Island dwarfism in a tropical Australian python (Simalia amethistina)
Alastair Freeman A C , Ian Bell B D and Daniel J. D. Natusch
A C , Ian Bell B D and Daniel J. D. Natusch  E *
E *
A
B
C Present address:
D Present address:
E
Abstract
Dwarfism in insular populations of snakes has been documented at several localities around the world and it has been largely attributed to a phenotypic response of these isolated populations to food availability and composition. The incidental collection of size data for the Milman Island population of Simalia amethistina in north Queensland has brought to light a previously unknown example of island dwarfism in a tropical python species in Australia. Simalia amethistina on Milman Island are significantly smaller than conspecifics from the nearby mainland, with the maximum recorded size being only as large as the smallest 50% of mainland specimens. We attribute this dwarfism to the lack of large-bodied prey species available on Milman Island.
Keywords: Cape York, food availability, insular, Lockerbie Scrub, Milman Island, prey composition, Simalia kinghorni, snake.
Introduction
Populations of snakes occurring on islands often exhibit markedly smaller body sizes than mainland conspecifics, a phenomenon referred to as island dwarfism (Boback 2003). This difference is typically correlated with resource composition and availability (Shine 1987; Boback 2003; Jessop et al. 2006; Tanaka 2011; Luiselli et al. 2015). Dwarfism has been documented in populations of the elapid snake Notechis scutatus inhabiting small islands off the coast of South Australia (Shine 1987) but is unknown in boid snakes inhabiting tropical Australia.
The scrub python (Simalia amethistina) occurs along the north-eastern coast of far north Queensland and the Torres Strait, predominately in closed forest habitat (Cogger 2014). Scrub pythons are among the longest snake species in the world. With a maximum confirmed length of 5.65 m and mass of 24 kg (Fearn and Sambono 2000), and an unconfirmed length of over 8 m (Greer 1997), they are arguably the largest terrestrial carnivore in Australia. Reflecting frequent arboreality, scrub pythons are slender-bodied (Fig. 1a). Males engage in combat bouts during the mating season (in cooler months of the year) and females lay a clutch of approximately 12 eggs in the late dry-season. Scrub pythons consume a wide variety of avian and mammalian prey and sometimes consume relatively large prey – including macropodid marsupials.
Size distribution of scrub pythons (Simalia amethistina) from (a) Milman Island and (b) the nearby mainland. The hatched line on each figure represents the largest specimen from Milman Island.
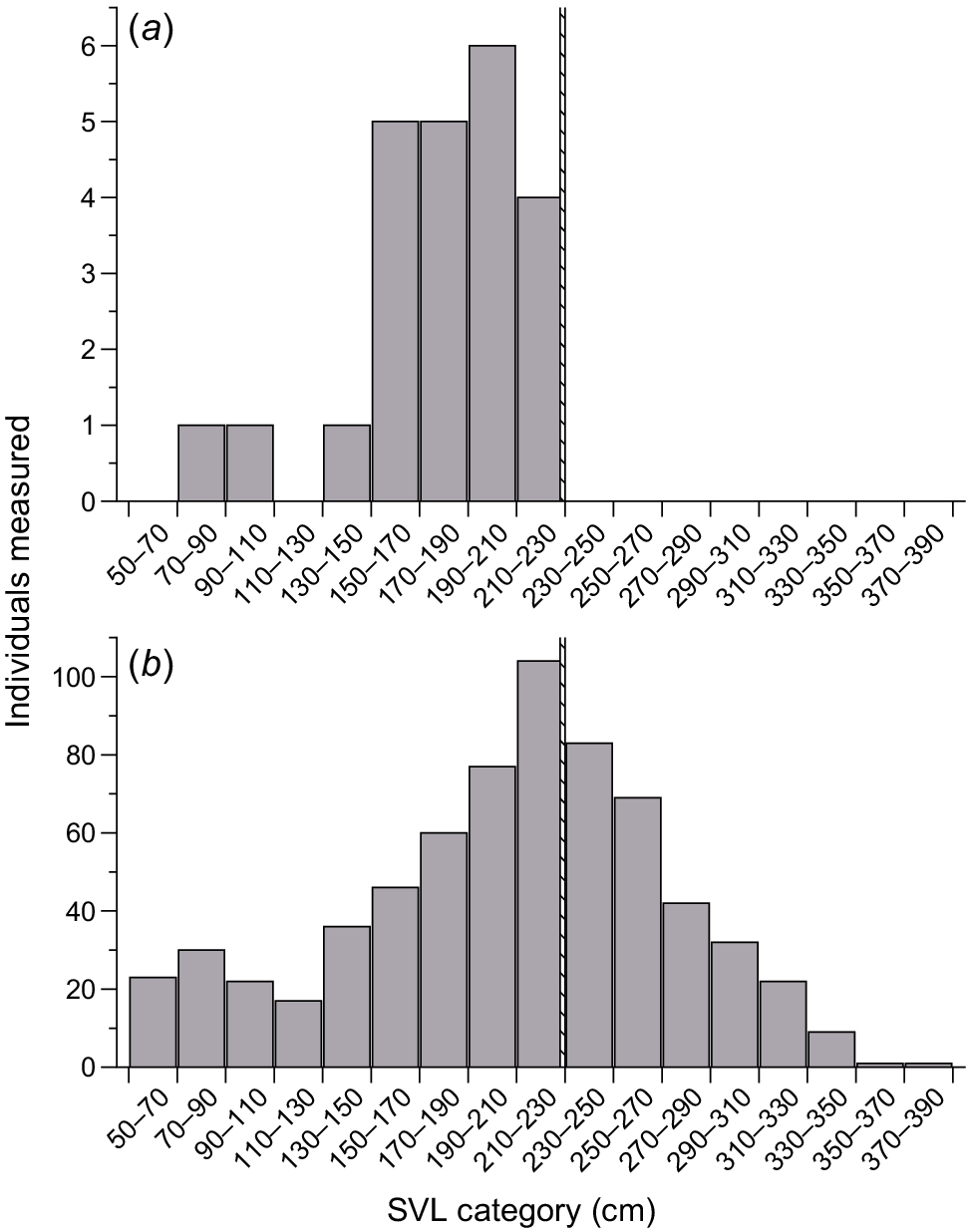
Small S. amethistina have been observed on Milman Island off the north Queensland coast since 1990 and it has been posited that a ‘dwarf’ population may occur there. To test whether Milman Island is home to a dwarf population of these pythons, we measured individuals on the island and compared their body sizes to pythons captured on the nearby mainland.
Study area
