Does the temporal variation of leaf terpene and moisture content trigger leaf flammability over time?
Anne Ganteaume A * , Bastien Romero A , Elena Ormeño B , Caroline Lecareux B and Catherine Fernandez BA
B
Abstract
It is widely assumed that plant flammability in the Mediterranean region peaks during the summer fire season. We currently lack data that could evaluate these assumptions and have not assessed the mechanisms, e.g. fuel moisture content (FMC) or terpenes, that might drive these patterns.
To determine the mechanistic drivers of species flammability, we used leaf burning experiments coupled with foliar chemical analyses focusing on Aleppo pine (Pinus halepensis) and three introduced cypresses commonly found at the wildland–urban interface (WUI) in southeastern France.
Terpenes, FMC and flammability varied over time and across the species studied, with contrasting patterns for each. Rare correlations between FMC and flammability occurred, in only one season and differing among species, while correlations between flammability and terpene compounds were diverse. The best flammability drivers were terpenes (mainly diterpenes), often changing among and within seasons, and their effect on flammability also differed. Overall, FMC was not a significant explanatory parameter of leaf flammability.
Highlighting the temporal variation between flammability and its drivers revealed that species flammability could also be enhanced by terpenes outside the fire season; this should be accounted for in fire prevention, especially at the WUI.
Keywords: flammability drivers, fuel moisture content, leaf flammability, leaf terpene content, Mediterranean region, Pinus halepensis, terpene temporal variation, wildland–urban interface vegetation.
Introduction
The concept of flammability has been widely studied and experimentally assessed under laboratory conditions as well as in situ, during prescribed fires, for instance. Very often, laboratory methods focus on deriving the metrics of the four components of flammability as described by Anderson (1970), i.e. ignitability, sustainability, combustibility, and Martin et al. (1993), i.e. consumability, typically via the study of individual species fuels (leaf, litter bed, shoot, etc.). For a given fuel scale, these flammability dimensions are not necessarily correlated (de Magalhães and Schwilk 2012; Engber and Varner 2012; Pausas and Moreira 2012; Cornwell et al. 2015) and each of them has relevance across fuel scales (Pausas and Moreira 2012). Previous studies showed that the components of flammability are primarily controlled by different plant traits (Scarff and Westoby 2006; Schwilk and Caprio 2011; Engber and Varner 2012; Clarke et al. 2014), including specific leaf area, surface area-to-volume ratio, thickness and moisture content (e.g. White and Zipperer 2010; Marino et al. 2011; Madrigal et al. 2013; Murray et al. 2013; Santoni et al. 2014; Grootemaat et al. 2015; Ormeño et al. 2020; Ganteaume et al. 2021; Popović et al. 2021). However, when multiple traits are taken into account, the importance of a given driver (significant when considered in isolation) may be surpassed by other traits, as was the case for leaf surface-to-volume ratio in Ganteaume (2018) or fuel moisture content (FMC) in Cawson et al. (2023). However, the latter trait has been widely recognised as a key driver of flammability (Varner et al. 2015) and fire behaviour (e.g. Rothermel 1972). Besides the obvious role of FMC in mitigating flammability by slowing down heat transmission to plant tissues (Pausas et al. 2016), this parameter can also interact with leaf-contained terpenes (Peñuelas and Llusià 1997; Alessio et al. 2008a, De Lillis et al. 2009) and the significant role of FMC on flammability has sometimes been mitigated by these compounds (Della Rocca et al. 2017; Ganteaume et al. 2021).
Mediterranean regions are characterised by climatic conditions conducive to fire and their vegetation is often composed of highly flammable species, therefore making it important to improve plant flammability knowledge. Most plants in the Mediterranean basin are known to synthesise and to emit volatile (the least complex compounds with lower molecular mass such as mono- or sesquiterpenes) or semi-volatile (heavier compounds such as diterpenes) terpenes (e.g. Llusià and Peñuelas 2000). The amount and composition of terpenes produced against a stressor (such as drought, herbivory) can be constrained by the plant’s physiological status (Sampedro et al. 2011) and its genetics (Pausas et al. 2016), but also by the nature and severity of and time since the stress (Page et al. 2012). The positive impact of terpenes on plant flammability could be an issue in Mediterranean areas where summer drought induces water stress that forces some species to increase their terpene production and possibly storage (Llusià and Peñuelas 1998; Blanch et al. 2009; Marchese et al. 2010), thereby making them potentially more flammable. Moreover, as large amounts of these compounds can be emitted in response to high temperatures (Centritto et al. 2011), episodes of massive terpene emission can result from wildfire events (Chetehouna et al. 2009; Courty et al. 2012). Given that most of these compounds can easily ignite in the presence of a heat source (owing to their low flash and boiling points), they could thereby enhance plant flammability, possibly affecting plant fire behaviour (White 1994; Cornelissen et al. 2003; Keeley et al. 2012). Terpenes have also been found to affect litter flammability (Ormeño et al. 2009), their content (and composition) hardly varying from green leaf to litter (Romero et al. 2019). Leaf flammability is often positively correlated with both monoterpene content (White 1994; Owens et al. 1998; Pausas et al. 2016) and sesquiterpene content (Guerrero et al. 2022), in contrast to diterpenes because of their higher complexity (Ganteaume et al. 2021). However, previous research indicates that terpenes can have complex and sometimes contradictory influences on flammability (Cappelli et al. 1983; Alessio et al. 2008b; Romero et al. 2019). Indeed, terpene content can also have antagonist effects on the components of flammability (Della Rocca et al. 2017; Romero et al. 2019; Ganteaume et al. 2021), highlighting the importance of assessing multiple components of flammability.
Change in plant flammability according to the variation of moisture content throughout the year has been demonstrated in numerous previous studies (e.g. Pellizzaro et al. 2007; Alessio et al. 2008a). However, the possible change in flammability could also be triggered by a temporal change in terpene content. Terpene content is known to vary throughout the year (Peñuelas and Llusià 1997; Llusià and Peñuelas 2000; Llusià et al. 2006; Alessio et al. 2008a) but the link with the temporal variation of flammability remains to be explored. Among the previous studies dealing with the temporal variation in flammability, FMC, or terpene content (Bernard-Degan 1988; Owens et al. 1998; Llusià and Peñuelas 2000; Weise et al. 2005; Pellizzaro et al. 2009), only a few have addressed the interactions between these parameters (Della Rocca et al. 2017; Ganteaume et al. 2021), and without tackling the temporal variation.
Among plant species commonly found at the wildland–urban interface (WUI) of southeastern (SE) France, only a few woody plant species contain terpenes, mostly conifers, including some native species (Romero et al. 2019). For these species, the composition and content of the main subgroups of terpenes (mono-, sesqui- and diterpenes) and of single terpene compounds as well as their impact on flammability were assessed. The effect of FMC and terpene content, combined or alone, on flammability has previously been investigated, pooling observations recorded over multiple seasons within a year (Ganteaume et al. 2021). These authors also showed that flammability differed among terpene-containing species and that each flammability variable was differently impacted by terpenes, but this prior research did not examine the temporal variation of such compounds and FMC and their influence on flammability over time.
The role of terpenes in flammability is therefore becoming even more important, now and in the near future, because their content could possibly increase in response to predicted warmer and drier climatic conditions in the context of the current climate change. Our work, therefore, aimed to explain the possible changes in leaf flammability of four conifer species commonly found at the WUI of SE France in response to changes in foliar moisture content and terpene content over three seasons. To achieve this goal, the specific objectives of the work were to:
characterise and quantify temporal changes in terpene content (at subgroup and compound levels), moisture content of live foliage and leaf flammability within each species. Further, we also checked if the four species used in this work – all conifers, with three species belonging to the same family (i.e. Cupressaceae) – presented the same pattern throughout the year, especially in terms of terpene content;
examine the influence of FMC and terpene content (subgroup and individual compound) on seasonal changes in leaf flammability within each species; and
develop models of leaf flammability by species and by season to determine the relative importance of individual terpene compounds and FMC.
Therefore, in the current work, we evaluated the following hypotheses: (i) flammability, terpene content and FMC temporal variation is species-specific; (ii) the influence of terpene content as well as of FMC on flammability varies among seasons; and (iii) the combined effect of individual terpene compound content and FMC on flammability varies among seasons, with terpenes being the main drivers.
Materials and methods
Species studied and sampling
The species studied in the current work were all conifers (one Pinaceae: Pinus halepensis Mill. 1768 and three Cupressaceae: Cupressus sempervirens L. 1753, Hesperocyparis arizonica Greene 1882 Bartel (Adams et al. 2009), formerly Cupressus arizonica, and Cupressocyparis leylandii A.B. Jacks. and Dallim 1926) that are commonly located within the WUI of the French Mediterranean region. P. halepensis is the only species native to SE France whereas the other species are exotic and used as ornamental vegetation (often in hedges). Indeed, H. arizonica comes from the southwestern USA and C. sempervirens var. horizontalis forms natural forest stands, mostly in the eastern part of the Mediterranean basin (in some parts of Tunisia (var. numidica), Italy and Greece, for instance). The latter species can present two distinct varieties: (i) horizontalis, characterised by a broad pyramidal crown with branches spreading horizontally (constituting the natural stands), and (ii) pyramidalis (or fastigata), characterised by a compact conical crown with a small angle between branches and trunk (Brofas et al. 2006), which is the variety studied in the current work. C. leylandii is an intergenic hybrid of the yellow cedar (Callitropsis nootkatensis D. Don 1824), native to northwestern America, and of the Monterey cypress (Hesperocyparis macrocarpa Hartw. 1847), a species endemic to Monterey Bay in California (USA). All of these species can be involved in fire propagation from wildland vegetation to nearby buildings, especially when they are used in ornamental hedges that provide strong horizontal fuel continuity.
Live leaves of the different species were sampled in Le Tholonet (SE France) where the climate is typically Mediterranean (i.e. mild winters and hot and dry, often windy summer). We focused our sampling on leaves because of their high terpene content and because they represent the primary fuel particle that drives crown fire behaviour. For the three cypress species, we used green branchlets composed of several green scale-leaves as a proxy for leaves, following the protocol developed in previous studies (Ganteaume 2018; Romero et al. 2019; Ganteaume et al. 2013, 2021). Leaf collection was carried out at three different times in 2016 (hereafter named ‘seasons’), corresponding to winter, spring and summer (i.e. between January and February, May and July (Supplementary Table S1)); the exact date within each season varied among species to capture a range of conditions when flammability might be expected to differ and to highlight possible temporal variations of FMC and terpene content linked to flammability. Moreover, the weather conditions being very different among years (which affects plant moisture content), it was not possible to anticipate the driest (or the wettest) period during the year for sampling. The leaves collected had to be healthy, well developed (i.e. mature), sun-exposed and located in the middle of the canopy, excluding newly developed tissue at the tip of the shoots (as the moisture content of these parts is higher). It is worth noting that the plants sampled were located as far as possible from housing (i.e. not in a backyard) in order to avoid any effect of watering or fertilising.
For each species and each season, a maximum of 40 g of mature and healthy (i.e. free of pests) green leaves was collected in the canopy of five different individual plants (i.e. 200 g in total per species and season) located at least 4 m apart. For each plant sampled, 30 g was used for the burning experiments, 5 g for FMC measurements and 0.5 g for terpene analysis. In the case of rain, sampling was conducted at least 48 h following the precipitation event to avoid any impact on FMC (mostly due to external moisture on the leaves). Collected leaves were placed in plastic bags that were stored in a cooler for immediate transportation to the laboratory to minimise the change in water content. The samples were burned the day of return to the laboratory.
Just before the burning experiments, three 5 g-leaf subsamples of each individual were oven-dried for 48 h at 60°C (until achieving constant weight) in order to measure their moisture content (i.e. FMC, calculated on a dry mass basis) at the time of burning. FMC (%) was calculated according to the following equation:
where Mf represents the fresh fuel mass (g) and Md represents the dry fuel mass (g).
Terpene identification and quantification
Leaves sampled for the chemical analyses were stored at −80°C to avoid any metabolic transformation when the analyses could not be performed right after sampling (but without exceeding 2 months). Terpene content was analysed for each season using 0.5 g of leaves collected from the five plants per species as presented in Romero et al. (2019). The low number of samples is consistent with that in previous studies (e.g. Peñuelas and Llusià 1997; Llusià and Peñuelas 2000; Della Rocca et al. 2017). Terpenes were qualitatively and quantitatively analysed using a gas chromatograph coupled to a mass spectrometer (GC-MS, 7890B Agilent Technologies). Leaves were ground using liquid nitrogen and put into a 4 mL vial of extraction solution (cyclohexane and dodecane). This vial was agitated for 30 min, filtered (0.22 μm PTFE filter) and put into a 3 mL vial for analysis. Dodecane was used as an internal standard and was not naturally present in the samples.
A sample of 1 μL was taken from the 3 mL vial (i.e. plant material and extraction solution) and injected into a 30 m × 0.25 mm × 0.25 μm-thickness capillary column (HP-5MS-Agilent J&W GC column) at a constant flow (1 mL min−1) and in splitless mode. The injection temperature was maintained at 250°C, with helium (99.99%) as the carrier gas. The initial temperature was 40°C and was increased at 3°C min−1 up to 300°C during analysis. A 5 min solvent delay was used and the total run time was 90 min. The very high temperatures reached in the GC oven allowed the detection of compounds with high molecular mass, such as diterpenes (as a whole, the chromatographic run applied allowed the detection of molecular hydrocarbons with up to 34 carbon atoms). Terpene identification was achieved based on the molecule retention time (which was compared with that of the pure standard when available) as well as the molecule mass spectrum, which was compared with available libraries (Adams 2007; NIST 2011). To complete this identification, experimental retention indexes were calculated for each molecule identified and compared with the theoretical retention indexes in these libraries. The terpene content of each sample was calculated following the methodology presented in Romero et al. (2019) and is expressed as milligrams per gram of dry matter (DM).
Following the results of a previous study (Ganteaume et al. 2021), the contribution of terpenes was investigated both at the subgroup (comprising monoterpenes, MT; sesquiterpenes, ST; and diterpenes, DT) and the single compound (within each subgroup) levels for each plant species. It is worth noting that, given the specifications of the GC-MS, only some of the diterpene compounds (especially with respect to oxygenated molecules) could be detected; some could not be identified because these molecules are less well-known than mono- and sesquiterpenes.
Flammability experiments
For each species, 30 samples (six trials for each of the five plants) of 1 g of leaf samples (mass required for burning on the epiradiator; see Hernando-Lara 2000) collected in each season were burned using a 500 W epiradiator composed of a 10 cm radiant disc, according to the methodology presented in Romero et al. (2019). The steady-state surface temperature achieved with the device was 420°C. The samples were in direct contact with the electric radiator, and the surface area of contact depended on the species. This surface was assumed to be close enough to the heat source to undergo homogeneous heat transfer effects. A pilot flame, which did not take part in the decomposition of the sample, was located 4 cm above the centre of the disc and allowed more regular ignition of the gases emitted during combustion of the leaf. The device was placed under a hood to prevent air currents from perturbing the convection column and gas plumes. This burning device is adapted for burning fuels at the particle level (leaf, twig, etc.) under laboratory conditions, as already highlighted in several past studies (e.g. Valette 1990; Hernando-Lara 2000; Ormeño et al. 2009; Ganteaume et al. 2013; Ganteaume 2018), given that the goal of the current work was not to mimic what goes on during a real fire, or describe or predict the flammability of fuels under natural and real conditions. Laboratory experiments help improve our knowledge of the effects of live and dead fuel properties (e.g. moisture content and other physical and chemical characteristics) on flammability; in our case, they represent basic information useful for assessing the fire risk of Mediterranean ornamental vegetation commonly planted for landscaping.
The variation in temperature was recorded every second during burning trials using a thermocouple (chromel–alumel, K type, 30 μm diameter) positioned 1 cm above the disc centre. As soon as the fuel was in contact with the epiradiator surface, time and temperature recordings were started. The five flammability variables recorded during the burning experiments encompassed the three flammability components (ignitability, sustainability and combustibility) as described by Anderson (1970): (i) time to ignition, (TTI, s): the time necessary for the fuel to ignite once laid on the radiant disk, and (ii) ignition temperature (Tign, °C): the temperature recorded when the flame appeared, for ignitability; (iii) flaming duration (FD, s): the time elapsed between flame occurrence and its extinction for sustainability; and (iv) the maximum temperature reached during burning (Tmax, °C) for combustibility. These flammability variables were taken into account because previous research has demonstrated that the effect of terpenes and FMC vary among these variables (Ganteaume et al. 2021). It is worth noting that the percentage of consumed biomass (i.e. consumability; Martin et al. 1993) was not accounted for because the leaves completely burned in each trial.
Data analyses
The statistical analyses were performed on each species’ seasonal dataset, taking into account the content of terpenes, assessed at the subgroup and single compound levels, as well as FMC, as explanatory factors of flammability. Separate statistical analyses were performed for each species because they differed in flammability as already shown in previous studies (e.g. Ganteaume 2018; Ganteaume et al. 2021). Although leaf thickness was an important driver of flammability (along with other structural leaf traits such as surface-to-volume ratio), it was not included in the present analyses as most morphological traits are less sensitive to temporal variations than chemical traits (Fajardo and Siefert 2016; Bloomfield et al. 2018). The different flammability variables (using a single mean value per individual of each species) were used as dependent variables. All tests were performed using StatGraphics Centurion XVII X64 software (StatPoint Technologies, Inc.).
First, we performed different variance analyses (one-way ANOVA, significance when P < 0.05) to highlight the effect of season on FMC, terpene content and flammability for each species. In these analyses, the Kruskal–Wallis test was performed instead of the F-test because of the small sample size for each season.
Then, Principal Component Analyses (PCAs) were performed on the four flammability variables (i.e. flaming duration, ignition temperature, time to ignition and maximum temperature) of each species seasonal dataset to determine the number of PCA components explaining most of the variance (>75%) as well as the link between the four variables and these components. From the coordinates of these main components, flammability indexes, accounting for the different variables, could be derived for each species and season. When the first component represented more than 75% of the variation, only one flammability index was used in the following regression analyses.
For each species and season, simple linear regression analyses were performed to highlight any significant positive or negative correlations (P < 0.05) existing between leaf traits (FMC, terpenes) and flammability (i.e. flammability indexes). In these analyses, the adjusted R2 value was used to quantify the amount of variation in flammability that was explained by the model. When several leaf traits were correlated with the flammability indexes and to avoid issues of multicollinearity among independent variables, Partial Least Square (PLS) regression analyses were performed to determine the relative importance of the different leaf traits (first using terpene content only, then adding FMC in the models) on each flammability index for each season (highlighted in the previous PCAs) and species. These analyses allowed the identification of a pool of significant terpene compounds, alone or with FMC, affecting flammability, as well as those that were the best drivers according to the season. The significance of components in the models was determined according to uncertainty tests carried out within one-at-a-time cross-validation. The scaled regression coefficients of the PLS models provided information on the effect (positive or negative) of each parameter on flammability and its relative weight in the fitted model (absolute value) indicated the relative importance in predicting each flammability index (parameters with |value| ≥ 0.2 were considered as significant). Model fitting was evaluated by the R2Y statistic, which is equivalent to the adjusted R2 of parametric methods.
Results
Temporal variation of terpenes, FMC and flammability
In total, 55 different terpene compounds were identified across all species; Cupressocyparis leylandii had the highest terpene diversity (34 compounds) regardless of the season (Table 1). The season showing the highest terpene diversity differed among species: summer for C. leylandii (displaying mostly diterpenes) as well as for Heterocyparis arizonica (displaying mostly sesquiterpenes), and winter and summer for Cupressus sempervirens (displaying mostly diterpenes and sesquiterpenes). For Pinus halepensis, terpene compound diversity was consistent across all three seasons (mostly monoterpenes). Overall, there was low inter-plant variation in the number of compounds (<20%), regardless of the season (except regarding diterpenes for H. arizonica in winter and for P. halepensis in spring and summer). For each species, the composition of extracted compounds as well as the percentage of each subgroup in the total amount of terpenes for each season are presented in Table 2.
| Terpene subgroup; season | Winter | Spring | Summer | |
|---|---|---|---|---|
| Cupressocyparis leylandii | ||||
| MT | 11 | 11 | 11 | |
| ST | 11 | 11 | 11 | |
| DT | 10 | 10 | 12 | |
| Total | 32 | 32 | 34 | |
| Hesperocyparis arizonica | ||||
| MT | 6 | 6 | 6 | |
| ST | 8 | 8 | 9 | |
| DT | 7 | 7 | 7 | |
| Total | 21 | 21 | 22 | |
| Cupressus sempervirens | ||||
| MT | 6 | 5 | 6 | |
| ST | 8 | 6 | 6 | |
| DT | 7 | 5 | 8 | |
| Total | 21 | 16 | 20 | |
| Pinus halepensis | ||||
| MT | 7 | 7 | 7 | |
| ST | 6 | 6 | 6 | |
| DT | 5 | 5 | 4 | |
| Total | 18 | 18 | 17 | |
MT, monoterpenes; ST, sesquiterpenes; DT, diterpenes.
| Cupressocyparis leylandii | Winter | Spring | Summer | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | s.d. | % | Mean | s.d. | % | Mean | s.d. | % | ||
| Monoterpenes | ||||||||||
| δ-3-carene | 0.37 | 0.16 | 9.44 | 0.39 | 0.25 | 11.19 | 0.42 | 0.11 | 9.39 | |
| α-pinene | 0.44 | 0.18 | 11.17 | 0.34 | 0.22 | 9.73 | 0.48 | 0.18 | 10.80 | |
| α-thujene | 0.07 | 0.03 | 1.68 | 0.08 | 0.06 | 2.36 | 0.08 | 0.02 | 1.89 | |
| β-pinene | 0.39 | 0.18 | 9.87 | 0.42 | 0.31 | 11.99 | 0.60 | 0.19 | 13.49 | |
| Fenchene | 0.04 | 0.01 | 1.09 | 0.06 | 0.04 | 1.83 | 0.05 | 0.01 | 1.14 | |
| ϒ-terpinene | 0.04 | 0.02 | 0.94 | 0.04 | 0.03 | 1.19 | 0.04 | 0.02 | 0.96 | |
| Limonene | 0.29 | 0.21 | 7.28 | 0.26 | 0.14 | 7.50 | 0.27 | 0.13 | 5.96 | |
| Myrcene | 0.20 | 0.05 | 4.98 | 0.27 | 0.15 | 7.78 | 0.21 | 0.01 | 4.77 | |
| Sabinene hydrate | 0.18 | 0.06 | 4.55 | 0.22 | 0.15 | 6.31 | 0.16 | 0.03 | 3.57 | |
| Terpinen-4-ol | 0.17 | 0.03 | 4.31 | 0.20 | 0.12 | 5.71 | 0.18 | 0.04 | 3.98 | |
| Terpinolene | 0.04 | 0.01 | 1.02 | 0.05 | 0.03 | 1.42 | 0.05 | 0.01 | 1.07 | |
| Sesquiterpenes | ||||||||||
| α-muurolene | 0.02 | 0.01 | 0.42 | 0.01 | 0.01 | 0.25 | 0.01 | 0.00 | 0.31 | |
| α bisabolol | 0.01 | 0.01 | 0.36 | 0.03 | 0.02 | 0.80 | 0.01 | 0.00 | 0.23 | |
| β-elemene | 0.02 | 0.00 | 0.57 | 0.03 | 0.02 | 0.88 | 0.06 | 0.02 | 1.43 | |
| Cadina-1(6),4-diene <cis> | 0.26 | 0.11 | 6.68 | 0.27 | 0.17 | 7.61 | 0.36 | 0.11 | 7.98 | |
| β-caryophyllene | 0.01 | 0.00 | 0.19 | 0.01 | 0.01 | 0.26 | 0.01 | 0.00 | 0.21 | |
| Cedrol | 0.01 | 0.00 | 0.17 | 0.01 | 0.00 | 0.15 | 0.01 | 0.01 | 0.21 | |
| Copaene | 0.01 | 0.00 | 0.24 | 0.01 | 0.01 | 0.33 | 0.01 | 0.00 | 0.20 | |
| Curcumen-12-ol <beta-(Z)> | 0.05 | 0.02 | 1.25 | 0.08 | 0.05 | 2.40 | 0.03 | 0.02 | 0.61 | |
| Elemol | 0.02 | 0.01 | 0.50 | 0.02 | 0.02 | 0.69 | 0.04 | 0.01 | 0.80 | |
| ϒ-elemene | 0.01 | 0.01 | 0.28 | 0.02 | 0.01 | 0.45 | 0.02 | 0.00 | 0.39 | |
| Nerodiol | 0.02 | 0.01 | 0.53 | 0.02 | 0.02 | 0.49 | 0.04 | 0.01 | 0.80 | |
| Diterpenes | ||||||||||
| Abietal-4-epi | – | – | – | – | – | – | 0.35 | 0.09 | 7.73 | |
| Abietatriene | 0.04 | 0.01 | 0.88 | 0.02 | 0.01 | 0.68 | 0.03 | 0.00 | 0.71 | |
| Cembrene | 0.29 | 0.08 | 7.28 | 0.07 | 0.04 | 2.06 | 0.14 | 0.03 | 3.14 | |
| Cembrene A | – | – | – | – | – | – | 0.14 | 0.03 | 3.16 | |
| Ferruginol (trans) | 0.02 | 0.01 | 0.61 | 0.01 | 0.01 | 0.26 | 0.02 | 0.01 | 0.45 | |
| Isophyllocladene | 0.10 | 0.04 | 2.63 | 0.03 | 0.02 | 0.95 | 0.06 | 0.01 | 1.36 | |
| Isopimara-9(11),15-diene | 0.02 | 0.01 | 0.39 | 0.01 | 0.00 | 0.16 | 0.01 | 0.00 | 0.14 | |
| Manool oxide | 0.13 | 0.04 | 3.24 | 0.05 | 0.03 | 1.51 | 0.07 | 0.02 | 1.67 | |
| Manool oxide <13-epi> | 0.11 | 0.04 | 2.66 | 0.01 | 0.01 | 0.39 | 0.05 | 0.01 | 1.04 | |
| Nezukol | 0.25 | 0.07 | 6.24 | 0.23 | 0.17 | 6.54 | 0.19 | 0.05 | 4.19 | |
| Sempervirol | 0.08 | 0.02 | 1.95 | 0.03 | 0.02 | 0.98 | 0.06 | 0.01 | 1.34 | |
| Totarol (trans) | 0.26 | 0.08 | 6.60 | 0.18 | 0.10 | 5.15 | 0.22 | 0.03 | 4.90 | |
| Hesperocyparis arizonica | Winter | Spring | Summer | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | s.d. | % | Mean | s.d. | % | Mean | s.d. | % | ||
| Monoterpenes | ||||||||||
| δ-3-carene | 0.02 | 0.03 | 1.92 | 0.01 | 0.02 | 1.31 | 0.01 | 0.01 | 0.99 | |
| α-pinene | 0.06 | 0.05 | 7.04 | 0.14 | 0.05 | 12.63 | 0.10 | 0.03 | 10.05 | |
| β-pinene | 0.02 | 0.02 | 2.68 | 0.06 | 0.04 | 5.10 | 0.04 | 0.03 | 4.05 | |
| Limonene | 0.08 | 0.11 | 9.06 | 0.13 | 0.13 | 12.07 | 0.07 | 0.04 | 6.82 | |
| Sabinene hydrate | 0.01 | 0.01 | 1.49 | 0.02 | 0.01 | 1.66 | 0.02 | 0.02 | 2.28 | |
| Terpinen-4-ol | 0.04 | 0.01 | 4.17 | 0.07 | 0.03 | 6.50 | 0.07 | 0.02 | 7.21 | |
| Sesquiterpenes | ||||||||||
| α-aconerol | 0.04 | 0.01 | 3.94 | 0.06 | 0.03 | 5.50 | 0.06 | 0.04 | 6.38 | |
| α-muurolene | 0.03 | 0.01 | 2.84 | 0.02 | 0.01 | 1.73 | 0.02 | 0.01 | 2.36 | |
| Aromadendrene | 0.13 | 0.07 | 14.17 | 0.11 | 0.04 | 9.79 | 0.09 | 0.03 | 9.85 | |
| Cadina-1(6),4-diene (cis) | 0.36 | 0.20 | 39.68 | 0.29 | 0.12 | 26.77 | 0.20 | 0.07 | 20.46 | |
| Cadinol | 0.01 | 0.00 | 1.14 | 0.02 | 0.01 | 1.82 | 0.01 | 0.01 | 1.52 | |
| Calamene | 0.02 | 0.01 | 2.10 | 0.01 | 0.01 | 1.24 | 0.02 | 0.01 | 2.35 | |
| Cedrol | 0.01 | 0.01 | 1.30 | 0.02 | 0.01 | 1.39 | 0.02 | 0.01 | 1.58 | |
| Cubebol <10-epi> | 0.02 | 0.01 | 1.92 | 0.03 | 0.02 | 3.16 | – | – | – | |
| Muurol-5-en-4-one | – | – | – | – | – | – | 0.06 | 0.04 | 6.40 | |
| Muurol-5-en-beta-ol (cis) | – | – | – | – | – | – | 0.08 | 0.04 | 8.28 | |
| Diterpenes | ||||||||||
| Abietadiene (trans) | 0.01 | 0.01 | 1.03 | 0.01 | 0.01 | 1.13 | 0.01 | 0.00 | 1.08 | |
| Abietal <4-epi> | 0.01 | 0.02 | 1.57 | 0.02 | 0.01 | 1.67 | 0.01 | 0.00 | 1.22 | |
| Abietol | 0.01 | 0.01 | 0.60 | 0.02 | 0.03 | 2.12 | 0.01 | 0.01 | 1.12 | |
| Ferruginol (trans) | 0.01 | 0.01 | 0.65 | 0.01 | 0.01 | 1.35 | 0.01 | 0.01 | 1.00 | |
| Nezukol | 0.01 | 0.00 | 0.66 | 0.01 | 0.01 | 0.80 | 0.01 | 0.01 | 1.44 | |
| Sempervirol | 0.01 | 0.01 | 1.22 | 0.01 | 0.01 | 1.33 | 0.02 | 0.02 | 2.26 | |
| Totarol (trans) | 0.01 | 0.00 | 0.80 | 0.01 | 0.00 | 0.93 | 0.01 | 0.00 | 1.30 | |
| Cupressus sempervirens | Winter | Spring | Summer | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | s.d. | % | Mean | s.d. | % | Mean | s.d. | % | ||
| Monoterpenes | ||||||||||
| δ-3-carene | 0.10 | 0.04 | 6.79 | 0.11 | 0.09 | 8.60 | 0.14 | 0.06 | 10.97 | |
| α-pinene | 0.39 | 0.19 | 26.15 | 0.26 | 0.16 | 19.66 | 0.29 | 0.13 | 22.06 | |
| α-thujene | 0.01 | 0.02 | 0.95 | 0.01 | 0.02 | 0.84 | 0.02 | 0.02 | 1.28 | |
| β-pinene | 0.02 | 0.01 | 1.28 | 0.03 | 0.02 | 2.35 | 0.03 | 0.02 | 2.63 | |
| Limonene | 0.01 | 0.00 | 0.82 | 0.01 | 0.01 | 0.58 | 0.02 | 0.00 | 1.17 | |
| Terpinolene | 0.00 | 0.00 | 0.33 | – | – | – | 0.02 | 0.01 | 1.31 | |
| Sesquiterpenes | ||||||||||
| α-humulene | 0.01 | 0.01 | 0.81 | 0.02 | 0.01 | 1.23 | 0.01 | 0.00 | 0.73 | |
| Aromadendrene | 0.01 | 0.01 | 0.82 | – | – | – | 0.02 | 0.01 | 1.47 | |
| β-ylangene | 0.03 | 0.01 | 1.75 | 0.02 | 0.02 | 1.68 | 0.02 | 0.00 | 1.25 | |
| Cadina-1(6),4-diene (cis) | 0.01 | 0.00 | 0.44 | 0.01 | 0.00 | 0.71 | – | – | – | |
| Cadinene | 0.01 | 0.00 | 0.68 | – | – | – | – | – | – | |
| β-caryophyllene | 0.02 | 0.01 | 1.35 | – | – | – | 0.01 | 0.00 | 1.00 | |
| Cedrol | 0.04 | 0.04 | 2.87 | 0.04 | 0.05 | 3.17 | 0.03 | 0.03 | 2.30 | |
| Ferruginol (trans) | – | – | – | 0.05 | 0.02 | 4.07 | – | – | – | |
| ϒ-muurolene | 0.12 | 0.04 | 7.88 | 0.16 | 0.08 | 12.10 | 0.14 | 0.05 | 10.35 | |
| Diterpenes | ||||||||||
| Abienol | 0.04 | 0.02 | 2.76 | 0.05 | 0.01 | 3.57 | 0.02 | 0.01 | 1.86 | |
| Abietadiene(trans) | 0.04 | 0.04 | 2.83 | 0.06 | 0.03 | 4.31 | 0.03 | 0.01 | 2.29 | |
| Manool oxide <13-epi> | – | – | – | – | – | – | 0.05 | 0.02 | 3.71 | |
| Sandaracopimarinal | 0.05 | 0.03 | 3.17 | 0.05 | 0.03 | 3.62 | 0.05 | 0.02 | 3.64 | |
| Sandaracopimarinol | 0.03 | 0.01 | 1.94 | 0.03 | 0.01 | 2.52 | 0.03 | 0.01 | 2.15 | |
| Sclarene | 0.04 | 0.05 | 2.48 | 0.03 | 0.03 | 2.10 | 0.03 | 0.03 | 2.07 | |
| Thunbergol | 0.03 | 0.02 | 2.25 | – | – | – | 0.02 | 0.02 | 1.56 | |
| Totarol (trans) | 0.48 | 0.17 | 31.67 | 0.38 | 0.14 | 28.88 | 0.34 | 0.10 | 26.20 | |
| Pinus halepensis | Winter | Spring | Summer | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | s.d. | % | Mean | s.d. | % | Mean | s.d. | % | ||
| Monoterpenes | ||||||||||
| δ-3-carene | 0.01 | 0.00 | 0.22 | 0.01 | 0.01 | 0.63 | 0.02 | 0.02 | 0.67 | |
| α-pinene | 0.13 | 0.03 | 5.31 | 0.15 | 0.08 | 7.03 | 0.22 | 0.10 | 8.98 | |
| α-thujene | 0.01 | 0.00 | 0.36 | 0.03 | 0.04 | 1.24 | 0.03 | 0.03 | 1.27 | |
| β-pinene | 0.04 | 0.01 | 1.89 | 0.11 | 0.11 | 5.48 | 0.15 | 0.11 | 6.17 | |
| Limonene | 0.01 | 0.00 | 0.51 | 0.02 | 0.02 | 1.06 | 0.03 | 0.01 | 1.05 | |
| Myrcene | 0.06 | 0.05 | 2.75 | 0.20 | 0.23 | 9.40 | 0.46 | 0.51 | 18.70 | |
| Terpinolene | 0.02 | 0.01 | 0.83 | 0.08 | 0.06 | 3.88 | 0.10 | 0.08 | 3.94 | |
| Sesquiterpenes | ||||||||||
| α-humulene | 0.16 | 0.03 | 6.76 | 0.18 | 0.07 | 8.61 | 0.14 | 0.04 | 5.91 | |
| Cadinene | 0.01 | 0.00 | 0.58 | 0.02 | 0.01 | 1.07 | 0.02 | 0.01 | 0.83 | |
| β-caryophyllene | 1.22 | 0.25 | 51.77 | 0.98 | 0.36 | 46.96 | 0.91 | 0.25 | 36.92 | |
| Cubebol | 0.02 | 0.00 | 0.89 | 0.05 | 0.01 | 2.29 | 0.03 | 0.01 | 1.19 | |
| Elemol | 0.02 | 0.00 | 0.84 | 0.04 | 0.01 | 1.70 | 0.01 | 0.00 | 0.56 | |
| Germacrene D | 0.01 | 0.00 | 0.50 | 0.04 | 0.02 | 2.15 | 0.08 | 0.14 | 3.42 | |
| Diterpenes | ||||||||||
| Cembrene | 0.34 | 0.15 | 14.62 | 0.07 | 0.05 | 3.29 | – | – | – | |
| Cembrene A | 0.03 | 0.01 | 1.41 | 0.01 | 0.01 | 0.67 | 0.02 | 0.01 | 1.00 | |
| Manool oxide | 0.04 | 0.01 | 1.56 | 0.01 | 0.03 | 0.67 | 0.04 | 0.03 | 1.46 | |
| Manool oxide <13-epi> | 0.16 | 0.20 | 7.00 | 0.05 | 0.06 | 2.37 | 0.16 | 0.14 | 6.67 | |
| Sclarene | 0.05 | 0.02 | 2.21 | 0.03 | 0.01 | 1.51 | 0.03 | 0.02 | 1.26 | |
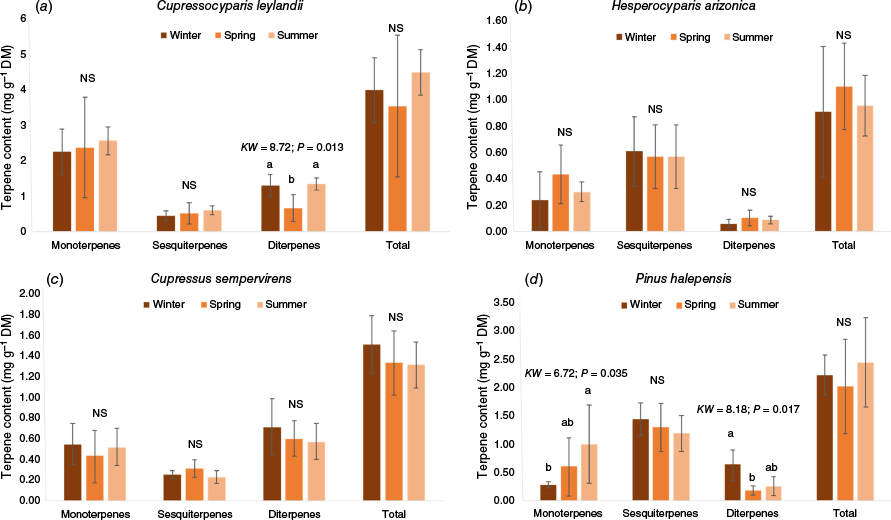
Regardless of the species, the annual total terpene content did not vary with season, but the species studied presented different temporal patterns for the different terpene subgroups (Fig. 1). For C. leylandii (Fig. 1a), only diterpene content varied significantly among seasons, the content being lower in spring than in the other seasons (95% increase from spring to winter and 101% from spring to summer). Monoterpenes presented the highest terpene content throughout the year. For P. halepensis (Fig. 1d), mono- and diterpene content varied among seasons but showed different temporal patterns: monoterpenes were more concentrated in summer (67% increase from winter to summer and 257% from spring to summer) whereas diterpenes showed higher values in winter (142% increase from summer to winter and 250% from spring to winter). Sesquiterpene showed the highest content throughout the year, mostly owing to β-caryophyllene (on average, 45% of the total content). The terpene content of H. arizonica and C. sempervirens did not significantly vary among seasons, regardless of the subgroup (Fig. 1b, c).
Variation in the terpene content (subgroups and total) throughout the year for the four species studied: (a) Cupressocyparis leylandii, (b) Hesperocyparis arizonica, (c) Cupressus sempervirens, (d) Pinus halepensis (NS, non-significant; when significant, the results of the tests are shown: KW, Kruskal–Wallis test; P, probability). Lowercase letters indicate significant differences among seasons for each terpene subgroup with a > b and error bars represent standard deviations. Values shown refer to the average for n = 6.
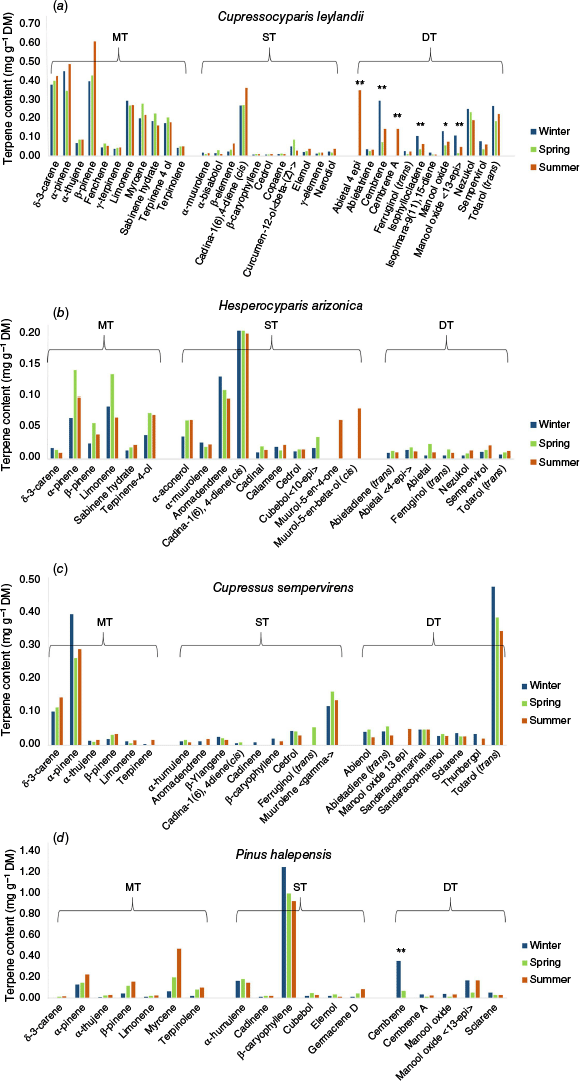
Seasonal patterns of terpene content at the compound level were consistent with terpene subgroup patterns among species (Fig. 2). For C. leylandii, only the content of six diterpene compounds differed among seasons, all of them main compounds (content ≥0.10 mg g−1 DM) of this subgroup in, at least, one season (e.g. cembrene being a major compound in winter and summer). Most diterpene compounds had their highest concentrations during winter. It is worth noting that, overall, the most concentrated compounds (i.e. monoterpenes) did not vary throughout the year. For P. halepensis, only the content of the diterpene cembrene varied among seasons and this compound was not identified in summer. As for C. leylandii, the most concentrated compound (sesquiterpene β-caryophyllene for P. halepensis) did not show any temporal variation. According to the results obtained at the subgroup level, the content of each terpene did not vary throughout the year for H. arizonica and C. sempervirens, even if, for these species, some compounds were not present all year round (therefore presenting marked differences throughout the year).
Temporal variation in single terpene compound content for the four species studied: (a) Cupressocyparis leylandii, (b) Hesperocyparis arizonica, (c) Cupressus sempervirens, (d) Pinus halepensis (Kruskal–Wallis test: *: P < 0.05, **: P < 0.01). Compounds within a terpene subgroup (MT, monoterpenes; ST, sesquiterpenes; DT, diterpenes) are shown in alphabetical order. Error bars are not shown for clarity’s sake (see Supplementary Table S2 for standard deviations).
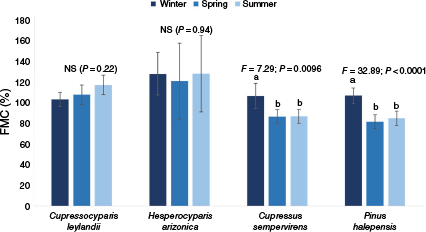
The leaf moisture content of the different conifer species ranged from 91.41% (±13.72) (P. halepensis) to 125.95% (±4.03) (H. arizonica). Except for H. arizonica (which showed the highest values regardless of season) and C. leylandii, FMC varied with season. Indeed, for C. sempervirens and P. halepensis, values of moisture content were higher in winter than in spring and summer (23% increase from both summer and spring to winter for C. sempervirens as well as 31 and 26% increase, respectively, for P. halepensis) (Fig. 3).
Temporal variation in the leaf moisture content (FMC) for the four species studied (when significant, the results of the test are shown: KW, Kruskal–Wallis test; P, probability; NS, not significant). Lowercase letters indicate significant differences among seasons with a > b. Bars represent the average for N = 5 and error bars represent standard deviations.
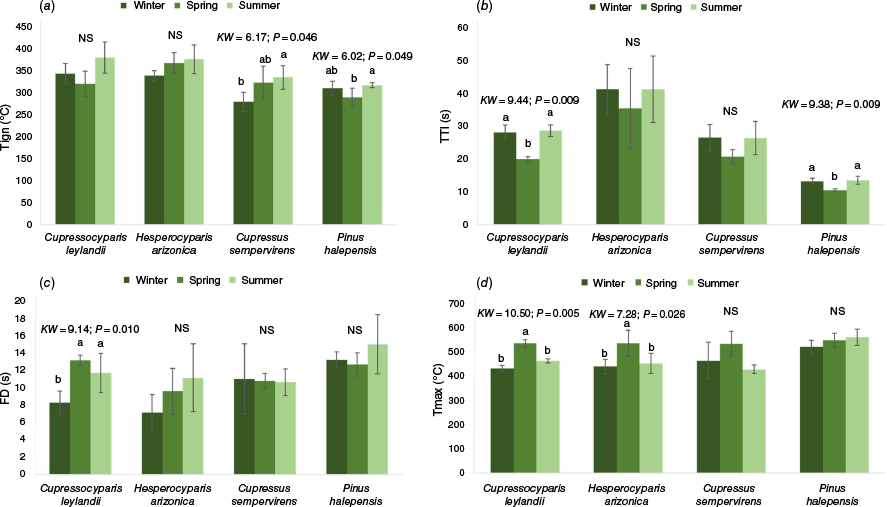
The influence of season on flammability varied among species (Fig. 4). Surprisingly, ignition temperature was consistently higher in summer (meaning leaves were less ignitable) than in winter or spring, but significant differences were only observed in C. sempervirens and P. halepensis (Fig. 4a). The former species had ignition temperatures 12 and 56°C lower in winter than in spring and summer, respectively, whereas the latter had an ignition temperature 30°C lower in spring than in summer. TTI was consistently lowest in spring across all species (meaning leaves were more ignitable) but was only significantly different in C. leylandii and P. halepensis. The former species had 8 and 9 s shorter TTI in spring compared with winter or summer, respectively, whereas TTI in spring was only 3 s shorter than those in both winter and summer for P. halepensis (Fig. 4b). Flaming duration did not show a clear seasonal pattern across most species but results were significant only for C. leylandii, for which flames lasted 5 and 3 s longer in spring and summer, respectively, than in winter (Fig. 4c). Maximum temperature was often higher in spring than in the other seasons but significant differences were only observed in C. leylandii and H. arizonica. The former species presented maximum temperatures that were 103 and 72°C lower in winter and summer, respectively, than in spring, whereas the temperatures were 96 and 84°C lower in winter and summer than in spring for the latter (Fig. 4d).
Temporal variation in the flammability of the four species studied: (a) Ignition temperature (b) Time-to-ignition, (c) Flaming duration, (d) Maximum temperature (Tign, ignition temperature; TTI, time to ignition; Tmax, maximum temperature; FD, flaming duration; NS, non-significant; KW, Kruskal–Wallis test; P, probability). Lowercase letters indicate significant differences among seasons and error bars represent standard deviations.
Role of terpenes and FMC on species’ flammability throughout the year
Most of the variance (>75%) was explained with one or two PCA components (from which the flammability indexes were derived) according to the species and the season. These flammability indexes encompassed the four flammability variables studied, which varied according to the season and species (Table 3): FI when the four variables were related to the first component (as for C. leylandii in spring, H. arizonica in winter and spring, and C. sempervirens in winter), FI1 and FI2 when the variables were spread over the two main components (in most cases).
| Species/Season | % variance explained | Winter | % variance explained | Spring | % variance explained | Summer | |
|---|---|---|---|---|---|---|---|
| Cupressocyparis leylandii | PC1 = 57% | FI1: Tmax (+), FD(+), TTI(−) | PC1 = 82% | FI: Tmax(+), FD(+), TTI(−), Tign(+) | PC1 = 43% | FI1: Tmax(+), Tign(+) | |
| PC2 = 37% | FI2: Tign(−) | PC2 = 36% | FI2: FD(+), TTI(−) | ||||
| Hesperocyparis arizonica | PC1 = 82% | FI: Tmax(+), FD(+), TTI(−), Tign(−) | PC1 = 91% | FI: Tmax(+), FD(+), TTI(−), Tign(−) | PC1 = 60% | FI1: Tmax(−), TTI(+), Tign(+) | |
| PC2 = 27% | FI2: FD(+) | ||||||
| Cupressus sempervirens | PC1 = 78% | FI: Tmax(+), FD(+), TTI(−), Tign(−) | PC1 = 53% | FI1: FD(+), TTI(+), Tign(−) | PC1 = 57% | FI1: FD(+), TTI(+) | |
| PC2 = 41% | FI2: Tmax(+) | PC2 = 38% | FI2: Tmax(+), Tign(+) | ||||
| Pinus halepensis | PC1 = 48% | FI1: Tmax(+), FD(+) | PC1 = 58% | FI1: Tmax(+), FD(+) | PC1 = 65% | FI1: Tmax(+), FD(+), TTI(−) | |
| PC2 = 34% | FI2: TTI(+), Tign(+) | PC2 = 23% | FI2: TTI(+), Tign(−) | PC2 = 27% | FI2: Tign(+) |
When only one component (PC1) explained at least 75% of the variance, the flammability index was denoted FI. The position of the variable on the component (negative or positive side) is given into brackets.
Surprisingly, there was no significant correlation between the flammability indexes and FMC, regardless of the season and the species. However, a significant positive correlation between FMC and TTI was found for H. arizonica (F = 58.57, P = 0.0046, R2 = 0.94) and C. sempervirens (F = 288.96, P = 0.0004, R2 = 0.99) as well as a negative effect on Tmax for P. halepensis (F = 14.2, P = 0.033, R2 = 0.77), but only in one season, differing for each species (summer and winter for the two former species and spring for the latter). Results were non-significant for C. leylandii.
The effect of the terpene subgroup content on flammability (using the flammability index) was significant only for P. halepensis (in spring and winter) and for C. sempervirens (in summer only). For the former species, monoterpenes and diterpenes negatively affected FI2 in both seasons whereas diterpenes were positively related to FI1 in the latter (Supplementary Fig. S1).
Individual terpene compounds correlating with leaf flammability indexes shifted among seasons for each species (Supplementary Fig. S2). For H. arizonica, these compounds were only diterpenes, with nezukol and sempervirol increasing flammability (FI) in winter, and ferruginol in summer (FI1: ignitability through TTI and Tign, and combustibility through Tmax) (Supplementary Fig. S2a). C. sempervirens presented a significant relationship only in summer with opposite effects between sesquiterpenes terpinolene and diterpene abienol decreasing flammability in contrast to monoterpene γ-muurolene (Supplementary Fig. S2b). For C. leylandii, significant results were obtained only in summer and winter, the sesquiterpene elemol decreasing flammability in the former season in contrast to the monoterpene fenchene. An opposite effect on flammability was also observed between some compounds belonging to the same terpene subgroups (the sesquiterpene cedrol increasing flammability in contrast to the sesquiterpene nerodiol) as well as a negative effect of diterpene totarol on sustainability (Supplementary Fig. S2c). P. halepensis had the most compounds significantly affecting flammability (five in winter, one in summer and six in spring; Supplementary Fig. S2d). For this species, monoterpene and sesquiterpene compounds were linked with an increase in flammability in winter and summer in contrast to diterpene manool oxide <13-epi> (only in winter, acting on ignitability through Tign, represented by FI2). In spring, all the significant compounds (regardless of their subgroup) were linked with an increase in flammability, impacting FI2 (Tign), except for diterpene cembrene impacting FI1 (negative effect on flammability).
Dominance of terpene compounds over FMC in their combined effect on flammability across seasons
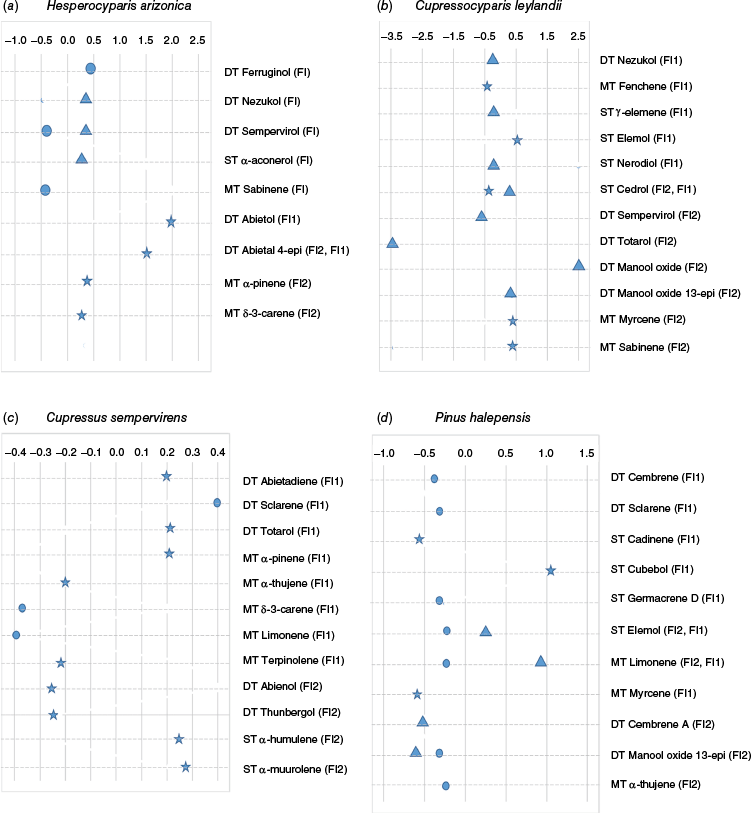
First, taking into account only the effect of terpene compounds on flammability, we found that the composition of terpene compounds (mostly diterpenes) significantly explained flammability and their effects throughout the year differed. The number of significant compounds ranged from 9 for H. arizonica to 12 compounds for C. leylandii and C. sempervirens, occurring mostly in summer for H. arizonica and C. sempervirens as well as in winter for the two other species (PLS regression analyses; Fig. 5). For H. arizonica, the compounds significantly related to FI were predominant only in spring and winter whereas those related to FI1 (abietol) or FI2 (abietal-4-epi) were found only in summer. Most compounds had a positive effect on the flammability indexes and were mainly diterpenes (Fig. 5a). For C. leylandii, there was no significant compound highlighted in spring, regardless of the flammability index. Most compounds related to FI1 were sesquiterpenes, mainly in winter, whereas diterpenes were mostly related to FI2, all found in winter, sometimes presenting an opposite effect on flammability (the diterpene totarol presented a strong negative effect on FI1 in contrast to diterpene manool oxide) (Fig. 5b). For C. sempervirens, apart from winter (non-significant results), the terpene compounds were mostly related to FI1 (8 compounds out of 12, only mono- and diterpenes). In spring, diterpene sclarene and monoterpene limonene best drove the relationship with FI1 (positively for the former in contrast to the latter). Compounds significantly related to FI2 were only highlighted in summer (diterpenes with a negative effect and sesquiterpenes with a positive effect) (Fig. 5c). For P. halepensis, most compounds were related to FI1, often with a negative effect, except sesquiterpene elemol and monoterpene limonene (in winter) and sesquiterpene cubebol (which best drove the relationship with FI1 in summer). Apart from in summer (non-significant results), the compounds affecting FI2 presented a negative effect and were diterpenes (cembrene A and manool oxide <13-epi>) in winter as well as diterpene (manool oxide <13-epi>) and monoterpene (α-thujene) in spring (Fig. 5d). It is worth noting that the significant compounds often did not correspond with the most concentrated molecules (those with a content ≥ 0.10 mg g−1 DM) (Table 2). The models showed that the temporal variation of the proportion of flammability explained by the terpene compounds, given by the R2, varied according to season. The season in which R2 was the highest (R2 > 0.84) varied according to the species (summer for H. arizonica and C. sempervirens, winter for C. leylandii, and winter and summer for P. halepensis; Supplementary Table S2) but this season changed (for C. leylandii and H. arizonica only) whenever FMC was added to the model (Supplementary Table S2).
Synthesis of the results of the different PLS regression analyses, performed for each season and species datasets, featuring the scaled and centred regression coefficients of the significant terpene compounds and the flammability indexes (FI, FI1, FI2) they are linked to: (a) Hesperocyparis arizonica; (b) Cupressocyparis leylandii; (c) Cupressus sempervirens; and (d) Pinus halepensis (DT, diterpene; MT, monoterpenes; ST, sesquiterpenes; star, summer; triangle, winter; circle, spring; significance of the compound when |value| > 0.2).
Adding FMC to the analyses, we found that this factor never was the best driver of flammability (i.e. never presented the highest correlation coefficient in the PLS regression analyses), regardless of the species, season and flammability index (Supplementary Table S2), terpenes always explaining at least 85% of the variance. The fit was slightly improved for 8 models out of 15 but this parameter was not significant in 7 of the 15 models (especially for C. leylandii, in contrast to H. arizonica for which FMC was always significant) (Supplementary Table S2). The fit improved in spring (for FI) and summer (only for FI2) for H. arizonica, in summer only for C. leylandii (for both FI1 and FI2, FMC non-significant in the latter) and for C. sempervirens (for FI2 only, FMC non-significant). A better fit also occurred in spring and summer (for FI1 only) as well as in winter (for FI2 only) for P. halepensis. In these cases, the difference between R2 rarely exceeded 15%.
Discussion
Temporal variation in flammability, terpene and leaf moisture content
In the current study, C. leylandii, P. halepensis and H. arizonica presented the highest flammability in spring in contrast to C. sempervirens, which had surprisingly higher flammability in winter although the driest periods at the time of sampling were spring and summer (Supplementary Fig. S3), therefore the most conducive to fire. This suggested that, for some species, other factors than moisture content could influence flammability, as suggested by Alessio et al. (2008b). Unsurprisingly, this temporal variation in flammability has already been reported by several authors for different Mediterranean species (e.g. Rodriguez Añon et al. 1995; Alessio et al. 2008a).
Seasonal variation in the terpene content was highlighted in the current work, which has already been reported in previous studies (e.g. