animal Production Science
| Advances in genetic selection and breeder practice improve commercial layer hen welfare.Crossref | GoogleScholarGoogle Scholar | (In press).
van Krimpen MM, Kwakkel RP, Van der Peet-Schwering CM, DenHartog LA, Verstegen MW (2008) Low dietary energy concentration, high nonstarch polysaccharide concentration, and coarse particle sizes of nonstarch polysaccharides affect the behavior of feather pecking-prone laying hens Poultry Science 87, 485–496.
| Low dietary energy concentration, high nonstarch polysaccharide concentration, and coarse particle sizes of nonstarch polysaccharides affect the behavior of feather pecking-prone laying hensCrossref | GoogleScholarGoogle Scholar | 18281575PubMed |
van Krimpen MM, Leenstra F, Maurer V, Bestman M (2016) How to fulfill EU requirements to feed organic laying hens 100% organic ingredients Journal of Applied Poultry Research 25, 129–138.
| How to fulfill EU requirements to feed organic laying hens 100% organic ingredientsCrossref | GoogleScholarGoogle Scholar |
Vilariño M, Picard ML, Melcion JP, Faure JW (1996) Behavioural adaption of laying hens to dilution of diets under mash and pellet form British Poultry Science 37, 895–907.
| Behavioural adaption of laying hens to dilution of diets under mash and pellet formCrossref | GoogleScholarGoogle Scholar | 9034580PubMed |
Villageliũ DN, Lyte M (2017) Microbial endocrinology: why the intersection of microbiology and neurobiology matters to poultry health. Poultry Science 96, 2501–2508.
| Microbial endocrinology: why the intersection of microbiology and neurobiology matters to poultry health.Crossref | GoogleScholarGoogle Scholar | 29050443PubMed |
Waldroup PW, Hellwig HM (1995) Methionine and total sulfur amino acid requirements influenced by stage of production. Journal of Applied Poultry Research 4, 283–292.
| Methionine and total sulfur amino acid requirements influenced by stage of production.Crossref | GoogleScholarGoogle Scholar |
Walker A, Gordon S (2003) Intake of nutrients from pasture by poultry The Proceedings of the Nutrition Society 62, 253–256.
| Intake of nutrients from pasture by poultryCrossref | GoogleScholarGoogle Scholar | 14506872PubMed |
Weber GM (2009) Improvement of flock productivity through supply of vitamins for higher laying performance and better egg quality. World’s Poultry Science Journal 65, 443–457.
| Improvement of flock productivity through supply of vitamins for higher laying performance and better egg quality.Crossref | GoogleScholarGoogle Scholar |
Weber G, Michalczuk M, Huyghebaert G, Juin H, Kwakernaak C, Gracia M (2012) Effects of a blend of essential oil compounds and benzoic acid on performance of broiler chickens as revealed by a meta-analysis of 4 growth trials in various locations Poultry Science 91, 2820–2828.
| Effects of a blend of essential oil compounds and benzoic acid on performance of broiler chickens as revealed by a meta-analysis of 4 growth trials in various locationsCrossref | GoogleScholarGoogle Scholar | 23091138PubMed |
Webster AJF (2001) Farm animal welfare: the five freedoms and the free market Veterinary Journal (London, UK) 161, 229–237.
| Farm animal welfare: the five freedoms and the free marketCrossref | GoogleScholarGoogle Scholar |
Webster J (2016) Animal welfare: freedoms, dominions and ‘a life worth living’ Animals (Basel) 6, 35
| Animal welfare: freedoms, dominions and ‘a life worth living’Crossref | GoogleScholarGoogle Scholar |
Whitehead CC (2004) Overview of bone biology in the egg-laying hen. Poultry Science 83, 193–199.
| Overview of bone biology in the egg-laying hen.Crossref | GoogleScholarGoogle Scholar | 14979569PubMed |
Whitehead CC, Fleming RH (2000) Osteoporosis in cage layers. Poultry Science 79, 1033–1041.
| Osteoporosis in cage layers.Crossref | GoogleScholarGoogle Scholar | 10901207PubMed |
Wils-Plotz EL, Klasing KC (2017) Effects of immunomodulatory nutrients on growth performance and immune-related gene expression in layer chicks challenged with lipopolysaccharide. Poultry Science 96, 548–555.
| Effects of immunomodulatory nutrients on growth performance and immune-related gene expression in layer chicks challenged with lipopolysaccharide.Crossref | GoogleScholarGoogle Scholar | 28394396PubMed |
Wu YB, Ravindran V, Thomas DG, Birtles MJ, Hendriks WH (2004) Influence of method of whole wheat inclusion and xylanase supplementation on the performance, apparent metabolisable energy, digestive tract measurements and gut morphology of broilers British Poultry Science 45, 385–394.
| Influence of method of whole wheat inclusion and xylanase supplementation on the performance, apparent metabolisable energy, digestive tract measurements and gut morphology of broilersCrossref | GoogleScholarGoogle Scholar | 15327125PubMed |
Yan F, Murugesan G, Cheng H (2019) Effects of probiotic supplementation on performance traits, bone mineralization, cecal microbial composition, cytokines and corticosterone in laying hens. Animal 13, 33–41.
| Effects of probiotic supplementation on performance traits, bone mineralization, cecal microbial composition, cytokines and corticosterone in laying hens.Crossref | GoogleScholarGoogle Scholar | 29785889PubMed |
Yeates NTM, Lee DHK, Hines RJG (1941) Reactions of domestic fowls to hot atmospheres. Proceedings of the Royal Society of Queensland 53, 105–128.
Yegani M, Korver DR (2008) Factors affecting intestinal health in poultry Poultry Science 87, 2052–2063.
| Factors affecting intestinal health in poultryCrossref | GoogleScholarGoogle Scholar | 18809868PubMed |
Yokhana JS, Parkinson G, Frankel TL (2016) Effect of insoluble fiber supplementation applied at different ages on digestive organ weight and digestive enzymes of layer-strain poultry Poultry Science 95, 550–559.
| Effect of insoluble fiber supplementation applied at different ages on digestive organ weight and digestive enzymes of layer-strain poultryCrossref | GoogleScholarGoogle Scholar | 26574026PubMed |
Yoselewitz I, Balnave D (1990) Strain responses in egg shell quality to saline drinking water. Proceedings of the Australian Poultry Science Symposium 2, 102.
Yoselewitz I, Klein E, Malka I, Pinchasov Y, Levav N, Katz T (1993) Influence of drinking water quality on layer performance and egg shell quality. In ‘Proceedings of the 31st annual convention, Israel Branch, World’s Poultry Science Association’, Tel Aviv, Israel. pp. 58–59.
Yousaf M, Chaudhry A (2008) History, changing scenarios and future strategies to induce moulting in laying hens. World’s Poultry Science Journal 64, 65–75.
| History, changing scenarios and future strategies to induce moulting in laying hens.Crossref | GoogleScholarGoogle Scholar |
Zhang D (1993) Shell gland function and egg shell quality. PhD Thesis, The University of Sydney, Sydney, NSW, Australia.
Zhang B, Coon CN (1997) The relationship of calcium intake, source, size, solubility in vitro and in vivo, and gizzard limestone retention in laying hens Poultry Science 76, 1702–1706.
| The relationship of calcium intake, source, size, solubility in vitro and in vivo, and gizzard limestone retention in laying hensCrossref | GoogleScholarGoogle Scholar | 9438285PubMed |