Milman Island (11.169540°S, 143.016623°E) is an uninhabited, densely wooded sand cay located approximately 23 km off mainland Australia, in the far northern section of the Great Barrier Reef Marine Park, Queensland. Milman Island is approximately 2.4 km in circumference and 24 ha in size. An intertidal reef flat (area ~ 560 ha) extends around much of the island. Since 1990, scientists from the Queensland Parks and Wildlife Service (QPWS) have been monitoring nesting sea turtles on the island. While on the island, the authors collected incidental data on the resident population of S. amethistina that occurs there. Pythons from the nearby mainland were examined to compare body sizes with snakes from Milman. The study area from which mainland snakes were collected is described in Natusch et al. (2022).
Methods
Between 2006 and 2024, pythons from Milman Island and the nearby mainland were hand caught and placed in cloth bags. Snout–vent length (SVL) was measured to the nearest centimetre using a stainless-steel tape measure and pythons were weighed to the nearest gram using Pesola™ spring scales. When possible, individuals were sexed using cloacal probing and were tagged with a Passive Integrated Transponder (PIT) or an individual scale was clipped and cauterized. Pythons were then released where they were caught and no recaptured specimens were included within analyses. The same procedures were followed for handling and measurement of mainland specimens captured between 2012 and 2017.
We used analysis of variance (ANOVA) with site and sex as factors and SVL as the dependent variable on our full dataset to test for differences in body size between island and mainland specimens. Because body mass is correlated with SVL, we included SVL as a covariate in an analysis of covariance (ANCOVA) to examine differences in body mass/condition between sites. Because island dwarfism in snakes is best tested by examination of mean and maximal sizes of adult snakes, we repeated our analyses but removed juveniles and restricted our tests to sexually mature specimens. Shine and Slip (1990) considered that 70% of mean adult body size is the typical size at maturity for Australian pythons. We set this threshold based on the mean of the smallest population in our study (in this case, Milman Island). In reality, we do not know the sizes of maturity for Milman Island S. amethistina, but this figure is nevertheless useful for standardising comparisons between sites (in the event that sampling of one population was skewed towards juveniles). Finally, to help understand whether the small body sizes of pythons from Milman Island are due to dwarfism and not small sample sizes, we took a random subsample from the dataset containing mainland specimens equal to the number of specimens measured on Milman and repeated this for 1000 bootstraps. We then compared the mean of these 1000 subsamples with the mean size of Milman Island pythons. We conducted all statistical analyses in JMP Pro 18 (SAS Institute, Cary, NC). Although our SVL measures conformed to a normal distribution, we ln-transformed body mass measures to meet the normality and homogeneity of variance assumptions of our statistical tests.
