Size matters: influence of dwarfing genes and the Lcol-A1 allele on coleoptile growth and crop establishment in wheat (Triticum aestivum)
Jordan A. Bathgate A B * , Juan S. Moroni A , Felicity A. J. Harris A B , Russell F. Eastwood C and Greg J. Rebetzke
A B * , Juan S. Moroni A , Felicity A. J. Harris A B , Russell F. Eastwood C and Greg J. Rebetzke  D
D
A
B
C
D
Abstract
Deep sowing allows growers to access deep soil moisture and ensure timely crop establishment in maximising yield potential. However, wheat cultivars containing the ‘Green Revolution’ Rht1 and Rht2 dwarfing genes often exhibit reduced crop establishment when sown deeper than 60 mm because of short coleoptile length (CL). Novel gibberellin biosynthetic mutant dwarfing genes associated with reduced plant height and greater CL offer potential alternatives to Rht1 and Rht2.
Determine CL and coleoptile diameter (CD) for genotypes and near-isogenic lines (NILs) with alternative dwarfing genes and a novel CL allele, and to validate plant establishment when sown deep in fields.
A diverse set of 101 wheat genotypes containing different dwarfing genes, including a novel long coleoptile allele, Lcol-A1, were assessed for CL and CD in temperature-controlled growth-chamber experiments (CE). The CE-phenotyped CL and CD were validated for plant establishment with shallow (4–5 cm) and deep (11–15 cm) sowing in two field experiments.
Compared with genotypes with Rht1 and Rht2, CL was significantly longer in genotypes with Rht13 (+23%), Rht18 (+24%), and Lcol-A1 (+16%) alleles across CE experiments. Longer CL significantly improved crop establishment with deep sowing in the field (16 and 33 plants m−2 for short- and long-coleoptiles respectively).
Longer CL associated with Rht13, Rht18 and Lcol-A1 allele improved crop establishment. Lcol-A1 allele was linked to a small increase in grain yield with deep sowing, while Rht18 was comparable to Rht2 with deep sowing.
Gibberellin-biosynthetic dwarfing genes, together with the Lcol-A1 allele, show promise for improving establishment with deep sowing.
Keywords: climate change, coleoptile diameter, coleoptile length, deep sowing, Lcol-A1, reduced height genes, Rht13, Rht18, seedling vigour, wheat.
Introduction
Timely sowing and emergence of winter cereals is a key consideration in ensuring even crop establishment and grain yield potential. In turn, seed must be either dry sown with the expectation of timely rain or placed into a moist seedbed to ensure germination and coleoptile elongation (Fletcher et al. 2016). However, increasing variability in the timing of seasonal breaks and large sowing programs extending the duration of sowing operations may require sowing deeper into soil moisture to assure timely germination and crop emergence (Flohr et al. 2021). Deep sowing is a strategic technique used by some growers to access soil moisture at depth when surface soil layers are dry (Mahdi et al. 1998; Schillinger et al. 1998; Matsui et al. 2002). Deep sowing is used to optimise timing of germination and to ensure that crop development aligns closely with seasonal conditions, thereby maximising yield potential (Schillinger et al. 1998; Gooding et al. 2006). However, growers commonly observe low rates of establishment and reduced grain yield when deep sowing is implemented (Rebetzke et al. 2007; Mohan et al. 2013).
Wheat cultivars commonly contain the gibberellic acid (GA)-insensitive dwarfing genes Rht1 (syn. Rht-B1b) and Rht2 (syn. Rht-D1b), both identified and delivered globally during the ‘Green Revolution’ (Allan 1980). These reduced-height genes shorten coleoptile length, reducing the adaptation of many wheat cultivars to deep sowing and slow leaf area development and early biomass (Richards and Lukacs 2002). Ideally, genotypes with increased coleoptile length and seedling vigour may reduce the risk associated with deep sowing practices and increase grain yield (Wildermuth et al. 2001; Rebetzke et al. 2007; Mohan et al. 2013). Field-validated, deep-sowing experiments using near-isogenic, long-coleoptile wheat lines showed an average of 18–20% yield increase with deep sowing across the Australian wheatbelt (Zhao et al. 2022). Alternative GA-sensitive dwarfing genes including Rht13 and Rht18 may be used in place of the current Rht1 and Rht2 genes to increase coleoptile length (Rebetzke et al. 2022). Additionally, the novel Lcol-A1 allele, associated with increased coleoptile length, may improve establishment and subsequent grain yield under deep sowing, although this gene has had limited evaluation (Bovill et al. 2019).
This paper presents controlled-environment and field studies aimed at evaluating coleoptile length and diameter for a range of genotypes containing dwarfing genes Rht1, Rht2, Rht13, and Rht18, in combination with the Lcol-A1 allele, and determining their suitability in breeding for improved adaptation to deep sowing. It is hypothesised that genotypes containing Rht13 or Rht18 dwarfing genes, and the Lcol-A1 allele will have longer coleoptiles, improved seedling establishment, and greater seedling vigour with deep sowing. These traits may improve grain yield under deep sowing conditions compared with the currently available cultivars containing Rht1 or Rht2 dwarfing genes.
Materials and methods
Germplasm
One hundred and one bread wheat genotypes varying in genetic background and representing commercial cultivars, near-isogenic lines (NILs), breeding lines, and experimental high vigour lines developed by CSIRO, were assembled for controlled-environment (CE) and field studies (Supplementary material Table S1). The genotypes represented different dwarfing genes, including Rht1 (n = 31 genotypes), Rht2 (n = 29), Rht13 (n = 3) and Rht18 (n = 33), and tall genotypes lacking known major dwarfing genes (n = 5), with some genotypes containing the Lcol-A1 allele (n = 26). Near-isogenic lines included Halberd (tall, Rht13 and Rht18), EGA Gregory (Rht1 and Rht18), Mace (Rht2 and Rht18), and Magenta (Rht2, Rht13 and Rht18). All seeds were derived from a single source to reduce confounding environment (maternal) with genetic effects on germination and seedling growth.
Genotyping
All entries were genotyped using a competitive allele-specific polymerase chain reaction (KASP) system utilising single nucleotide polymorphisms (SNPs) for dwarfing genes Rht1 and Rht2 (Ellis et al. 2002), Rht13 (Borrill et al. 2022), Rht18 (Ford et al. 2018) and the Lcol-A1 allele (Bovill et al. 2019).
Plant growth chamber coleoptile measurements
Seeds were visually inspected for any damage and then screened over a 2.5 mm sieve before sowing into plastic containers [53 (L) × 32 (W) × 20 cm (H)] containing a commercially-available seed-raising potting mix, Osmocote Professional® (ICL Growing Solutions, Bella Vista, NSW, Australia). Containers were evenly watered and allowed to drain before enclosing in large opaque plastic bags to maintain humidity and minimise drying. The covered containers were placed into Conviron® Adaptis A1000 plant growth chambers (Conviron, Grovedale, VIC, Australia) at two temperatures, 15°C or 27°C, in darkness for 200 degree-days (degree days or thermal time at base temperature 0°C), according to the methods of Rebetzke et al. (2007). The experimental design was a randomised complete block with four replicates. Seedlings were removed from containers at 200 degree-days and measured for coleoptile and total shoot length (i.e. coleoptile plus first leaf length), and coleoptile diameter by using a Mitutoyo Series 293® 25 mm micrometer (M.T.I. Qualos Pty. Ltd., Heidelberg West, VIC, Australia). All measurements were performed on two seedlings per genotype in all replicates. Coleoptile diameter was determined under a Leica® EZ4 HD dissecting microscope (Leica Biosystems, Mount Waverley, VIC, Australia) at two positions along the coleoptile, namely, 10 mm above the seed and 10 mm below the tip of the coleoptile. As the coleoptile is elliptical, two measurements were made of the minor and major axis (perpendicular to each other) at each measuring point. Coleoptile diameter was calculated using the formula for the area of an ellipse (a b) π/4 as described by Matsui et al. (2002).
Field evaluation
A subset of 67 genotypes from the coleoptile growth study representing the different dwarfing and Lcol-A1 genes was selected for field evaluation (Table S2) in 2020 at two sites, namely, Collingullie, southern New South Wales (35.0589°S, 147.0514°E) and Narrabri, northern New South Wales (30.2807°S, 149.7519°E). At the Collingullie site, the field experiment was sown with a custom-built, rigid tine trial seeder at two sowing depths, ~45 mm (shallow) and 110 mm (deep). Treated granular starter fertiliser [1 mL kg flutriofol + monoammonium phosphate (MAP) – N: 10 kg ha−1, P: 21.9 kg ha−1] was banded with seed on 15 May 2020, into 5-m-long, 0.21-m-spaced, five-row plots at a rate of 205 seeds m−2. At Narrabri, genotypes were sown at ~50 and 150 mm depth with granular starter fertiliser (MAP – N: 10 kg ha−1, P: 21.9 kg ha−1) banded with seed and an additional granular nitrogen fertiliser (urea – N: 92 kg ha−1) incorporated at sowing on 7 May 2020 into 6-m-long, 0.21-m-spaced, five-row plots at a seeding rate of 171 seeds m−2. Both experiments were conducted as a two-replicate, split-plot design with genotype as whole plots and sowing depth as subplots. Weather stations were positioned at each site to record air temperature, relative humidity (RH), soil temperature at both sowing depths (Collingullie only), and rainfall. Experiments were managed to exclude weeds and disease throughout the growing season, with additional nitrogen being supplied at early stem elongation at 44.6 kg ha−1 as urea ammonium nitrate at Collingullie and 36.8 kg ha−1 applied as granular urea at Narrabri. Plant establishment counts at Collingullie and Narrabri were performed along 1 and 0.5 m of sowing row respectively, ~30 days after sowing. Heading date was recorded for each plot along with plant height at Collingullie. At maturity, plots were end-trimmed to 3 and 4 m length at Collingullie and Narrabri respectively, and then machine-harvested for grain yield.
Field site descriptions
The Collingullie experiment site was an acidic light brown clay-loam, whereas the Narrabri site was a self-mulching neutral to alkaline grey clay. Both sites recorded decile 5 rainfall, with growing-season rainfall (April–October) of 362 mm (Collingullie) and 274 mm (Narrabri) (Tables S3, S4). At Collingullie, two low-temperature events of −0.8°C and −0.5°C occurred during the critical development period, along with three events of −2.6°C, −1.2°C and −1.0°C at Narrabri (see Figs S1, S2). No ear frost damage was observed.
Seedling emergence counts
At the Collingullie site, seedling counts were performed daily on 2 m of randomly marked furrow (separated into four 0.5 m sections across plots to encompass variability) and continued for 38 days after sowing until emergence ceased. Seedling count data were fitted against thermal time [(degree-days) air temperature at 1.2 m above ground] by using a non-linear Gompertz growth model with the following formula:
where A is the lower asymptote (set at 0 because no seedlings emerge prior to this point), C is the upper asymptote, M is the value of the midpoint (50% of maximum plant number), and B is the slope of the curve at X (Tipton 1984; Gan et al. 1992; Rebetzke et al. 2007). Estimated parameters included rate of emergence (B, slope of curve), final plant number (95% of maximum) and thermal time to final plant number (Fig. S3).
Early seedling growth and development measurements
Seedling growth and development were determined at the Collingullie site when seedlings in the 45 mm sowing depth reached ~4.5 Haun development stage at 47 days (504 degree-days, air temperature) after sowing. Seedlings were carefully excavated from four corners of each plot by using a shovel to dig emerged plants from 30 cm of row, before placing into plastic bags and storing in a darkened cool room (~3°C) until measuring. The following day, five randomly chosen plants from each bag were washed to remove soil from roots and the plants were measured for first leaf length and width (for leaf area estimate), coleoptile length, crown depth, and leaf and tiller number. Roots were removed and shoots were then dried in a forced-air dehydrator at 70°C for 4 days before weighing. Measurements were performed by replicate over 4 days and were made with a ruler to the nearest 0.5 mm. First leaf length was measured from the ligule to the leaf tip, and leaf width was measured at the widest position along the leaf and multiplied by 0.835, following Miralles and Slafer (1991). Crown depth was measured from the crown to the point of stem colour change from white (chlorotic) to green.
Statistical analysis
All continuous data were analysed using the linear mixed model procedure, as described by Gilmour et al. (1997) with spatial components fitted when appropriate using the ASReml-R package (version 4.2) within the R software environment (version 2024.04.0; Butler et al. 2017; R Core Team 2020). First, model assumptions were checked for homogeneity of variance and normality of residuals before interpreting model output. Mean effects and interactions were tested via Wald’s tests utilising approximate F-statistics. Statistical comparisons of treatments and interactions were performed by using posteriori contrasts, and pairwise comparisons among means were conducted using Dunnett’s T3 test. Additionally, post hoc comparisons were performed using the protected Fisher’s least significant difference (l.s.d.) test, with the average l.s.d. calculated.
Single point-in-time plant emergence count data were fitted to several statistical models for diagnostic comparison, including a linear mixed model (LMM), a generalised linear mixed model (GLMM) with Poisson family of distributions, and a negative binomial model using the GLM function in ASReml-R. Finally, count data were modelled using an LMM. Plant emergence count data collected from sowing to 38 days after sowing was fitted to a Gompertz non-linear regression against thermal time (base temperature 0°C) by using statistical software Genstat (version 21 for windows; VSN International 2020).
Statistical significance is reported at the α = 0.05 level, unless otherwise stated. Figures were generated using GraphPad Prism (version 10.4.1; GraphPad Prism 2020).
Results
Plant growth chamber: assessment of coleoptile length and diameter
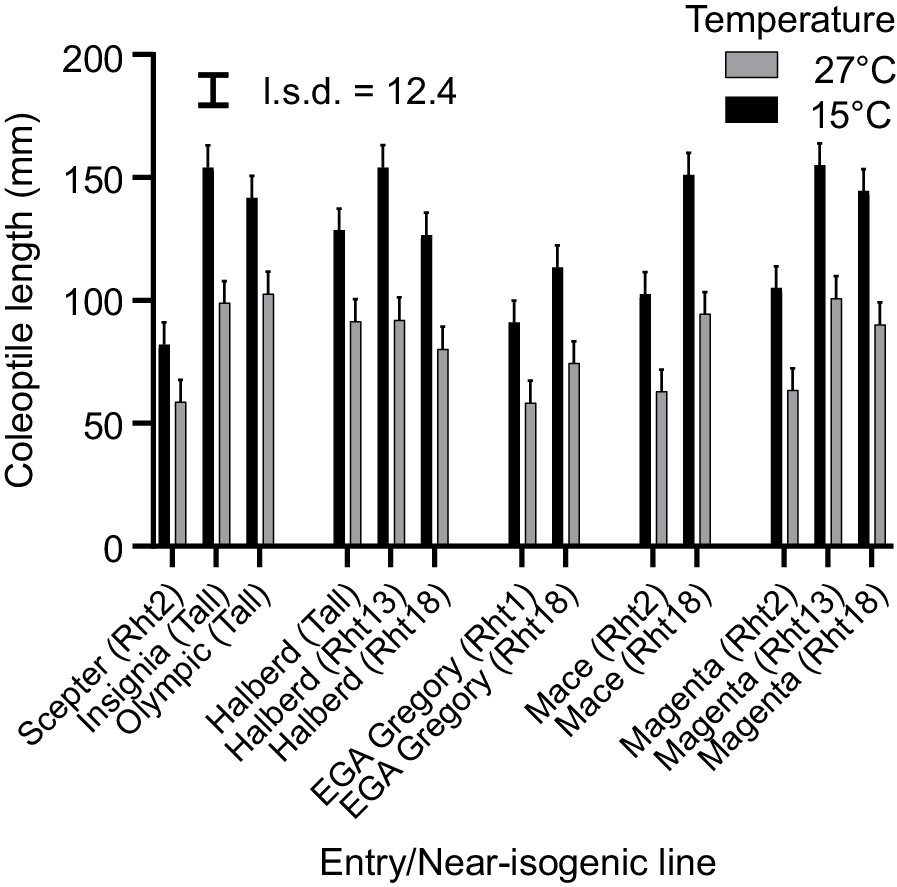
The mean coleoptile length in the CE was 128 mm at 15°C and 82 mm at 27°C, with ranges of 82–158 mm and 44–103 mm for 15°C and 27°C respectively (Table 1). Genotypic ranking for coleoptile length was consistent across both temperatures, reflecting a strong genetic correlation (rg = 0.92; P < 0.001). On a percentage change basis, comparing a genotype coleoptile length at 27°C with its maximum length at 15°C, we observed a variation in response ranging from 51.1% to 76.4% of the length at 15°C. This relationship was positively correlated with coleoptile length (rp = 0.45, P < 0.001). The coleoptile length of the tall wheat cultivar Olympic (absent of any dwarfing and Lcol-A1 genes) ranked among the longest of all genotypes, producing 142 and 103 mm at 15°C and 27°C respectively (Fig. 1). Similarly, coleoptile lengths of tall cultivars Insignia and Halberd were among the longest genotypes at both temperatures. The Rht2-containing semi-dwarf cultivar Mace had coleoptile lengths of 103 and 63 mm, whereas its Rht18-containing NIL Mace + Rht18 had coleoptile lengths of 151 and 95 mm at 15°C and 27°C respectively (Fig. 1). Genotypes having Rht1 or Rht2 and lacking the long Lcol-A1 allele produced the shortest coleoptiles across both temperature treatments (Table S1). That said, replacement of Rht2 with Rht18 in the Lcol-A1-containing Magenta NIL pair was associated with a significant increase in coleoptile length at both 15°C and 27°C (Fig. 1). Coleoptile lengths of Rht13 and Rht18 Halberd NILs were comparable at 15°C and 27°C to that of their tall parent Halberd.
| Dwarfing gene | Lcol-A1 allele | Coleoptile length (mm) | Coleoptile diameter (mm) | Genotypes (=n) | ||||
|---|---|---|---|---|---|---|---|---|
| 15°C treatment | 27°C treatment | 15°C treatment | 27°C treatment | |||||
| Rht1 | Short | 100 | 60 | 1.38 | 1.49 | 31 | ||
| Rht2 | Short | 96 | 59 | 1.55 | 1.74 | 26 | ||
| Long | 115 | 65 | 1.40 | 1.58 | 3 | |||
| Rht13 | Short | 119 | 80 | 1.29 | 1.38 | 2 | ||
| Long | 155 | 101 | 1.26 | 1.47 | 1 | |||
| Rht18 | Short | 130 | 82 | 1.40 | 1.45 | 15 | ||
| HET | 141 | 90 | 1.36 | 1.46 | 16 | |||
| Long | 150 | 90 | 1.34 | 1.40 | 2 | |||
| Tall | Short | 142 | 103 | 1.18 | 1.25 | 1 | ||
| Long | 132 | 87 | 1.23 | 1.33 | 4 | |||
| Mean | 128 | 82 | 1.34 | 1.45 | ||||
| l.s.d. | 10 | 10 | 0.14 | 0.14 | Total | 101 | ||
l.s.d. compares Lcol.A1 alleles within dwarfing gene groups across both temperature treatments.
Predicted coleoptile length of cultivars and near-isogenic lines from controlled environment phenotyping at 15°C or 27°C. Error bars above each bar represent +1 standard error of the mean. l.s.d. bar compares cultivars or lines.
Coleoptile diameter was strongly correlated for genotypes across both temperature treatments (rg = 0.77; P < 0.001), and mean diameters at 15°C and 27°C were significantly different at 1.34 and 1.45 mm respectively (Table 1). Genotypes containing the Rht2 and ‘short’ Lcol-A1 alleles produced the largest average coleoptile diameter across temperatures. By contrast, the other dwarfing and Lcol-A1 alleles varied in their association with coleoptile diameter across genotypes. Generally, coleoptile length increases were associated with reductions in coleoptile diameter across all genotypes (rg = −0.38; P < 0.001).
Field study
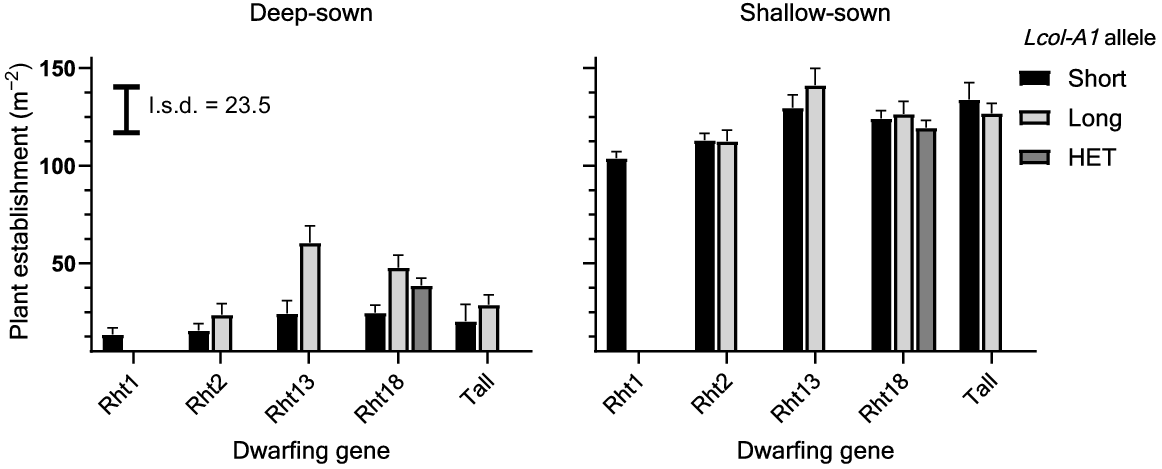
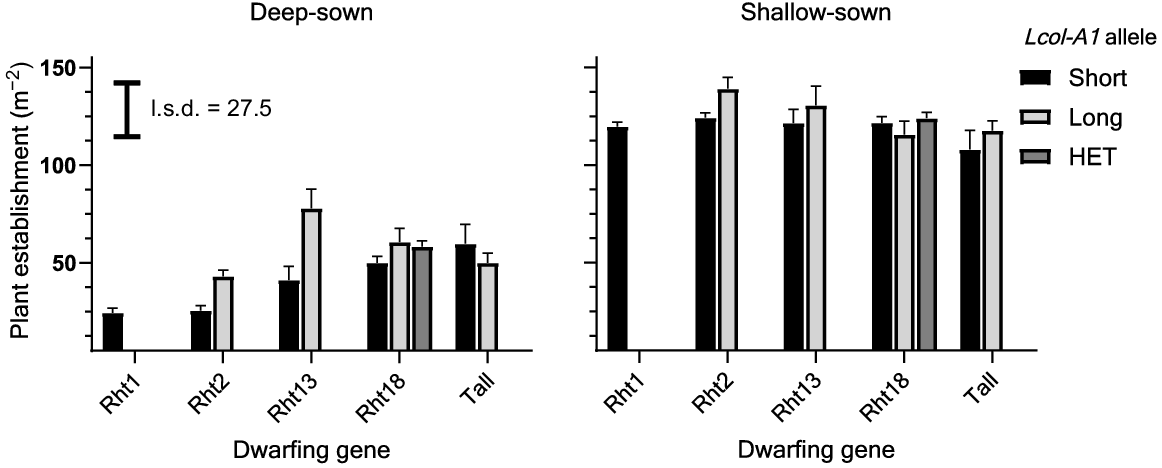
Average plant numbers with shallow sowing were 124 and 123 m−2 at Collingullie and Narrabri respectively. Deeper sowing significantly (P < 0.001) reduced seedling emergence and plant establishment at Collingullie and Narrabri, with reductions in mean plant number of 74% and 54% respectively, when compared with shallow sowing (Figs 2, 3). Deep sowing reduced plant number in Rht1 and Rht2 genotypes to a larger extent than in Rht13, Rht18 or tall genotypes, which produced the largest plant numbers along with those containing the Lcol-A1 allele. The Rht1 and Rht2 dwarfs produced significantly fewer plants across both sites (Figs 2, 3).
Predicted number of plants for different dwarfing and long-coleoptile Lcol-A1 genes for deep and shallow sowing at Collingullie, southern NSW, in 2020. Error bars above each bar represent +1 standard error of the mean. l.s.d. bar compares dwarfing-gene and Lcol-A1 allele groups. HET indicates that lines are heterozygous for the Lcol-A1 long-coleoptile allele.
Predicted number of plants for different dwarfing and long-coleoptile Lcol-A1 genes for deep and shallow sowing at Narrabri, northern NSW, in 2020. Error bars above each bar represent +1 standard error of the mean. l.s.d. compares between dwarfing genes and Lcol-A1 allele groups. HET indicates that lines are heterozygous for the Lcol-A1 long-coleoptile allele.
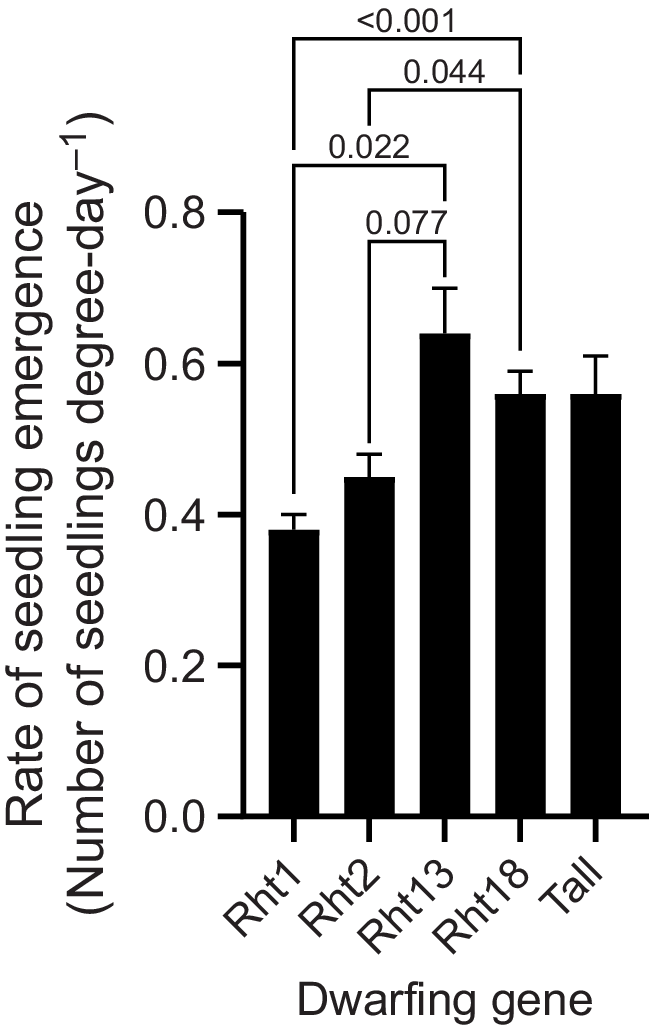
Deep sowing significantly influenced all three estimated emergence parameters from Gompertz non-linear regression (Table 2). Plant establishment was reduced 77% from 120 to 28 plants m−2 (P < 0.001), and thermal time to final establishment (95% of total emerged plants) increased from 226 to 265 degree-days (P < 0.001). Rate of seedling emergence was reduced from an average 0.828 with shallow sowing to 0.244 plants degree-day−1 (P < 0.001) with deep sowing. Genotypes containing the Rht1 and Rht2 dwarfing genes emerged slowest (Fig. 4) and with the smallest establishment counts compared with both Rht13 and Rht18 and tall genotypes. Additionally, presence of the long coleoptile Lcol-A1 allele resulted in significantly (P < 0.001) greater plant numbers with deep sowing. Thermal time to 95% emergence did not differ significantly among the dwarfing-gene groups or in the interaction with the Lcol-A1 alleles. Rate of emergence was significantly different among dwarfing-gene groups but not for the interaction with the Lcol-A1 allele. Shallow sowing resulted in similar establishment across all genotypes (Table 2).
| Dwarfing gene | Lcol-A1 allele | Rate of seedling emergence (seedlings degree-day−1) | 95% maximum plant number (m−2) | Thermal time to 95% maximum plant number (degree-days) | ||||
|---|---|---|---|---|---|---|---|---|
| Shallow sown | Deep sown | Shallow sown | Deep sown | Shallow sown | Deep sown | |||
| Rht1 | Short | 0.630 | 0.137 | 102 | 12 | 236 | 266 | |
| Rht2 | Short | 0.744 | 0.144 | 112 | 14 | 231 | 262 | |
| Long | 0.745 | 0.166 | 110 | 22 | 228 | 263 | ||
| Rht13 | Short | 1.038 | 0.152 | 126 | 24 | 214 | 278 | |
| Long | 0.877 | 0.498 | 140 | 50 | 231 | 253 | ||
| Rht18 | Short | 0.805 | 0.206 | 120 | 22 | 228 | 269 | |
| HET | 0.800 | 0.354 | 118 | 36 | 229 | 264 | ||
| Long | 0.860 | 0.313 | 124 | 46 | 223 | 267 | ||
| Tall | Short | 0.922 | 0.179 | 132 | 24 | 215 | 260 | |
| Long | 0.854 | 0.291 | 124 | 28 | 228 | 263 | ||
| Mean | 0.828 | 0.244 | 120 | 28 | 226 | 265 | ||
| l.s.d. | 0.242A | 16B | 16B | 24A | ||||
Rate of seedling emergence at Collingullie estimated from fitted Gompertz non-linear regression. Means for dwarfing-gene groups pooled for deep and shallow sowing. Error bars above each bar represent +1 standard error of the mean. Bars with P-values represent the significance of posteriori contrasts.
