Associations between illness perceptions, distress, self-reported cognitive difficulties and cognitive performance after mild traumatic brain injury
Deborah L. Snell A * , Josh W. Faulkner
A * , Josh W. Faulkner  B , Jonathan A. Williman C , Noah D. Silverberg D E F , Alice Theadom G , Lois J. Surgenor H and Richard J. Siegert G
B , Jonathan A. Williman C , Noah D. Silverberg D E F , Alice Theadom G , Lois J. Surgenor H and Richard J. Siegert G
A
B
C
D
E
F
G
H
Abstract
To examine associations between subjective and objective cognitive problems, and factors potentially modifying these relationships, after mild traumatic brain injury (mTBI).
Treatment-seeking adults (n = 95) were assessed 6 weeks (Time 1) and then 6 months later (Time 2) after mTBI. Validated questionnaires assessed cognitive, emotional and somatic mTBI symptoms, distress, catastrophising, and beliefs about symptoms and recovery. Cognitive performance was measured using the National Institutes of Health Toolbox Cognition Battery. Using correlations and linear regression, we explored associations between psychological factors, objectively measured cognitive performance and self-reported cognitive symptoms.
There were only modest correlations between subjective cognitive symptoms and objective cognitive performance at assessment timepoints. In contrast, there were medium to large correlations between subjective cognitive symptoms, post-concussion symptom burden and psychological factors. Post-concussion symptom burden and beliefs about symptoms and recovery at Time 1 predicted persisting self-reported cognitive symptoms at Time 2.
High post-concussion symptom burden and non-recovery expectations may increase risk for persistent subjectively experienced cognitive symptoms. Our findings may guide targeted treatment efforts focusing on factors with potential to influence cognitive symptom reporting after mTBI.
Keywords: adults, cognitive outcomes, cognitive performance, expectations, mild traumatic brain injury (mTBI), National Institutes of Health Toolbox Cognition Battery, post-concussion symptoms, self reported cognitive symptoms.
Introduction
Recovery after mild traumatic brain injury (mTBI) is expected within the first few weeks to months after injury (Belanger et al. 2010; Karr et al. 2014; Iverson et al. 2019). However, for a subgroup, persisting cognitive, emotional and somatic symptoms, along with functional difficulties, continue beyond this timeframe. The size of this subgroup is contentious and estimates vary across studies, ranging from 13%, (Schneider et al. 2022) to >50% of cases (Theadom et al. 2016; Dikmen et al. 2017; Theadom et al. 2017; Cancelliere et al. 2023). For this subgroup, persisting symptoms can become increasingly interrelated, mutually amplifying and reinforcing with time (Iverson 2019). Many factors can contribute to the development and maintenance of this cycle, including acute biological impacts of the injury itself, as well as a range of interacting pre-, peri- and early post-injury cognitive, psychological and psychosocial factors (Karr et al. 2014; Faulkner and Snell 2023; Silverberg and Mikolić 2023).
Cognitive signs and symptoms early after mTBI are common, with potential to impact functioning, and are a frequently identified source of distress (Brands et al. 2019; Levy et al. 2023). There seems to be good agreement that mTBI can, at least temporarily, affect cognitive function, but that cognitive improvement occurs with time (McCrea et al. 2009). A single mTBI is not thought to be associated with a high incidence of chronic cognitive impairment (Iverson et al. 2019). Several systematic reviews and meta-analyses evaluating objectively measured cognitive outcomes among people with mTBI provide strong evidence for recovery of cognitive function within early weeks to months (Binder et al. 1997; Schretlen and Shapiro 2003; Pertab et al. 2009; Boone et al. 2024). Additionally, studies using neuroimaging modalities to isolate diagnostic and prognostic biomarkers suggest that for some, occult brain changes occurring after mTBI may contribute to cognitive difficulties (Biagianti et al. 2020). However, determining cognitive recovery after mTBI is complex, with no agreed on criteria sets for defining what cognitive recovery means. This is further complicated by the range of factors that can influence cognitive function. Sleep, post-concussion symptom burden, expectations and anxiety, among others, can all affect cognitive performance (Silverberg et al. 2020; Pieroth 2022).
There is also a growing body of evidence noting a discrepancy between self-reported cognitive symptoms and objective cognitive performance after mTBI (French et al. 2014; Stenberg et al. 2020; Vos et al. 2020). That is, with time, objectively assessed cognitive performances can improve and fall within expected ranges, but subjectively experienced cognitive symptoms persist. Such outcomes can be confusing for both clinicians and consumers of health services (Snell et al. 2017). Factors that might influence the reduction or persistence of self-reported cognitive symptoms after mTBI remain unclear. A small number of studies have shown self-reported cognitive difficulties after mTBI are more closely associated with psychological factors, such as depression and anxiety, than with objective cognitive performance, and this relationship could be bidirectional (Vos et al. 2020). A recent study by Stenberg et al. (2020) showed that change in self-reported cognitive symptoms over 3 months after mTBI was more closely associated with changes in emotional and somatic symptoms than with changes in objective cognitive performance. We have previously shown that along with general distress and depression, injury expectations and responses to symptoms impact mTBI symptomatic outcomes (Snell et al. 2015). Furthermore, a recent study demonstrated catastrophising was associated with higher levels of cognitive symptom-reporting relative to objectively measured cognitive performance (Shi et al. 2024). Unravelling these complex relationships and exploring factors beyond mood and anxiety that influence a person’s experience of their cognitive functioning after mTBI may guide treatment efforts and targeting of factors that influence subjectively experienced cognitive symptoms. These additional factors include symptom and recovery expectations, and catastrophising.
Study objectives
We aimed to investigate relationships between self-reported cognitive symptoms and objective cognitive performances after mTBI. Specifically, we were interested in: (i) associations between self-reported cognitive symptoms and objective cognitive performance at 6 weeks (Time 1) and 6–9 months (Time 2) after mTBI, and (ii) which demographic, injury or psychological factors were associated with persisting cognitive symptoms 6–9 months after mTBI.
Methods
Design and setting
This is a secondary analysis of data collected as part of a prospective observational study examining impacts of psychological factors on recovery after mTBI. Participants were recruited from five outpatient clinics providing rehabilitation services for mTBI patients across both the North and South Islands of New Zealand between February 2019 and June 2022. Participating clinics were funded by New Zealand’s government-funded no-fault injury insurance scheme with standard operating procedures for mTBI management across all clinics. This means all participants had an accepted injury claim for mTBI and no participants had to litigate or were involved in litigation. Main data were collected at enrolment (Time 1: mean = 6.0 weeks after injury (s.d. = 2.5), range 2–13 weeks) and 6 months later (Time 2: mean = 31.0 weeks post-injury (s.d. = 4.0), range 23–41 weeks). These timeframes were selected noting a large body of literature, including many robust systematic reviews, showing in the majority of cases a return to cognitive baseline is evident within the first 3–4 months after injury. Thus, at 6–9 months, persisting cognitive symptoms might be considered atypical (Boone et al. 2024). There is also evidence from various studies that improvement in general flattens after approximately 6 months post-injury (Machamer et al. 2022). In addition, monthly phone follow-ups were conducted in between Time 1 and 2 assessments to monitor symptomatic recovery. Data collection was undertaken remotely during periods of restriction and lockdown in New Zealand during the COVID-19 pandemic. This meant that any participant who was seen for a study visit during these periods did not have cognitive assessment data available, as these were completed face-to-face only. The present study focuses on secondary cognitive outcomes data, with primary outcomes of the study not yet published.
Ethical approval was received from New Zealand’s National Health and Disability Ethics Committee (ref 18/CEN/79).