Results
In total, we captured 24 specimens of S. amethistina on Milman Island and 687 specimens from the nearby mainland. The mean body size of pythons from Milman Island was 177.9 cm ± 7.5 cm SVL versus 206 ± 2.5 cm SVL from the nearby mainland (Fig. 2). After deletion of a non-significant two-way interaction between sex and site, the result of our ANOVA on the full dataset revealed that pythons from Milman were shorter than mainland conspecifics (F1,657 = 4.23, P = 0.04). We found no statistically significant differences in body mass/condition between sites (F1,657 = 0.84, P = 0.36) or between the sexes (P > 0.05 for both SVL and mass). Our ANCOVA with SVL as a covariate revealed that pythons from Milman were heavier than Mainland specimens at similar body lengths (F1,652 = 19.6, P < 0.001).
Differences in mean snout–vent length (SVL) between (a) all scrub pythons (Simalia amethistina) and (b) adult scrub pythons from Milman Island and the nearby mainland.
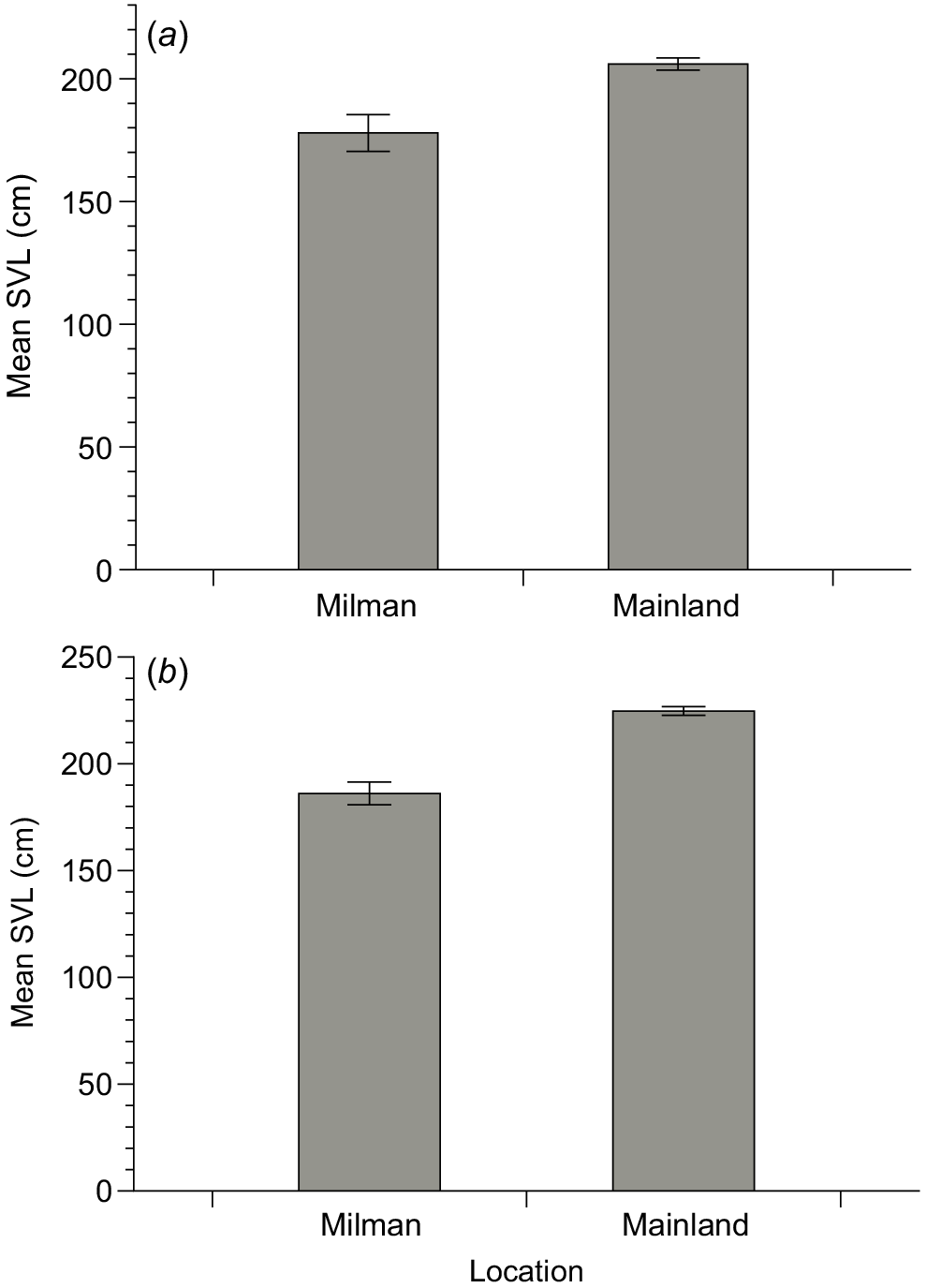
Restriction of our analysis to adult specimens (i.e. those > 130 cm SVL) resulted in mean body sizes of 186.1 ± 5.3 cm SVL for Milman Island and 224.7 ± 2.0 cm SVL for nearby mainland specimens, strengthening the result of our full analysis (i.e. smaller SVL in Milman specimens: F1,602 = 6.6, P < 0.001) (Fig. 2) and reconfirming that, at similar body lengths, Milman Island pythons were heavier-bodied than their mainland counterparts (F1,562 = 6.97, P = 0.009).
Our analysis of maximal body sizes of mainland versus Milman Island pythons showed that 50% of all adult mainland specimens were larger than the largest (223 cm SVL) Milman Island specimen (Fig. 1). The largest Milman Island specimen measured only 60% of the maximum size recorded for the mainland population (223 versus 370 cm SVL, respectively).
Finally, our analysis of 1000 bootstraps randomly subsampling the mainland dataset revealed that on only 20 (2%) occasions were mean body sizes of pythons from the mainland smaller than the sample from Milman Island, supporting the theory of dwarfism despite the relatively small sample size.
Discussion
Our analyses strongly support the idea that a dwarf population of S. amethistina occurs on Milman Island. Mean body sizes are 40 cm smaller than mainland pythons and all Milman specimens sit within the smallest 50% of the size range of their mainland conspecifics. Dwarfism in insular populations of snakes is not uncommon and has been reported in tiger snakes (Notechis scutatus) in southern Australia (Shine 1987), boa constrictors (Boa constrictor) on offshore islands of Belize (Boback 2006), and asp vipers (Vipera aspis) and western whip snakes (Hierophis viridiflavus) in the Italian Mediterranean (Luiselli et al. 2015).
The only plausible scenario that would invalidate our dwarfism theory is that larger specimens occur on Milman Island but were never located by us. Although this cannot be ruled out, we consider the likelihood to be low. First, the island is exceptionally small, is covered in stunted vegetation, and is relatively easy to survey in its entirety. The number of places for large snakes to hide (and thus be missed by us) is limited. Secondly, our random bootstrapping analysis accounting for our small sample size suggests the probability that Milman and mainland specimens have similar body sizes is extremely low (2%). Moreover, our comparative analysis of maximal body sizes suggests that, if dwarfism was not valid, half of the 24 specimens we located should have been larger than the largest individual we recorded on the island.
Dwarfism in island snakes is thought to be a response to food availability (Boback 2003; Tanaka 2011; Luiselli et al. 2015). On the mainland, the diet of adult S. amethistina includes numerous species of small to large-sized mammals, including rats (Rattus sp.), long-nosed (Perameles nasuta) and northern brown (Isoodon macrourus) bandicoots, brush tailed possums (Trichosurus vulpecula), spectacled flying foxes (Pteropus conspicillatus), feral cats (Felis catus), red-legged pademelons (Thylogale stigmatica) and agile wallabies (Macropus agilis) (Shine and Slip 1990; Fill et al. 2012; Natusch et al. 2021). Other than flying foxes, which have been recorded rarely on Milman Island, there are no terrestrial or arboreal mammals resident on the island. There have been only three observations of S. amethistina feeding on Milman Island: in 1994, pythons were recorded on two occasions feeding on rainbow bee-eaters (Merops ornatus), a small passerine (Dobbs et al. 1997), and in 2018 a snake was observed feeding on a frigate bird (Frigata sp.) on the forest floor (S. Menzies, pers. comm.).
Pythons have been observed on the beach and rock flats at night in the intertidal zone, either actively foraging or in ambush (A.B. Freeman and I. Bell, pers obs.). Foraging at night in the intertidal zone has not been documented elsewhere for this species and appears to be unique to Milman Island. Ghost crabs (Ocypode sp.) are common on Milman Island beaches, and during the summer sea turtle nesting season, turtle hatchlings (Chelonia mydas and Eretmochelys imbricata) are regularly observed making their way to the water. Both would appear to be potential prey items. The intertidal zone and foreshore area are also wading-bird roosting and tern nesting areas. These species are also potential prey items that may encourage the intertidal and beach foraging behaviour we have observed in S. amethistina.
Our finding that Milman Island pythons are heavier than mainland snakes of the same body length is intriguing. Plausibly, the dearth of large-bodied prey on Milman has removed the need for large body size, but the smaller pythons on the island still enjoy high abundances of smaller prey that have a favourable influence on body condition. Clearly, further sampling of the Milman Island population is needed to better understand the observed dwarfism. Experimental trials would help to elucidate whether the observed size difference between mainland and island populations is a local genetic adaption or a phenotypically adaptive response to differences in prey composition (Aubret and Shine 2009). If the former, the Milman Island S. amethistina may be a genetically distinct taxon worthy of specific conservation management.
Acknowledgements
Data from Milman Island were collected by the authors during marine turtle monitoring fieldtrips to Milman Island by Queensland Parks and Wildlife Service (QPWS) over several years. We thank the Traditional Owners of the area for allowing us access to undertake research on their Country. We kindly thank two anonymous reviewers for comments that improved the quality of this manuscript.
References
Aubret F, Shine R (2009) Genetic assimilation and the postcolonization erosion of phenotypic plasticity in island tiger snakes. Current Biology 19(22), 1932-1936.
| Crossref | Google Scholar | PubMed |
Boback SM (2003) Body size evolution in snakes: evidence from Island populations. Copeia 2003(1), 81-94.
| Crossref | Google Scholar |
Boback SM (2006) A morphometric comparison of Island and mainland boas (Boa constricta) in Belize. Copeia 2006, 261-267.
| Crossref | Google Scholar |
Dobbs KAL, Miller JD, Card MA, Mather M, Haselmeyer J (1997) Birds of Milman Island. Corella 21(2), 37-43.
| Google Scholar |
Fearn S, Sambono J (2000) A reliable size record for the scrub python Morelia amethistina (Serpentes: Pythonidae) in north east Queensland. Herpetofauna 30(1), 2-6.
| Google Scholar |
Fill JM, McBride P, Powell AJ, Shanahan LK, Stark JR, Freeman AB, Curran TJ (2012) Diet of amethystine (Morelia kinghorni) and carpet pythons (Morelia spilota) in north Queensland, Australia. Herpetological Review 43(1), 30-34.
| Google Scholar |
Jessop TS, Madsen T, Sumner J, Rudiharto H, Phillips JA, Ciofi C (2006) Maximum body size among insular Komodo dragon populations covaries with large prey density. Oikos 112(2), 422-429.
| Crossref | Google Scholar |
Luiselli L, Petrozzi F, Mebert K, Zuffi MA, Amori G (2015) Resource partitioning and dwarfism patterns between sympatric snakes in a micro-insular Mediterranean environment. Ecological Research 30(3), 527-535.
| Crossref | Google Scholar |
Natusch DJD, Lyons JA, Shine R (2021) Rainforest pythons flexibly adjust foraging ecology to exploit seasonal concentrations of prey. Journal of Zoology 313(2), 114-123.
| Crossref | Google Scholar |
Natusch D, Lyons JA, Shine R (2022) Spatial ecology, activity patterns, and habitat use by giant pythons (Simalia amethistina) in tropical Australia. Scientific Reports 12, 5274.
| Crossref | Google Scholar | PubMed |
Shine R (1987) Ecological comparisons of island and mainland populations of Australian tigersnakes (Notechis: Elapidae). Herpetologica 43(2), 233-240.
| Google Scholar |
Shine R, Slip DJ (1990) Biological aspects of the adaptive radiation of Australasian pythons (Serpentes: Boidae). Herpetologica 46, 283-290.
| Google Scholar |
Tanaka K (2011) Phenotypic plasticity of body size in an insular population of a snake. Herpetologica 67(1), 46-57.
| Crossref | Google Scholar |