Across both field sites, increases in coleoptile length were positively correlated with increases in plant number (rp = 0.87; P < 0.01) with deep sowing (Table 3). This relationship was not significant (r = 0.09; n.s.) for shallow sowing at Narrabri but was significant (r = 0.39; P < 0.01) for shallow sowing at Collingullie. Coleoptile diameter had a weak negative relationship with establishment in deep sowing at Collingullie (r = −0.31; P < 0.05) and Narrabri (r = −0.36; P < 0.05), whereas the association was not statistically significant at shallow sowing depths at both sites.
| Site | Sowing depth (mm) | Pearson correlation of plant number with coleoptile | ||
|---|---|---|---|---|
| Coleoptile length | Coleoptile diameterA | |||
| Collingullie | 45 | 0.39* | −0.11n.s. | |
| 110 | 0.87** | −0.31* | ||
| Narrabri | 50 | 0.09n.s. | −0.00n.s. | |
| 150 | 0.87** | −0.36* | ||
Mean coleoptile length of all genotypes was significantly (P < 0.001) greater in the deep-sowing treatment (45 and 92 mm for shallow and deep respectively). Coleoptile length was significantly longer in tall genotypes and breeding lines containing the Rht13, Rht18 and Lcol-A1 alleles (Table 4). Deep sowing increased mean size of the first leaf (+7.9%; P < 0.01), with genotypes containing the Rht18 dwarfing gene having the largest leaves (Fig. 5). Leaf number per plant with deep sowing was reduced from 4.0 to 3.2 leaves per plant (Haun stage) (P < 0.01), with a corresponding reduction in tiller number from 1.3 to 0.4 tillers per plant (P < 0.01), whereas above-ground dry weight per plant was significantly (P < 0.01) reduced from 0.115 to 0.054 g. Shoot weight was largest in the Rht13, Rht18 and tall wheats, and genotypes containing the Lcol-A1 allele. Crown depth was increased with deep sowing (P < 0.01).
| Dwarfing gene | Lcol-A1 allele | Genotypes (n) | Coleoptile length (mm) | Crown depth (mm) | Above-ground shoot tissue weightA (g) | ||||
|---|---|---|---|---|---|---|---|---|---|
| Shallow sown | Deep sown | Shallow sown | Deep sown | Shallow sown | Deep sown | ||||
| Rht1 | Short | 18 | 45 | 73 | 30 | 62 | 0.1093 | 0.0495 | |
| Rht2 | Short | 16 | 43 | 76 | 29 | 65 | 0.1140 | 0.0416 | |
| Long | 3 | 45 | 89 | 23 | 55 | 0.0968 | 0.0595 | ||
| Rht13 | Short | 2 | 49 | 91 | 29 | 65 | 0.1267 | 0.0578 | |
| Long | 1 | 49 | 101 | 18 | 44 | 0.1234 | 0.0937 | ||
| Rht18 | Short | 9 | 46 | 86 | 28 | 60 | 0.1158 | 0.0540 | |
| HETB | 11 | 50 | 98 | 24 | 58 | 0.1242 | 0.0645 | ||
| Long | 2 | 51 | 108 | 22 | 68 | 0.1296 | 0.0563 | ||
| Tall | Short | 1 | 31 | 98 | 21 | 57 | 0.1183 | 0.0708 | |
| Long | 4 | 45 | 97 | 20 | 46 | 0.1159 | 0.0642 | ||
| Mean | 45 | 92 | 24 | 58 | 0.1150 | 0.0542 | |||
| l.s.d.C | 13 | 12 | 0.0069 | ||||||
Predicted first leaf area of cultivars and near-isogenic lines sown at Collingullie in southern NSW, with data averaged across deep and shallow sowings. Error bars above each bar represent +1 standard error of the mean. Asterisks and ‘n.s.’ indicate the significance and non-significance of posterior contrasts for each NIL pair. Tall (Halberd), Rht1 or Rht2 and the alternative Rht13 or Rht18 gene in each background.
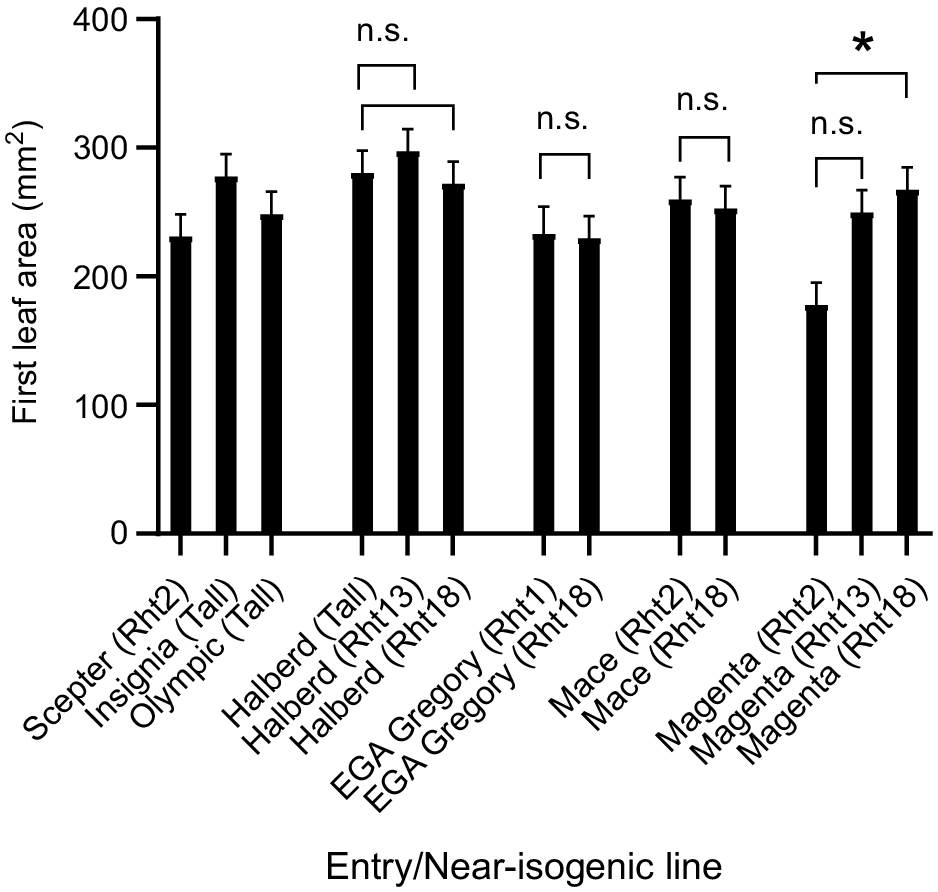
Deep sowing reduced mean plant height at Collingullie to a similar extent across Rht2- and Rht18-containing genotypes (Table 5).
| Dwarfing gene | Lcol-A1 allele | Genotypes (n) | Plant height Collingullie (cm) | Grain yield Collingullie (Mg ha−1) | Grain yield Narrabri (Mg ha−1) | |||
|---|---|---|---|---|---|---|---|---|
| Mean of shallow and deep sown | Shallow sown | Deep sown | Shallow sown | Deep sown | ||||
| Rht2 | Short | 16 | 87 | 7.829 | 4.363 | 4.605 | 2.802 | |
| Long | 3 | 98 | 6.722 | 5.370 | 4.614 | 3.341 | ||
| Rht18 | Short | 9 | 107 | 5.745 | 4.215 | 3.421 | 2.778 | |
| HETA | 11 | 109 | 6.126 | 4.979 | 3.392 | 3.014 | ||
| Long | 2 | 113 | 6.310 | 4.834 | 3.125 | 2.994 | ||
| Tall | Short | 1 | 121 | 6.015 | 5.546 | 3.777 | 2.726 | |
| Long | 4 | 126 | 5.098 | 4.371 | 3.088 | 2.426 | ||
| Mean | 109 | 6.264 | 4.811 | 3.717 | 2.869 | |||
| l.s.d. | n.s. | 0.990B | 0.695C | |||||
At Collingullie, grain yield was reduced (P < 0.001) 23% with deep sowing, from 6.26 to 4.81 Mg ha−1, being similar to Narrabri where grain yield was reduced (P < 0.001) 23%, from 3.72 to 2.87 Mg ha−1. Genotypes containing Rht2 produced the highest grain yields with shallow sowing across both sites. However, in the deep-sowing treatment, lines containing the Rht18 dwarfing gene achieved grain yields equivalent to the Rht2 groupings. The Lcol-A1 allele was associated with increased grain yield under deep sowing, but there was no interaction with grain yield under shallow sowing. Plant number and grain yield were positively correlated at Collingullie and Narrabri (r = 0.74, 0.66, P < 0.001).
Discussion
Plant growth chamber experiments
We observed an average 36% reduction in coleoptile length as soil temperature increased from 15°C to 27°C, consistent with findings reported in Bhatt and Qualset (1976) and Rebetzke et al. (2016), we also observed that the extent of this reduction, expressed as a percentage of the maximum coleoptile length at 15°C, was correlated with coleoptile length. This suggests that genotypes with longer coleoptiles are less sensitive to elevated soil temperatures, which are likely to be experienced with climate change in Australia and elsewhere.
However, the increase in coleoptile diameter with an increasing temperature was not consistent with that reported by Rebetzke et al. (2004). The latter showed a reduction in diameter with a small temperature increase from 15°C to 19°C. Differences in diameter may be drawn from Allan et al. (1962), who showed that changes in coleoptile morphology were associated with coleoptile parenchyma cell length and number across wheat genotypes. It is likely that coleoptile parenchyma cell anatomy, and therefore coleoptile diameter, respond differently to temperature across genotypes. Together, these findings suggest that increasing temperatures are likely to affect coleoptile length and diameter to influence plant number and establishment. Increases in soil temperature will reduce coleoptile length to reduce emergence with deeper sowing. Therefore, increasing coleoptile length of commercial wheat varieties is important for adaptation to increased early season soil temperatures and to ensure crop establishment and production in future climates (Kirkegaard et al. 2014; Hunt et al. 2018).
Wheat genotypes containing the Rht1 and Rht2 dwarfing genes were, on average, 29% and 30% respectively, shorter in coleoptile length than mean was the length of all tall wheat genotypes, consistent with other reports (Wang et al. 2014; Tang et al. 2021). By contrast, coleoptiles in Rht13- and Rht18-containing genotypes were of comparable lengths to tall wheats (difference in length of less than 3%), suggesting that Rht13 and Rht18 were not associated with reductions in coleoptile length. Indeed, coleoptile length was maintained with Rht13 and Rht18 dwarfing genes in the tall, long-coleoptile Halberd background. Specific comparisons within near-isogenic dwarfing-gene pairs confirmed the greater coleoptile lengths associated with Rht13 and Rht18 than with Rht1 and Rht2 parents across multiple genetic backgrounds. The influence of Lcol-A1 allele could be evaluated only in Rht2, Rht13, Rht18, and tall wheat genotypes. Across Lcol-A1 allele-containing dwarfing-gene backgrounds, coleoptile length increased with Lcol-A1 allele, except in the tall-genotype group. However, both Rht13 and tall-wheat groups contained only one entry each for Lcol-A1, limiting conclusions of the allele effect on coleoptile length. Nonetheless, replacement of Rht2 with Rht18 in the Magenta dwarfing-gene NIL fixed for Lcol-A1 indicated a large increase in coleoptile length at both cooler and warmer temperatures with Rht18. Bovill et al. (2019) reported that the presence of the Lcol-A1 allele increased coleoptile length by an average of 18%, consistent with our results representing different dwarfing genes and genetic backgrounds. However, the Magenta NILs suggest the possibility of a dwarfing gene × Lcol-A1 interaction in some genetic backgrounds.
Differences in coleoptile diameter were not consistent among dwarfing-gene groups. There was a significant negative relationship between coleoptile diameter and length (r = −0.38, P < 0.001), and this relationship appeared independent of the dwarfing gene. Presence of the Lcol-A1 long-coleoptile allele had no influence on coleoptile diameter.
Plant establishment and the relationship to dwarfing genes and the Lcol-A1 allele
Deep sowing reduced plant numbers in all genotypes and particularly for shorter-coleoptile genotypes. Reduced plant number was likely to be due to the inability of shorter coleoptile genotypes to elongate to the soil surface, particularly in the surface-crusting clay loam at Collingullie. In turn, there is likely to be a greater reliance on elongation of the first leaf to grow through to the soil surface. The physical structure of the first leaf was reported to bend and buckle in response to uneven soil aggregate and surface crusting to slow emergence and reduce plant number (Lutcher et al. 2019). Soil characteristics were recorded only through soil tests (Table S5) in this study; nonetheless, the strong relationship between coleoptile length and plant number with deep sowing highlighted the importance of greater coleoptile length in Rht13, Rht18, and tall and Lcol-A1-containing genotypes. This is especially notable when compared with the reduced plant number in short-coleoptile Rht1 or Rht2 dwarfing genes, and short Lcol-A1 allele-containing wheats. Although flutriafol, a fungicide known to reduce coleoptile length (Radford et al. 1989), was applied at the Collingullie site, genotype rankings for establishment were consistent across both field locations. This suggests that genotype performance was comparably estimated, even if overall emergence was slightly reduced at the Collingullie site. It is also likely that environmental factors, such as the crusting soil type at this location, contributed to the reduced establishment.
Similar seedling emergence rates with shallow sowing across dwarfing gene and Lcol-A1 allele groups suggest that limitations to emergence associated with Rht1 or Rht2 are not likely to be associated with reduced establishment at shallow sowing depths. This has been established in other studies evaluating Rht1- and Rht2-containing genotypes (e.g. Rebetzke et al. 2007; Tang et al. 2021). Herein, changes to seedling coleoptile length and diameter associated with dwarfing genes Rht1, Rht2, Rht13, Rht18, and Lcol-A1 did not improve establishment at shallow sowing depths, nor did they show negative effects.
Deeper sowing increased thermal time for seedling emergence and decreased final seedling number. Increased time to seedling emergence with deep sowing was observed in other studies (Mahdi et al. 1998; Matsui et al. 2002). Differences associated with dwarfing gene groups and Lcol-A1 allele suggest similar rates of emergence at shallow sowing. However, with deep sowing, longer coleoptiles of Lcol-A1 and Rht13, Rht18, and tall wheat genotypes produced greater plant numbers than did non-Lcol-A1 or the Rht1 and Rht2 dwarfing genotypes.
Rate of emergence from the fitted plant number curves was a measure of how rapidly seedlings emerged to achieve the final plant number. With shallow sowing, the Rht2 group had the smallest rate of emergence. However, time to maximum plant number was not different from other dwarfing groups. The Rht13, Rht18 and tall genotypes all had greater rates of seedling emergence with deep sowing. The increase in coleoptile length associated with Lcol-A1 showed small but non-significant increases in the rate at which seedlings emerged for all dwarfing gene groups. The incorporation of alternative dwarfing genes Rht13, Rht18 and the Lcol-A1 allele in elite genotypes would likely increase establishment and seedling vigour from deep sowing, with no negative effect when shallow sowing.
Leaf size remained unchanged with the addition of Rht13 or Rht18 in NILs, except in Magenta where leaf area increased with Rht18. These results are consistent with those of Rebetzke et al. (2022) but not with those of Tang et al. (2021), who indicated that leaf size is reduced with Rht18.
Plant height, grain yield and the relationship to dwarfing genes and the Lcol-A1 allele
Reduction in mature plant height associated with the Rht18 dwarfing gene was consistent with reductions reported elsewhere (e.g. Rebetzke et al. 2011; Wang et al. 2014), and illustrate the potential for its use in breeding and varietal development. The interaction of different dwarfing genes with Lcol-A1 observed for plant height in this study was small, but needs further investigation.
The Rht18 gene was associated with reduced grain yield under shallow sowing, but similar yield under deep sowing to Rht2 genotypes. Interpretation of grain yield responses of genotypes with dwarfing genes and Lcol-A1 allele and their interactions was difficult because of fewer lines. The elite Rht2-containing cultivars yielded the most in shallow-sown treatments, because of years of breeding and selection for grain yield and harvestability. Genotypes containing Rht18 had not been selected for yield, making comparisons with established cultivars for grain yield inappropriate. Although Rht2 genotypes produced the highest grain yields under shallow sowing conditions, this is likely to reflect the extensive breeding effort and selection history involving Rht2 in Australian breeding programs. As breeding with alternative dwarfing genes, such as Rht13 and Rht18, evolves, it is anticipated that these genotypes may likely reach comparable, if not superior, yield performance under a wider range of sowing depths.
The Lcol-A1 allele under deep sowing led to increased grain yield in most cases, with no real negative impacts noted in this study. It is likely that the Lcol-A1 allele may be actively incorporated into breeding programs, and at the time of writing this paper, this is occurring (R. Eastwood, pers. comm.).
Plant number and yield are not always correlated, as was observed at both field sites in this study. Anderson (1986) demonstrated that in low-rainfall environments, yields can be maintained with as few as 30 plants m−2. However, this low plant density is suboptimal for weed suppression. For example, Lemerle et al. (2004) reported that a minimum of 200 plants m−2 is typically required to suppress annual ryegrass (Lolium rigidum). In our study, genotypes containing Rht1 and Rht2 often fell below the 30 plants m−2 threshold under deep sowing, whereas those containing Rht13, Rht18, and the addition of the Lcol-A1 allele, more reliably exceeded this level. These results highlight the need to consider both agronomic context and management goals (e.g. weed control versus yield resilience) when interpreting establishment–yield relationships.
Currently, growers wishing to use deep sowing to reach subsoil moisture must establish crops using old tall wheat varieties. Older varieties often lodge and have lower grain yields than modern semi-dwarf wheat (Anderson and Smith 1990). Incorporation of the Rht13, Rht18, and Lcol-A1 allele will allow growers to sow seed deeper than currently practiced, without the issues of reduced yield and lodging associated with tall wheat. Efforts in Australia and elsewhere are focusing on GA biosynthetic mutant dwarfing genes such as Rht18 in breeding long-coleoptile wheats (Zhao et al. 2022). Continued research and breeding will deliver long-coleoptile semi-dwarf wheats for deep sowing to ensure timely germination and emergence when surface soils are too dry.
Conclusions
Controlled growth chamber experiments demonstrated the effect of dwarfing genes Rht1, Rht2, Rht13, Rht18 and the coleoptile-modifying Lcol-A1 allele on coleoptile length in 101 genotypes. A subset of genotypes was sown at multiple depths in field experiments at two separate locations and showed that longer coleoptiles in Rht13-, Rht18- and Lcol-A1-containing wheats conferred greater establishment with deep sowing. Deep sowing reduced grain yield across all genotypes compared with shallow sowing. However, genotypes with longer coleoptiles showed improved crop establishment and, in most cases, produced higher yields under deep sowing, highlighting their potential value in mitigating yield loss under suboptimal sowing conditions.
Data availability
Data from this research are published as main or supplementary illustrations. However, raw data are available on request to the corresponding author.
Declaration of funding
This research was partially funded by the Graham Centre for Agricultural Innovation and Australian Grain Technologies.
Author contributions
G. Rebetzke, J. Bathgate and R. Eastwood conceived and designed the experiments. J. Bathgate and R. Eastwood performed the experiments. J. Bathgate and G. Rebetzke analysed the data. R. Eastwood, S. Moroni, G. Rebetzke, J. Bathgate and F. Harris contributed materials/analysis tools. J. Bathgate, G. Rebetzke, S. Moroni, F. Harris and R. Eastwood wrote the paper.
Acknowledgements
We express our gratitude for the dedicated technical support provided by staff and students at Charles Sturt University. Special thanks go to Jack Maloney, Patrick Hawkins, Hoang Han Nguyen, and Vincent West. Additionally, we thank Grace Bathgate, the spouse of the corresponding author, for her support of the project. Our thanks also go to Bernie Dominiak and Andrew Carmichael for reviewing a pre-submission version of the manuscript.
References
Allan RE (1980) Influence of semidwarfism and genetic background on stand establishment of wheat. Crop Science 20(5), 634-638.
| Crossref | Google Scholar |
Allan RE, Vogel OA, Burleigh JR (1962) Length and estimated number of coleoptile parenchyma cells of six wheat selections grown at two temperatures. Crop Science 2(6), 522-524.
| Crossref | Google Scholar |
Anderson WK (1986) Some relationships between plant population, yield components and grain yield of wheat in a Mediterranean environment. Australian Journal of Agricultural Research 37(3), 219-233.
| Crossref | Google Scholar |
Anderson WK, Smith WR (1990) Yield advantage of two semi-dwarf compared with two tall wheats depends on sowing time. Australian Journal of Agricultural Research 41(5), 811-826.
| Crossref | Google Scholar |
Bhatt GM, Qualset CO (1976) Genotype–environment interactions in wheat: effects of temperature on coleoptile length. Experimental Agriculture 12(1), 17-22.
| Crossref | Google Scholar |
Borrill P, Mago R, Xu T, Ford B, Williams SJ, Derkx A, Bovill WD, Hyles J, Bhatt D, Xia X, MacMillan C, White R, Buss W, Molnár I, Walkowiak S, Olsen O-A, Doležel J, Pozniak CJ, Spielmeyer W (2022) An autoactive NB-LRR gene causes Rht13 dwarfism in wheat. Proceedings of the National Academy of Sciences 119(48), e2209875119.
| Crossref | Google Scholar |
Bovill WD, Hyles J, Zwart AB, Ford BA, Perera G, Phongkham T, Brooks BJ, Rebetzke GJ, Hayden MJ, Hunt JR, Spielmeyer W (2019) Increase in coleoptile length and establishment by Lcol-A1, a genetic locus with major effect in wheat. BMC Plant Biology 19(1), 332.
| Crossref | Google Scholar |
Ellis M, Spielmeyer W, Gale K, Rebetzke G, Richards R (2002) ‘Perfect’ markers for the Rht-B1b and Rht-D1b dwarfing genes in wheat. Theoretical and Applied Genetics 105, 1038-1042.
| Crossref | Google Scholar | PubMed |
Fletcher A, Lawes R, Weeks C (2016) Crop area increases drive earlier and dry sowing in Western Australia: implications for farming systems. Crop & Pasture Science 67(12), 1268-1280.
| Crossref | Google Scholar |
Flohr BM, Ouzman J, McBeath TM, Rebetzke GJ, Kirkegaard JA, Llewellyn RS (2021) Redefining the link between rainfall and crop establishment in dryland cropping systems. Agricultural Systems 190, 103-105.
| Crossref | Google Scholar |
Ford BA, Foo E, Sharwood R, Karafiatova M, Vrána J, MacMillan C, Nichols DS, Steuernagel B, Uauy C, Doležel J, Chandler PM, Spielmeyer W (2018) Rht18 semidwarfism in wheat is due to increased GA 2-oxidaseA9 expression and reduced GA content. Plant Physiology 177(1), 168-180.
| Crossref | Google Scholar | PubMed |
Gan Y, Stobbe EH, Moes J (1992) Relative date of wheat seedling emergence and its impact on grain yield. Crop Science 32(5), 1275-1281.
| Crossref | Google Scholar |
Gilmour AR, Cullis BR, Verbyla AP (1997) Accounting for natural and extraneous variation in the analysis of field experiments. Journal of Agricultural, Biological, and Environmental Statistics 2, 269-293.
| Crossref | Google Scholar |
Gooding S, Botwright Acuña TL, Fox PN, Wade LJ (2006) Emergence, stand establishment and vigour of deep-sown Australian and CIMMYT wheats. Australian Journal of Experimental Agriculture 46(9), 1167-1175.
| Crossref | Google Scholar |
GraphPad Prism (2020) ‘GraphPad Prism for Windows.’ Version 10.6.0. (GraphPad Software: Boston, MA, USA) Available at https://www.graphpad.com/
Hunt JR, Hayman PT, Richards RA, Passioura JB (2018) Opportunities to reduce heat damage in rain-fed wheat crops based on plant breeding and agronomic management. Field Crops Research 224, 126-138.
| Crossref | Google Scholar |
Kirkegaard JA, Hunt JR, McBeath TM, Lilley JM, Moore A, Verburg K, Robertson M, Oliver Y, Ward PR, Milroy S, Whitbread AM (2014) Improving water productivity in the Australian Grains industry – a nationally coordinated approach. Crop & Pasture Science 65(7), 583-601.
| Crossref | Google Scholar |
Lemerle D, Cousens RD, Gill GS, Peltzer SJ, Moerkerk M, Murphy CE, Collins D, Cullis BR (2004) Reliability of higher seeding rates of wheat for increased competitiveness with weeds in low rainfall environments. The Journal of Agricultural Science 142(4), 395-409.
| Crossref | Google Scholar |
Lutcher LK, Wuest SB, Johlke TR (2019) First leaf emergence force of three deep-planted winter wheat cultivars. Crop Science 59(2), 772-777.
| Crossref | Google Scholar |
Mahdi L, Bell CJ, Ryan J (1998) Establishment and yield of wheat (Triticum turgidum L.) after early sowing at various depths in a semi-arid Mediterranean environment. Field Crops Research 58(3), 187-196.
| Crossref | Google Scholar |
Matsui T, Inanaga S, Shimotashior T, An P, Sugimoto Y (2002) Morphological characters related to varietal differences in tolerance to deep sowing in wheat. Plant Production Science 5(2), 169-174.
| Crossref | Google Scholar |
Miralles DJ, Slafer GA (1991) A simple model for non-destructive estimates of leaf area in wheat. Cereal Research Communications 19, 439-444.
| Google Scholar |
Mohan A, Schillinger WF, Gill KS (2013) Wheat seedling emergence from deep planting depths and its relationship with coleoptile length. PLoS ONE 8(9), e73314.
| Crossref | Google Scholar |
R Core Team (2020) ‘R: a language and environment for statistical computing.’ (R Foundation for Statistical Computing: Vienna, Austria) Available at https://cran.r-project.org/
Radford BJ, Strong WM, Wilderminth GB (1989) Effects of urea and flutriafol on germination, coleptile length and establishment of wheat and barley. Australian Journal of Experimental Agriculture 29(4), 551-557.
| Crossref | Google Scholar |
Rebetzke GJ, Richards RA, Sirault XRR, Morrison AD (2004) Genetic analysis of coleoptile length and diameter in wheat. Australian Journal of Agricultural Research 55(7), 733-743.
| Crossref | Google Scholar |
Rebetzke GJ, Richards RA, Fettell NA, Long M, Condon AG, Forrester RI, Botwright TL (2007) Genotypic increases in coleoptile length improves stand establishment, vigour and grain yield of deep-sown wheat. Field Crops Research 100(1), 10-23.
| Crossref | Google Scholar |
Rebetzke GJ, Ellis MH, Bonnett DG, Condon AG, Falk D, Richards RA (2011) The Rht13 dwarfing gene reduces peduncle length and plant height to increase grain number and yield of wheat. Field Crops Research 124(3), 323-331.
| Crossref | Google Scholar |
Rebetzke GJ, Zheng B, Chapman SC (2016) Do wheat breeders have suitable genetic variation to overcome short coleoptiles and poor establishment in the warmer soils of future climates? Functional Plant Biology 43(10), 961-972.
| Crossref | Google Scholar | PubMed |
Rebetzke GJ, Rattey AR, Bovill WD, Richards RA, Brooks BJ, Ellis M (2022) Agronomic assessment of the durum Rht18 dwarfing gene in bread wheat. Crop & Pasture Science 73(4), 325-336.
| Crossref | Google Scholar |
Richards RA, Lukacs Z (2002) Seedling vigour in wheat-sources of variation for genetic and agronomic improvement. Australian Journal of Agricultural Research 53(1), 41-50.
| Crossref | Google Scholar |
Schillinger WF, Donaldson E, Allan RE, Jones SS (1998) Winter wheat seedling emergence from deep sowing depths. Agronomy Journal 90(5), 582-586.
| Crossref | Google Scholar |
Tang T, Botwright Acuña T, Spielmeyer W, Richards RA (2021) Effect of gibberellin-sensitive Rht18 and gibberellin-insensitive Rht-D1b dwarfing genes on vegetative and reproductive growth in bread wheat. Journal of Experimental Botany 72(2), 445-458.
| Crossref | Google Scholar | PubMed |
Tipton JL (1984) Evaluation of three growth curve models for germination data analysis. Journal of the American Society for Horticultural Science 109(4), 451-454.
| Crossref | Google Scholar |
VSN International (2020) ‘Genstat for Windows.’ 21st edn. (VSN International: Hemel Hempstead, UK) Available at Genstat.co.uk
Wang Y, Chen L, Du Y, Yang Z, Condon AG, Hu Y-G (2014) Genetic effect of dwarfing gene Rht13 compared with Rht-D1b on plant height and some agronomic traits in common wheat (Triticum aestivum L.). Field Crops Research 162, 39-47.
| Crossref | Google Scholar |
Wildermuth GB, McNamara RB, Quick JS (2001) Crown depth and susceptibility to crown rot in wheat. Euphytica 122(2), 397-405.
| Crossref | Google Scholar |
Zhao Z, Wang E, Kirkegaard JA, Rebetzke GJ (2022) Novel wheat varieties facilitate deep sowing to beat the heat of changing climates. Nature Climate Change 12(3), 291-296.
| Crossref | Google Scholar |