Participants
Eligible consecutively referred participants were approached by a clinician from the outpatient clinic to invite participation. Eligibility criteria were: (1) aged ≥16 years, (2) sustained an mTBI according to World Health Organization Neurotrauma Taskforce criteria (Holm et al. 2005), (3) were <3 months post-injury at enrolment, (4) were fluent in English, (5) had no prior neurological condition or severe unstable medical condition (e.g. respiratory illness, cancer), including a past history of moderate-to-severe traumatic brain injury, and (6) had at least one set of cognitive assessment scores at either Time 1 or Time 2. There were no inclusion or exclusion criteria based on post-concussion or psychological symptoms.
Data collection and measures
Demographic and clinical data were collected via self-report, and included age at injury, sex, ethnicity, highest education level, mental health history and history of previous mTBI. Clinical variables included mechanism of injury, self-reported post-traumatic amnesia, Glasgow Coma Scale score at hospital arrival if seen on the day of injury, time from injury to the first study visit (Time 1) and time from injury to the second study visit (Time 2).
Cognitive functioning was assessed face-to-face at Times 1 and 2 using the National Institutes of Health Tool Box Cognition Battery (NIHTB-CB; Gershon et al. 2010; Gershon et al. 2013). A trained research assistant administered the NIHTB-CB on a 9.7-inch iPad Pro (Apple) in a quiet, distraction-free room. Administration times ranged between 30 and 45 min. The assessment was conducted in English. The NIHTB-CB was developed for ages 7–85 years, appears sensitive to cognitive impairment following traumatic brain injury (Holdnack et al. 2017), with evidence of adequate psychometric properties (Heaton et al. 2014; Scott et al. 2019). For example, the NIHTB-CB has shown acceptable internal consistency (Cronbach's alpha 0.8; test–retest reliability r = 0.8–0.9; convergent validity r = 0.8–0.90; and discriminant validity r = 0.2–0.4) against gold standard composites, in general and traumatic brain injury samples (Heaton et al. 2014; Tulsky et al. 2017). We have also shown that it is well tolerated by people early after mTBI (Macleod et al. 2021). The NIHTB-CB includes seven subtests measuring executive function, attention, episodic memory, language, processing speed and working memory (see Table 1). Individual subtest and composite scores were calculated. Raw scores were converted to age- and sex-corrected scaled scores; that is, normally distributed standard scores with a mean of 100 and standard deviation of 15.
| Subtest | Description and cognitive domain | Estimated administration time (min) | |
|---|---|---|---|
| (1) Picture sequence memory test | Evaluates episodic memory. Participants are asked to reproduce a sequence of pictures that is shown on the screen depicting a typical day in the park. | 7 | |
| (2) Flanker inhibitory control and attention test | Measures attention and inhibitory control. Participants focus on a given stimulus while inhibiting attention to stimuli flanking it. | 3 | |
| (3) List sorting working memory | Measures working memory. Participants recall and sequence different visually and orally presented stimuli. | 7 | |
| (4) Dimensional Change Card Sort Test | Measures cognitive flexibility and attention. Pictures are presented varying along two dimensions (e.g. shape and colour). The dimension for sorting is indicated by a cue word on the screen. | 3 | |
| (5) Pattern Comparison Processing Speed Test | Measures speed of processing. Participants discern whether two side-by-side pictures are the same or not and are required to respond as fast as possible. | 4 | |
| (6) Picture Vocabulary Test | Measures receptive vocabulary administered in a computer-adaptive test format. Participants select the picture that most closely matches the meaning of the word. | 3 | |
| (7) Oral Reading Recognition Test | Measures reading decoding skill and crystallised abilities. Participants are asked to read and pronounce letters and words as accurately as possible, and the test administrator scores using a pronunciation guide. | 3 | |
| Crystallised Composite Cognition Score | Subtests contributing to the crystallised composite cognition score are tests 6 and 7. | – | |
| Fluid Composite Cognition Score | Subtests contributing to the fluid composite cognition score are tests 1–5. | – |
Overestimation of cognitive impairment based on normally occurring variability is a noted concern in the wider neuropsychological literature (Brooks et al. 2008; Binder et al. 2009). To manage this with the NIHTB-CB, we determined the presence or absence of cognitive impairment using a premorbid ability-stratified multivariate base rate approach using the algorithms developed by Holdnack et al. (2017). Following these algorithms, cognitive impairment was defined as two or more low scores considering the five fluid subtests of the NIHTB-CB relative to the Crystallised Composite Cognition score. For individuals with acquired brain injury, the Crystallised Cognition Composite score may act as a proxy for estimated pre-injury general ability or baseline (Tulsky et al. 2017). The application of the algorithms and final determination of impairment was made by a neuropsychologist with experience undertaking cognitive assessment in mTBI populations (DS).
To check performance validity, we administered the Advanced Clinical Solutions for WAIS-IV Word Choice (Pearson 2009). This is a 50-item forced choice procedure that has the appearance of a memory test, but is designed to detect below capacity performance. In this study, we followed base rates published in the Advanced Clinical Solutions for WAIS-IV Word Choice manual and considered the threshold for valid performance to be a score of >45 out of 50. Participants were not excluded from analyses based on performance validity outcomes, because this would not preclude examination of psychological factors contributing to cognitive concerns.
Self-reported cognitive difficulties were measured either face-to-face or remotely, using the three cognitive items of the Rivermead Post-Concussion Symptom Questionnaire (RPQ). The RPQ is a 16-item self-report questionnaire that assesses common symptoms following mTBI (King et al. 1995). The RPQ consists of somatic symptoms (headaches, dizziness, nausea and vomiting, noise and light sensitivity, fatigue, sleep disturbance, double vision), cognitive symptoms (forgetfulness/poor memory, poor concentration taking longer to think) and emotional symptoms (being irritable/easily angered feeling depressed or tearful, feeling frustrated or impatient). Participants rated the presence and problem status of these symptoms on a scale of 0–4 (0 = not experienced at all; 1 = no more of a problem than before injury; 2 = a mild problem; 3 = a moderate problem; 4 = a severe problem). Scores of 1 were recoded to 0 following recommendations of King et al. (1995). Rasch analysis has shown that the RPQ demonstrated good internal consistency in a traumatic brain injury sample (person separation index = 0.87; Balalla et al. 2020). The RPQ was administered at Time 1 (M = 6.0 weeks post-injury, s.d. 2.5) and at Time 2 (M = 31 weeks after injury, s.d. 4.0).
For this study, we calculated the physical, cognition and emotion subscale scores following the factor analysis of Potter et al. (2006). The cognitive subscale of the RPQ is a commonly selected measure of subjective cognitive symptoms after mTBI, and scores range from 0 to 12 (Ngwenya et al. 2018). We then summed somatic and emotion subscale scores to create a post-concussion symptom datapoint that was not contaminated by inclusion of cognitive symptoms.
The Depression, Anxiety and Stress Scale-21 (DASS-21; Lovibond and Lovibond 1995) is a 21-item self-report scale measuring depression, anxiety and stress symptoms with a focus on the previous 7 days. Higher scores on this measure reflect elevated levels of depression, anxiety and stress symptoms. The DASS-21 has good psychometric properties, and is a valid measure of depression, anxiety and stress symptoms in people with brain injury (Ownsworth et al. 2008). For this study, total scale scores were calculated (range 0–63), with higher scores indicating higher distress (Zanon et al. 2021). The DASS-21 as a unidimensional scale has shown good internal consistency in general brain injury and mTBI samples (Zanon et al. 2021; Faulkner et al. 2024). The DASS-21 was administered at Time 1 and Time 2.
The Illness Perception Questionnaire-Revised (IPQ-R) provides a quantitative assessment of participant beliefs about their injury and recovery expectations (Moss-Morris et al. 2002). We have previously shown that such beliefs and expectations are associated with mTBI outcome (Snell et al. 2011, 2015). In this study, we used the timeline and consequences subscales of the IPQ-R; that is, perceptions of injury recovery and consequences. These two subscales have shown the strongest associations with outcomes in our previous work (Snell et al. 2013). Participants were asked to rate their agreement with statements about symptom beliefs on a 5-point Likert scale from ‘strongly disagree’ to ‘strongly agree’. As in our previous research, the IPQ-R was modified for mTBI by changing the word ‘illness’ to ‘injury’. The IPQ-R consequences and timeline subscales were administered at Time 1 and Time 2. Item responses from each scale were summed together to form an IPQ-R consequences and timeline total score, with scores ranging from 12 to 60, and higher scores reflecting stronger beliefs. We summed these scales to provide a single injury perception datapoint for the regression analyses.
The Pain Catastrophising Scale (PCS) is a 13-item scale evaluating the extent to which participants experienced various thoughts and feelings associated with their symptoms, scored on a 5-point Likert scale from ‘not at all’ to ‘all the time’ (Sullivan et al. 1995). Items included experiencing fears about non-recovery, feelings of loss of control of symptoms, feeling overwhelmed by symptoms and intrusiveness of symptoms. The PCS was originally developed for the chronic pain population. There is a body of evidence noting the similarity in patterns of catastrophising between people with chronic pain and those with persisting post-concussion symptoms following mTBI (Smith-Seemiller et al. 2003; Snell et al. 2018). Further recent work has highlighted that catastrophising may be associated with high cognitive symptom reporting after mTBI (Shi et al. 2024). In our study, we calculated a total score for the PCS, with scores ranging from 0 to 52, and adapted items, where the word ‘pain’ was replaced with ‘symptoms,’ following Terpstra et al. (2021). The PCS was administered at Time 1 and Time 2.
Statistical analyses
Data were analysed using SPSS version 28 (IBM Corp. Released 2020. IBM SPSS Statistics for MacIntosh). Findings are reported following Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines (Vandenbroucke et al. 2007). Demographic and clinical characteristics were summarised using descriptive statistics, such as means, standard deviation, ranges and frequencies. Differences between cognitive performance scores between Times 1 and 2 were examined using paired samples t-tests.
Bivariate correlations (Pearson’s r) were used to examine associations between self-reported cognitive symptoms (RPQ-Cognition subscale), objectively measured cognitive performance using the NIHTB-CB, the somatic and emotion RPQ subscales, and psychological measures at Times 1 and 2. Second, a series of unadjusted linear regression models were conducted to examine how much variance in self-reported cognitive symptoms at Time 2 (dependent variable) was explained by each of the Time 1 covariates. A multivariable model further examined associations between demographic and clinical variables including all covariates in the model together. For the linear regression models, the somatic and emotion subscales of the RPQ were summed to create a post-concussion symptoms datapoint. In a final step, a multivariable model using stepwise backward selection showed covariates remaining in the model.
Data are reported as 95% confidence intervals (CI). Effect sizes (ES) were calculated for significant results using Pearson’s r and Cohen’s interpretation rules (Cohen 1988), as appropriate. Small, medium and large effect sizes were respectively defined as |0.1–0.3|, |0.3–0.5| and |≥0.5| for Pearson’s r; 0.2, 0.5 and 0.8 for Cohen’s d; and 0.01, 0.09 and 0.25 for R2 (Ellis 2010). A two-tailed P < 0.05 was used to evaluate statistical significance. The approach to missing data was list-wise deletion reflecting the pragmatic nature of the study, embedded within clinical practice.
Results
Description of study sample
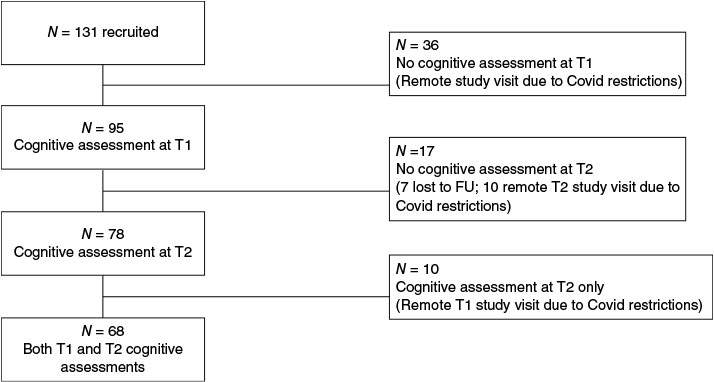
Of recruited participants to the main study (n = 131), n = 95 were seen face-to-face at Time 1 and had cognitive assessment, n = 78 were seen face-to-face at Time 2, and n = 68 had full sets of scores at both Times 1 and 2 (Fig. 1). The sample at Time 1 (n = 95) was on average aged 39.3 years (s.d. 16.1 years, range 16–76 years). There were more women (61.1%), and a majority reported their ethnicity as New Zealand European (90.5%). In terms of education, 43.2% described their highest education level as tertiary or university. The most common injury mechanisms were falls (31.6%) and transport-related accidents (30.5%). Over half of the sample reported a mental health history (60.6%), and 47.4% reported a prior history of mTBI. Much of the cohort had post-concussion symptoms that could be considered severe at Time 1 (Voormolen et al. 2018; Zeldovich et al. 2022), with an average symptom burden of 31.0 (s.d. 11.9) on the RPQ (total score). Levels of distress (DASS-21) fell in mild ranges overall at Time 1 (stress subscore: M = 8.1 (s.d. 4.3); anxiety subscore: M = 3.9 (s.d. 3.4); depression subscore: M = 5.0 (s.d. 4.2)). In terms of treatment accessed in concussion services, almost all participants reported having seen an occupational therapist (88.5%), and the majority participated in physiotherapy (78.6%) and a medical evaluation (57.3%). Few participants reported having seen a psychologist (9.2%) or neuropsychologist (9.9%). Demographic and clinical information is shown in Table 2.
| Variable | Mean (s.d.) or n (%) | |
|---|---|---|
| Demographic characteristics | ||
| Age [(years), M (s.d.), range] | 39.3 (16.1), 16.8–75.6 | |
| Sex [male, n (%)] | 37 (38.9) | |
| Ethnicity [n (%)] | ||
| New Zealand Māori | 9 (9.5) | |
| Non-New Zealand Māori | 86 (90.5) | |
| Highest education level [n (%)] | ||
| High school or less | 54 (56.8) | |
| University/tertiary | 41 (43.2) | |
| Previous traumatic brain injury [yes, n (%)] | 45 (47.4) | |
| Previous mental health history [yes, n (%)] | 57 (60.6) | |
| Injury and clinical characteristics | ||
| Time to baseline assessment in weeks [Time 1, M (s.d.)] | 6.4 (2.6) | |
| Mechanism of Injury [n (%)] | ||
| Transport-related | 29 (30.5) | |
| Fall | 30 (31.6) | |
| Assault | 9 (9.5) | |
| Hit by object | 7 (7.4) | |
| Other | 20 (21.1) | |
| PTA [n (%)] | ||
| No PTA | 34 (35.8) | |
| PTA <1 h | 45 (47.4) | |
| PTA >1 h, <24 h | 16 (16.8) | |
| Glasgow Coma Scale Score [M (s.d.)] | 14.9 (0.3) | |
| Symptoms at baseline [T1 RPQ total, M (s.d.)] | 31.7 (11.7) | |
| DASS21 total score at time 1 [M (s.d.)] | 17.4 (10.5) | |
| IPQ-R Consequences + Timeline score at Time 1 [M (s.d.)] | 35.4 (5.3) | |
| Pain catastrophising scale score at time 1 [M (s.d.)] | 21.2 (7.7) | |
T1 = Time 1; RPQ = Rivermead Post-Concussion Symptom Questionnaire; PTA = post-traumatic amnesia; DASS21 = Depression Anxiety and Stress Scale 21; NIH = National Institutes of Health; IPQ-R = Illness Perception Questionnaire-Revised sum of consequences and timeline scales; PCS = Pain Catastrophising Scale.
Using the multivariable base rate approach to defining cognitive assessment outcomes using the NIHTB-CB, over half the sample (n = 51; 54%) met criteria for cognitive impairment at Time 1. At Time 2, 17% (n = 13) of those meeting criteria for cognitive impairment at Time 1 still met these criteria. A small number of participants met the criterion for performance invalidity at Time 1 (n = 11; 11.6%), with just three (3.8%) participants still meeting this criterion at Time 2. Participants were not excluded from analyses based on performance validity. Effect size differences comparing performances for NIHTB-CB subtests and Fluid and Crystallised composite scores at Times 1 and 2 were large for subtests contributing to the fluid composite and small for crystallised subtests. Cognitive performance data are shown in Table 3.
| Variable | Time 1 | Time 2 | Mean difference (95% CI) | Effect size A | |
|---|---|---|---|---|---|
| Time 1 | |||||
| Crystallised composite score [M (s.d.), range] | 106.42 (14.53), 72–144 | 104.31 (15.64), 75–138 | 2.19 (−4.64, 0.26) | 0.22 | |
| Fluid composite score [M (s.d.), range] | 91.35 (17.72), 54–130 | 104.67 (18.16), 63–144 | 12.70 (9.21, 16.19) | 0.89 | |
| Picture sequence memory test [M (s.d.), range] | 100.96 (15.96), 71–146 | 110.03 (17.55), 72–146 | 9.02 (5.32, 12.71) | 0.59 | |
| Flanker inhibitory control and attention test [M (s.d.), range] | 81.67 (13.75), 54–114 | 89.51 (13.88), 56–126 | 7.91 (4.79, 11.03) | 0.63 | |
| List sorting working memory [M (s.d.), range] | 99.11 (13.65), 64–136 | 104.04 (12.65), 72–128 | 4.65 (1.59, 7.70) | 0.38 | |
| Dimensional change card sort test [M (s.d.), range] | 90.40 (17.87), 54–136 | 101.00 (19.79), 59–146 | 9.60 (4.99, 14.21) | 0.50 | |
| Pattern comparison processing speed test [M (s.d.), range] | 98.26 (22.54), 54–140 | 110.27 (20.36), 63–146 | 11.40 (6.84, 15.97) | 0.61 | |
| Picture vocabulary test [M (s.d.), range] | 102.94 (12.64), 64–136 | 102.76 (12.88), 79–127 | 0.38 (−2.21, 2.97) | 0.04 | |
| Oral reading recognition test [M (s.d.), range] | 109.19 (17.43), 65–145 | 105.36 (17.90), 65–146 | −4.60 (−7.02, −1.59) | 0.37 | |
| Met criteria for cognitive impairment [yes, n (%)] B | 51 (53.7) | 13 (16.7) | |||
| Met criteria for invalid performance [yes, n (%)] C | 11 (11.6) | 3 (3.8) | |||
Associations between self-reported cognitive symptoms, objective cognitive performance and psychological variables
Table 4 shows correlations between self-reported cognitive symptoms (RPQ cognitive score), cognitive performance as measured using the fluid composite score of the NIHTB-CB and psychological symptoms. At Time 1, self-reported cognitive symptoms were negatively correlated with cognitive performances, although the effect size was small. There were medium to large positive correlations between self-reported cognitive symptoms, emotional and somatic post-concussion symptoms (RPQ) and injury recovery beliefs (IPQ-R).
| Variable | RPQ-cognitive score | Fluid composite score | RPQ-physical | RPQ-emotional | IPQ-R | DASS21 total score | PCS total score | |
|---|---|---|---|---|---|---|---|---|
| Time 1 (n = 95) | ||||||||
| RPQ-cognitive score | 1 | −0.21* | 0.65** | 0.51** | 0.37** | 0.17 | 0.09 | |
| Fluid composite cognition score | 1 | −0.34** | −0.36** | −0.28** | −0.18 | −0.07 | ||
| RPQ-physical score | 1 | 0.51** | 0.52** | 0.38** | 0.40** | |||
| RPQ-emotional score | 1 | 0.40** | 0.45** | 0.31** | ||||
| IPQ-R consequences + timeline | 1 | 0.46** | 0.53** | |||||
| DASS21 total score | 1 | 0.58** | ||||||
| PCS total score | 1 | |||||||
| Time 2 (n = 78) | ||||||||
| RPQ-cognitive score | 1 | −0.16 | 0.68** | 0.55** | 0.37** | 0.24* | 0.23* | |
| Fluid composite cognition score | −0.07 | −0.10 | −0.30** | −0.23* | −0.17 | |||
| RPQ-physical score | 1 | 0.56** | 0.53** | 0.28** | 0.26** | |||
| RPQ-emotional score | 1 | 0.47** | 0.54** | 0.45** | ||||
| IPQ-R consequences + timeline | 1 | 0.38** | 0.50** | |||||
| DASS21 total score | 1 | 0.56** | ||||||
| PCS total score | 1 | |||||||
*P < 0.05; **P < 0.01; 1. Fluid Composite Cognition Score from the National Institutes of Health Toolbox Cognition Battery; mTBI = mild traumatic brain injury; RPQ = Rivermead Post-Concussion Symptom Questionnaire; IPQ-R = Illness Perceptions Questionnaire-Revised Consequences and Timeline scales combined; DASS21 = 21-item Depression, Stress and Anxiety Scale.
At Time 2, self-reported cognitive symptoms were again negatively correlated with cognitive performances, but these relationships were small and not significant. In contrast, there were medium to large correlations between self-reported cognitive symptoms, post-concussion symptoms (RPQ) and injury recovery beliefs (IPQ-R). Associations between self-reported cognitive symptoms and other psychological variables (DASS21 and PCS) were generally small.
Time 1 predictors of persisting self-reported cognitive symptoms at Time 2
In the unadjusted linear regression models, post-concussion symptoms and psychological variables each contributed to variance in Time 2 RPQ-Cognitive scores (Table 5). Table 5 also shows that after controlling for variables in the adjusted model, no variable remained significant. In the adjusted model, 25.0% of variance was explained (R2 = 0.37; adjusted R2 = 0.25). Using a stepwise backward selection process, two variables remained, with an adjusted R2 = 0.24 (Table 6). These were Time 1 post-concussion symptom burden (RPQ physical and emotional symptoms) and Time 1 injury recovery beliefs (IPQ-R).
| Model | Unadjusted | Adjusted A | |||||
|---|---|---|---|---|---|---|---|
| B | 95% CI | P | B | 95% CI | P | ||
| Demographic variables | |||||||
| Sex (male) | −0.32 | −1.63, 0.99 | 0.63 | −0.22 | −1.82, 1.39 | 0.79 | |
| Age at injury | 0.00 | −0.04, 0.04 | 0.98 | −0.01 | −0.06, 0.04 | 0.69 | |
| Pre-injury ability (NIHTB-CB crystallised composite score) T1 | −0.02 | −0.07, 0.03 | 0.46 | 0.02 | −0.03, 0.07 | 0.44 | |
| Clinical variables | |||||||
| Previous depression (yes) | 0.43 | −0.86, 1.72 | 0.51 | −1.29 | −2.84, 0.26 | 0.10 | |
| Previous concussion (yes) | −0.25 | −1.52, 1.02 | 0.70 | 0.25 | −1.18, 1.68 | 0.73 | |
| Injury severity (any PTA yes) | −0.23 | −1.51, 1.04 | 0.72 | −1.40 | −3.27, 0.47 | 0.14 | |
| LOC (yes) | −0.09 | −1.37, 1.19 | 0.89 | 0.53 | −1.39, 2.45 | 0.57 | |
| Objective cognitive performance | |||||||
| NIHTB-CB fluid composite score T1 | −0.04 | −0.08, 0.01 | 0.10 | 0.00 | −0.06, 0.05 | 0.91 | |
| RPQ | |||||||
| Physical + emotional symptoms T1 | 0.16 | 0.09, 0.22 | <0.01 | 0.09 | −0.04, 0.21 | 0.18 | |
| Injury beliefs/expectations | |||||||
| IPQ-R (total consequences and timeline beliefs) T1 | 0.24 | 0.12, 0.34 | <0.01 | 0.13 | −0.06, 0.31 | 0.17 | |
| Distress | |||||||
| DASS21 total score T1 | 0.12 | 0.04, 0.17 | <0.01 | 0.03 | −0.05, 0.12 | 0.46 | |
| PCS total score T1 | 0.17 | 0.09, 0.26 | <0.01 | 0.08 | −0.06. 0.22 | 0.24 | |
| Variable | B | 95% CI | P | |
|---|---|---|---|---|
| Post-concussion symptoms at T1: | ||||
| RPQ total physical and emotion symptom score T1 | 0.13 | 0.03, 0.24 | 0.02 | |
| Injury beliefs/expectations at T1: | ||||
| IPQ-R (total consequences and timeline beliefs) T1 | 0.16 | −0.01, 0.34 | 0.05 |
R2 = 0.35; adjusted R2 = 0.24.
Dependent variable = self-reported cognitive difficulties (RPQ-Cognitive score) at Time 2 (T2).
CI = confidence interval; T1 = time 1; RPQ = Rivermead Post-Concussion Symptom Questionnaire; IPQ-R = Illness Perceptions Questionnaire-Revised Consequences and Timeline combined subscale scores.
Discussion
In this study, we explored associations between self-reported cognitive symptoms and objectively measured cognitive performance within 3 months of an mTBI and 6 months later. We also investigated demographic and clinical predictors of persisting subjectively experienced cognitive symptoms. Although there were slightly different patterns of associations depending on time post-injury, at both assessment points, subjectively experienced cognitive symptoms were more strongly associated with post-concussion symptoms and psychological variables than with cognitive performance. Early predictors of persisting self-reported cognitive symptoms included post-concussion symptom burden and injury recovery beliefs.
Rates of cognitive impairment early and late after mTBI in our study are consistent with a large body of research identifying that measurable cognitive difficulties can be discerned early after mTBI, generally resolving for a majority within early the weeks of injury (Karr et al. 2014; Boone et al. 2024). Our findings of modest negative correlations between subjectively experienced cognitive symptoms and objectively measured cognitive performance early after mTBI are also consistent with other work. In a cross-sectional study investigating cognitive complaints within 3 months of injury in a highly symptomatic civilian sample (n = 100), subjective cognitive measures modestly negatively correlated with actual cognitive/neurological performance, mostly only for complex attentional tasks (Stillman et al. 2020).
Later after injury, at Time 2, correlations between subjectively experienced cognitive symptoms and objectively measured cognitive performances were weaker than those between subjective cognitive symptoms, post-concussion symptoms and psychological factors. This adds to other published research noting that factors contributing to persisting cognitive symptoms after mTBI are complex and multifactorial, with psychological symptoms being important drivers of persistent self-reported cognitive outcomes (French et al. 2014; Barker-Collo et al. 2015; Ngwenya et al. 2018; Levy et al. 2024). Among these, the impacts of depression and anxiety on subjectively experienced cognitive symptoms after mTBI have been most studied. We have previously shown that recovery expectations and beliefs appear important in terms of general symptom outcomes (Snell et al. 2013, 2015). In the present study, injury recovery beliefs also appeared to be associated more specifically with persisting self-reported cognitive symptoms. Factors that might contribute to experiences of cognitive symptoms after mTBI include the representations of the injury, including expectations of cognitive recovery and the nocebo effect. The nocebo effect refers to symptoms that develop in response to negative health-related information, beliefs and/or experiences that can influence outcomes through their contextualised, psychosocial significance (Polich et al. 2020).
There has been a recent upsurge in literature describing functional cognitive complaints; that is, where self-reported cognitive difficulties are not fully attributable to brain injury or disease, or that are in excess of objectively measured or observed performance (Silverberg and Rush 2023). Mechanisms underlying such discrepancies are unclear, but could include increased concern about brain health following a neurological event, such as an mTBI, which could contribute to expectations of persistent poor cognitive functioning (Bhome et al. 2022). Bhome et al. (2022) suggest expectations of cognitive impairment reinforced by attribution bias might override actual experience of good or improving cognitive performance by not experiencing opportunities to update beliefs about cognition.
Clinical implications
Our findings add to work noting the impact of psychological symptoms on mTBI outcomes and the importance of early identification of at-risk individuals who may need prompt referral for treatment (Silverberg et al. 2020). Although generic education and reassurance interventions early after mTBI have an evidence base (Snell et al. 2009; Silverberg et al. 2020), there may be a specific opportunity to focus on factors thought to perpetuate cognitive symptoms (Silverberg and Rush 2023). For example, better understanding of the expectations people hold about their cognitive recovery after mTBI and encouraging more normal (pre-injury) use of functions, such as memory, may provide an opportunity to challenge negative expectations, experience cognitive successes and update beliefs about cognitive function (Griem et al. 2016; Mollica et al. 2022).
Limitations
First, we acknowledge the impact of the COVID-19 pandemic on missing datapoints in our sample and losses to follow up, with a small number of participants assessed only at either Time 1 or Time 2, but not both. Overestimating poor outcomes is a noted concern associated with attrition bias in mTBI studies (Cancelliere et al. 2023), although in this study, datapoints for cognitive assessment were missing at random and largely linked to the effects of the pandemic. In addition, it is possible that impacts of the pandemic contributed to higher distress levels among participants recruited during 2020–2022; however, New Zealand’s COVID-19 cases were much lower than comparable countries in the first 2 years of the pandemic, New Zealanders spent less time in lockdown than many other countries and the economy recovered faster (Anon 2024; French et al. 2025). Second, we did not have additional severity information, such as imaging, to explore whether cognitive outcomes were influenced by injury severity, beyond estimated post- traumatic amnesia and the Glasgow Coma Scale score. Third, it is worth noting the issue of ecological validity of neuropsychological assessment and the possibility that available cognitive tests and batteries, including the NIHTB-CB, may not be sensitive to day-to-day cognitive symptoms experienced by people after mTBI (Bigler et al. 2013). Furthermore, practice effects on the NIHTB-CB could have contributed to improved performances at 6–9 months post-injury. Although the test–retest reliability of the NIHTB-CB is generally considered to be excellent (Heaton et al. 2014), there are some accounts of practice effects evident at least at 3-month retest (Kuzmuk et al. 2023). The NIHTB-CB practice effects have been described as similar to those obtained for the widely used gold standard neuropsychological measures, and practice effects would not undermine examination of associations between cognitive performance and other variables (Heaton et al. 2014). In addition, we did not exclude participants if they failed performance validity tests. This may have been a confounding factor, especially at Time 1. Fourth, we evaluated self-reported cognitive symptoms using the three cognitive symptom items of the RPQ. More robust measures of subjectively experienced cognitive symptoms (such as the Cognitive Failures Questionnaire (Broadbent et al. 1982) or Cognitive Complaint After Mild Closed Head Injury scale (Anderson 2021)) are recommended to better understand and measure cognitive recovery after mTBI (Al Sayegh et al. 2010). Finally, the study recruitment procedure involved recruitment on presentation to an outpatient healthcare service following injury and consent to being contacted about the study. This means our findings may not generalise to the wider mTBI population, and those who declined to be contacted and those sustaining mTBI, but not being referred or presenting to our recruitment sites or to health services at all, were missed (McCullagh and Feinstein 2003). Furthermore, those referred for outpatient rehabilitation are likely to be referred because they demonstrate risk factors for slow recovery, such as a high post-concussion and psychological symptom burden, prior history of mTBI, and mental health complications (Silverberg et al. 2015). Thus, although our sample reflects a subpopulation of people sustaining mTBI, it appears similar to the population of people referred to mTBI outpatient clinics (Forrest et al. 2018).
Conclusions
Factors contributing to subjectively experienced cognitive symptoms after mTBI appear complex. High early post-concussion symptom burden and psychological mechanisms, such as pessimistic recovery expectations, may increase risk for persistent subjectively experienced cognitive symptoms. Our findings may be helpful for clinicians by guiding targeted treatment efforts towards addressing factors with the potential to influence cognitive symptom reporting after mTBI.
Declaration of funding
This research was supported by a grant from the Health Research Council of NZ (ref 18/046). Professor Alice Theadom is supported by a Rutherford Discovery Fellowship, administered by The Royal Society – Te Aparangi.
Acknowledgements
We acknowledge the work of our two research assistants (Corrin Bourke and Maureen O’Reilly) for keeping recruitment going under the challenging circumstances of the COVID-19 pandemic.
References
Al Sayegh A, Sandford D, Carson AJ (2010) Psychological approaches to treatment of postconcussion syndrome: a systematic review. Journal of Neurology, Neurosurgery and Psychiatry 81, 1128-1134.
| Crossref | Google Scholar | PubMed |
Anderson J (2021) Cognitive complaint and objective cognition during the post-acute period after mild traumatic brain injury in pre-morbidly healthy adults. Brain Injury 35, 103-113.
| Crossref | Google Scholar | PubMed |
Balalla S, Krägeloh C, Medvedev O, Siegert R (2020) Is the Rivermead Post-Concussion Symptoms Questionnaire a Reliable and Valid Measure to Assess Long-Term Symptoms in Traumatic Brain Injury and Orthopedic Injury Patients? A Novel Investigation Using Rasch Analysis. Neurotrauma Reports 1, 63-72.
| Crossref | Google Scholar | PubMed |
Barker-Collo S, Jones K, Theadom A, Starkey N, Dowell A, Mcpherson K, Ameratunga S, Dudley M, Te Ao B, Feigin V, on behalf of the BIONIC Research Group (2015) Neuropsychological outcome and its correlates in the first year after adult mild traumatic brain injury: A population-based New Zealand study. Brain Injury 29, 1604-16.
| Crossref | Google Scholar | PubMed |
Belanger HG, SpiegeL E, Vanderploeg RD (2010) Neuropsychological performance following a history of multiple self-reported concussions: A meta-analysis. Journal of the International Neuropsychological Society 16, 262-267.
| Crossref | Google Scholar | PubMed |
Bhome R, Mcwilliams A, Price G, Poole NA, Howard RJ, Fleming SM, Huntley JD (2022) Metacognition in functional cognitive disorder. Brain Communications 4, fcac041.
| Crossref | Google Scholar | PubMed |
Biagianti B, Stocchetti N, Brambilla P, Van Vleet T (2020) Brain dysfunction underlying prolonged post-concussive syndrome: a systematic review. Journal of Affective Disorders 262, 71-76.
| Crossref | Google Scholar | PubMed |
Bigler ED, Farrer TJ, Pertab JL, James K, Petrie JA, Hedges DW (2013) Reaffirmed limitations of meta-analytic methods in the study of mild traumatic brain injury: A response to Rohling et al. The Clinical Neuropsychologist 27, 176-214.
| Crossref | Google Scholar | PubMed |
Binder LM, Rohling ML, Larrabee GJ (1997) A review of mild head trauma. Part I: Meta-analytic review of neuropsychological studies. Journal of Clinical and Experimental Neuropsychology 19, 421-431.
| Crossref | Google Scholar | PubMed |
Binder LM, Iverson GL, Brooks BL (2009) To err is human: “abnormal” neuropsychological scores and variability are common in healthy adults. Archives of Clinical Neuropsychology 24, 31-46.
| Crossref | Google Scholar | PubMed |
Boone K, Vane R, Victor T (2024) Critical Review of Recently Published Studies Claiming Long Term Neurocognitive Abnormalities in Mild Traumatic Brain Injury. Archives of Clinical Neuropsychology 40, 272-288.
| Crossref | Google Scholar | PubMed |
Brands IM, Verlinden I, Ribbers GM (2019) A study of the influence of cognitive complaints, cognitive performance and symptoms of anxiety and depression on self-efficacy in patients with acquired brain injury. Clinical Rehabilitation 33, 327-334.
| Crossref | Google Scholar | PubMed |
Broadbent DE, Cooper PF, Fitzgerald P, Parkes KR (1982) The Cognitive Failures Questionnaire (CFQ) and its correlates. British Journal of Clinical Psychology 21, 1-16.
| Crossref | Google Scholar | PubMed |
Brooks BL, Iverson GL, Holdnack JA, Feldman HH (2008) Potential for misclassification of mild cognitive impairment: A study of memory scores on the Wechsler Memory Scale-III in healthy older adults. Journal of the International Neuropsychological Society 14, 463-478.
| Crossref | Google Scholar | PubMed |
Cancelliere C, Verville L, Stubbs JL, Yu H, Hincapié CA, Cassidy JD, Wong JJ, Shearer HM, Connell G, Southerst D, Howitt S, Guist B, Silverberg ND (2023) Post-Concussion Symptoms and Disability in Adults With Mild Traumatic Brain Injury: A Systematic Review and Meta-Analysis. Journal of Neurotrauma 40, 1045-1059.
| Crossref | Google Scholar | PubMed |
Dikmen S, Machamer J, Temkin N (2017) Mild traumatic brain injury: longitudinal study of cognition, functional status, and post-traumatic symptoms. Journal of Neurotrauma 34, 1524-1530.
| Crossref | Google Scholar | PubMed |
Faulkner JW, Snell DL (2023) A framework for understanding the contribution of psychosocial factors in biopsychosocial explanatory models of persistent postconcussion symptoms. Physical Therapy 103, pzac156.
| Crossref | Google Scholar | PubMed |
Faulkner J, Snell D, Siegert R (2024) Rasch analysis of the depression anxiety stress scales-21 (DASS-21) in a mild traumatic brain injury sample. Brain Injury 39, 136-144 Epub ahead of print.
| Crossref | Google Scholar | PubMed |
Forrest R, Henry JD, Mcgarry PJ, Marshall RN (2018) Mild traumatic brain injury in New Zealand: factors influencing post-concussion symptom recovery time in a specialised concussion service. Journal of Primary Health Care 10, 159-166.
| Crossref | Google Scholar | PubMed |
French LM, Lange RT, Brickell T (2014) Subjective cognitive complaints and neuropsychological test performance following military-related traumatic brain injury. Journal of Rehabilitation Research and Development 51, 933-950.
| Crossref | Google Scholar | PubMed |
French NP, Maxwell H, Baker MG, Callaghan F, Dyet K, Geoghegan JL, Hayman D, Huang QS, Kvalsvig A, Russell E, Scott P, Thompson TP, Plank MJ (2025) Preparing for the next pandemic: insights from Aotearoa New Zealand’s Covid-19 response. The Lancet Regional Health - Western Pacific 56, 101525.
| Crossref | Google Scholar | PubMed |
Gershon RC, Cella D, Fox NA, Havlik RJ, Hendrie HC, Wagster MV (2010) Assessment of neurological and behavioural function: The NIH Toolbox. The Lancet Neurology 9, 138-139.
| Crossref | Google Scholar | PubMed |
Gershon RC, Wagster MV, Hendrie HC, Fox NA, Cook KF, Nowinski CJ (2013) NIH toolbox for assessment of neurological and behavioral function. Neurology 80, S2-S6.
| Crossref | Google Scholar | PubMed |
Griem J, Stone J, Carson A, Kopelman MD (2016) Psychologic/functional forms of memory disorder. Handbook of Clinical Neurology 139, 407-417.
| Crossref | Google Scholar | PubMed |
Heaton RK, Akshoomoff N, Tulsky D, Mungas D, Weintraub S, Dikmen S, Beaumont J, Casaletto KB, Conway K, Slotkin J, Gershon R (2014) Reliability and validity of composite scores from the NIH Toolbox Cognition Battery in adults. Journal of the International Neuropsychological Society 20, 588-598.
| Crossref | Google Scholar | PubMed |
Holdnack JA, Iverson GL, Silverberg ND, Tulsky DS, Heinemann AW (2017) NIH toolbox cognition tests following traumatic brain injury: Frequency of low scores. Rehabilitation Psychology 62, 478-484.
| Crossref | Google Scholar | PubMed |
Holm L, Cassidy JD, Carroll LJ, Borg J (2005) Summary of the WHO Collaborating Centre for Neurotrauma Task Force on Mild Traumatic Brain Injury. Journal of Rehabilitation Medicine 37, 137-141.
| Crossref | Google Scholar | PubMed |
Iverson GL (2019) Network analysis and precision rehabilitation for the post-concussion syndrome. Frontiers in Neurology 10, 489.
| Crossref | Google Scholar | PubMed |
Iverson GL, Karr JE, Gardner AJ, Silverberg ND, Terry DP (2019) Results of scoping review do not support mild traumatic brain injury being associated with a high incidence of chronic cognitive impairment: Commentary on McInnes et al. 2017. PLoS One 14, e0218997.
| Crossref | Google Scholar | PubMed |
Karr JE, Areshenkoff CN, Garcia-Barrera MA (2014) The neuropsychological outcomes of concussion: a systematic review of meta-analyses on the cognitive sequelae of mild traumatic brain injury. Neuropsychology 28, 321-336.
| Crossref | Google Scholar | PubMed |
King NS, Crawford S, Wenden FJ, Moss NE, Wade DT (1995) The Rivermead Post Concussion Symptoms Questionnaire: a measure of symptoms commonly experienced after head injury and its reliability. Journal of Neurology 242, 587-592.
| Crossref | Google Scholar | PubMed |
Kuzmuk LE, Rebchuk AD, Deptuck HM, Cairncross M, Silverberg ND, Field TS (2023) Three-month Practice Effect of the National Institutes of Health Toolbox Cognition Battery in Young Healthy Adults. The Canadian Journal of Neurological Sciences 50, 769-772.
| Crossref | Google Scholar | PubMed |
Levy AM, Saling MM, Anderson J (2023) Frequency and extent of cognitive complaint following adult civilian mild traumatic brain injury: A systematic review and meta-analysis. Brain Impairment 24, 309-332.
| Crossref | Google Scholar | PubMed |
Levy A, Saling M, Anderson J (2024) Cognitive Symptoms Are Not Associated with Cognitive Performance in Post-Acute mTBI. Archives of Clinical Neuropsychology 40, 63-74.
| Crossref | Google Scholar |
Lovibond PF, Lovibond SH (1995) The structure of negative emotional states: comparison of the Depression Anxiety Stress Scales (DASS) with the Beck Depression and Anxiety Inventories. Behaviour Research and Therapy 33, 335-343.
| Crossref | Google Scholar | PubMed |
Machamer J, Temkin N, Dikmen S, Nelson LD, Barber J, Hwang P, Boase K, Stein MB, Sun X, Giacino J, Mccrea MA, Taylor SR, Jain S, Manley G, TRACK-TBI Investigators (2022) Symptom Frequency and Persistence in the First Year after Traumatic Brain Injury: A TRACK-TBI Study. Journal of Neurotrauma 39, 358-370.
| Crossref | Google Scholar | PubMed |
Macleod C, Surgenor L, Levack W, Hackney J, Theadom A, Siegert R, silverberg N, Snell D (2021) Patient and clinician experiences of a computerised cognitive battery for use after concussion: a preliminary qualitative study. Brain Impairment 22, 189-204.
| Google Scholar |
McCrea M, Iverson GL, Mcallister TW, Hammeke TA, Powell MR, Barr WB, Kelly JP (2009) An integrated review of recovery after mild traumatic brain injury (MTBI): implications for clinical management. The Clinical Neuropsychologist 23, 1368-1390.
| Crossref | Google Scholar | PubMed |
McCullagh S, Feinstein A (2003) Outcome after mild traumatic brain injury: An examination of recruitment bias. Journal of Neurology, Neurosurgery & Psychiatry 74, 39-43.
| Crossref | Google Scholar | PubMed |
Mollica A, Dey A, Cairncross M, Silverberg N, Burke MJ (2022) Neuropsychiatric treat-ment for mild traumatic brain injury: Nonpharmacological approaches. Seminars in Neurology 42, 168-181.
| Crossref | Google Scholar | PubMed |
Moss-Morris R, Weinman J, Petrie K, Horne R, Cameron L, Buick D (2002) The Revised Illness Perception Questionnaire (IPQ-R). Psychology & Health 17, 1-16.
| Crossref | Google Scholar |
Ngwenya LB, Gardner RC, Yue JK, Burke JF, Ferguson AR, Huang MC, Winkler EA, Pirracchio R, Satris GG, Yuh EL, Mukherjee P, Valadka AB, Okonkwo DO, Manley GT (2018) Concordance of common data elements for assessment of subjective cognitive complaints after mild-traumatic brain injury: a TRACK-TBI Pilot Study. Brain Injury 32, 1071-1078.
| Crossref | Google Scholar | PubMed |
Ownsworth T, Little T, Turner B, Hawkes A, Shum D (2008) Assessing emotional status following acquired brain injury: The clinical potential of the depression, anxiety and stress scales. Brain Injury 22, 858-869.
| Crossref | Google Scholar | PubMed |
Pertab JL, James KM, Bigler ED (2009) Limitations of mild traumatic brain injury meta-analyses. Brain Injury 23, 498-508.
| Crossref | Google Scholar | PubMed |
Pieroth E (2022) Assessment and management of persistent post-concussion symptoms. Operative Techniques in Sports Medicine 30, 150894.
| Crossref | Google Scholar |
Polich G, Iaccarino MA, Kaptchuk TJ, Morales-Quezada L, Zafonte R (2020) Nocebo effects in concussion. Is all that is told beneficial? American Journal of Physical Medicine and Rehabilitation 99, 71-80.
| Crossref | Google Scholar | PubMed |
Potter S, Leigh E, Wade D, Fleminger S (2006) The Rivermead Post Concussion Symptoms Questionnaire: A confirmatory factor analysis. Journal of Neurology 253, 1603-14.
| Crossref | Google Scholar | PubMed |
Schneider A, Huie JR, Boscardin WJ, Nelson L, Barber JK, Yaffe K, Diaz-Arrastia R, Ferguson AR, Kramer J, Jain S, Temkin N, Yuh E, Manley GT, Gardner RC, TRACK-TBI Investigators (2022) Cognitive Outcome 1 Year After Mild Traumatic Brain Injury: Results From the TRACK-TBI Study. Neurology 98, e1248-e1261.
| Crossref | Google Scholar | PubMed |
Schretlen DJ, Shapiro AM (2003) A quantitative review of the effects of traumatic brain injury on cognitive functioning. International Review of Psychiatry 15, 341-349.
| Crossref | Google Scholar | PubMed |
Scott EP, Sorrell A, Benitez A (2019) Psychometric properties of the NIH toolbox cognition battery in healthy older adults: reliability, validity, and agreement with standard neuropsychological tests. Journal of the International Neuropsychological Society 25, 857-867.
| Crossref | Google Scholar | PubMed |
Shi S, Picon EL, Rioux M, Panenka WJ, Silverberg ND (2024) Catastrophizing is associated with excess cognitive symptom reporting after mild traumatic brain injury. Neuropsychology 38, 126-133.
| Crossref | Google Scholar | PubMed |
Silverberg ND, Mikolić A (2023) Management of Psychological Complications Following Mild Traumatic Brain Injury. Current Neurology and Neuroscience Reports 23, 49-58.
| Crossref | Google Scholar | PubMed |
Silverberg N, Rush B (2023) Neuropsychological evaluation of functional cognitive disorder: a narrative review. The Clinical Neuropsychologist 38, 302-325.
| Crossref | Google Scholar |
Silverberg ND, Gardner AJ, Brubacher JR, Panenka WJ, Li JJ, Iverson GL (2015) Systematic Review of Multivariable Prognostic Models for Mild Traumatic Brain Injury. Journal of Neurotrauma 32, 517-526.
| Crossref | Google Scholar | PubMed |
Silverberg ND, Iaccarino MA, Panenka WJ, Iverson GL, McCulloch KL, Dams-O’connor K, Reed N, American Congress of Rehabilitation Medicine Brain Injury Interdisciplinary Special Interest Group Mild TBI Task Force (2020) Management of Concussion and Mild Traumatic Brain Injury: A Synthesis of Practice Guidelines. Archives of Physical Medicine and Rehabilitation 101, 382-393.
| Crossref | Google Scholar | PubMed |
Smith-Seemiller L, Fow NR, Kant R, Franzen MD (2003) Presence of post-concussion syndrome symptoms in patients with chronic pain vs mild traumatic brain injury. Brain Injury 17, 199-206.
| Crossref | Google Scholar | PubMed |
Snell DL, Surgenor LJ, Hay-Smith EJ, Siegert RJ (2009) A systematic review of psychological treatments for mild traumatic brain injury: an update on the evidence. Journal of Clinical & Experimental Neuropsychology 31, 20-38.
| Crossref | Google Scholar | PubMed |
Snell DL, Siegert RJ, Hay-Smith EJ, Surgenor LJ (2011) Associations between illness perceptions, coping styles and outcome after mild traumatic brain injury: preliminary results from a cohort study. Brain Injury 25, 1126-1138.
| Crossref | Google Scholar | PubMed |
Snell D, Hay-Smith E, Surgenor L, Siegert R (2013) Predicting long-term outcome after mild traumatic brain injury: The contribution of injury beliefs and Leventhal’s Common Sense Model. Brain Injury 23, 333-362.
| Google Scholar |
Snell DL, Surgenor LJ, Hay-Smith EJ, Williman J, Siegert RJ (2015) The contribution of psychological factors to recovery after mild traumatic brain injury: Is cluster analysis a useful approach? Brain Injury 29, 291-299.
| Crossref | Google Scholar | PubMed |
Snell DL, Martin R, Surgenor LJ, Siegert RJ, Hay-Smith E (2017) What’s wrong with me? seeking a coherent understanding of recovery after mild traumatic brain injury. Disability and Rehabilitation 39, 1968-1975.
| Crossref | Google Scholar | PubMed |
Snell D, Martin R, Macleod A, Surgenor L, Siegert R, Hay-Smith E, Melzer T, Hooper G, Anderson T (2018) Untangling chronic pain and post concussion symptoms: the significance of depression. Brain Injury 32, 583-592 [Epub ahead of print].
| Crossref | Google Scholar |
Stenberg J, Karr JE, Terry DP, Håberg AK, Vik A, Skandsen T, Iverson GL (2020) Change in self-reported cognitive symptoms after mild traumatic brain injury is associated with changes in emotional and somatic symptoms and not changes in cognitive performance. Neuropsychology 34, 560-568.
| Crossref | Google Scholar | PubMed |
Stillman AM, Madigan N, Torres K, Swan N, Alexander MP (2020) Subjective Cognitive Complaints in Concussion. Journal of Neurotrauma 37, 305-311.
| Crossref | Google Scholar | PubMed |
Sullivan M, Bishop S, Pivik J (1995) The pain catastrophizing scale: development and validation. Psychological Assessment 7, 524-532.
| Google Scholar |
Terpstra AR, Cairncross M, Yeates KO, Vranceanu AM, Greenberg J, Hunt C, Silverberg ND (2021) Psychological Mediators of Avoidance and Endurance Behavior After Concussion. Rehabilitation Psychology 66, 470-478.
| Crossref | Google Scholar | PubMed |
Theadom A, Parag V, Dowell T, Mcpherson K, Starkey N, Barker-Collo S, Jones K, Ameratunga S, Feigin VL, BIONIC Research Group (2016) Persistent problems one year after mild traumatic brain injury: A longitudinal population study in New Zealand. British Journal of General Practice 66, e16-23.
| Crossref | Google Scholar | PubMed |
Theadom A, Barker-Collo S, Jones K, Kahan M, Te Ao K, Mcpherson N, Starkey V, Feigin F, BIONIC4you Research Group (2017) Work limitations 4 years after mild traumatic brain injury: a cohort study. Archives of Physical Medicine and Rehabilitation 98, 1560-1566.
| Crossref | Google Scholar | PubMed |
Tulsky DS, Carlozzi NE, Holdnack J, Heaton RK, Wong A, Goldsmith A, Heinemann AW (2017) Using the NIH Toolbox Cognition Battery (NIHTB-CB) in individuals with traumatic brain injury. Rehabilitation Psychology 62, 413-424.
| Crossref | Google Scholar | PubMed |
Vandenbroucke J, Von Elm E, Altman D, Gøtzsche P, Mulrow C, Pocock S, Poole C, Schlesselman J, Egger M, For the STROBE Initiative (2007) Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and Elaboration. PLoS Medicine 4, 1628-1654.
| Google Scholar |
Voormolen DC, Cnossen MC, Polinder S, Von Steinbuechel N, Vos PE, Haagsma JA (2018) Divergent Classification Methods of Post-Concussion Syndrome after Mild Traumatic Brain Injury: Prevalence Rates, Risk Factors, and Functional Outcome. Journal of Neurotrauma 35, 1233-1241.
| Crossref | Google Scholar | PubMed |
Vos L, Williams MW, Poritz J, Ngan E, Leon-Novelo L, Sherer M (2020) The Discrepancy Between Cognitive Complaints and Neuropsychological Test Findings in Persons With Traumatic Brain Injury. Journal of Head Trauma Rehabilitation 35, E382-E392.
| Crossref | Google Scholar | PubMed |
Zanon C, Brenner RE, Baptista MN, Vogel DL, Rubin M, Al-Darmaki FR, Gonçalves M, Heath PJ, Liao H-Y, Mackenzie CS, Topkaya N, Wade NG, Zlati A (2021) Examining the dimensionality, reliability, and invariance of the depression, anxiety, and stress Scale-21 (DASS-21) across eight countries. Assessment 28, 1531-1544.
| Crossref | Google Scholar | PubMed |
Zeldovich M, Bockhop F, Covic A, Cunitz K, Polinder S, Haagsma JA, Von Steinbuechel N (2022) Reference Values for the Rivermead Post-Concussion Symptoms Questionnaire (RPQ) from General Population Samples in the United Kingdom, Italy, and The Netherlands. Journal of Clinical Medicine 11, 4658.
| Crossref | Google Scholar | PubMed |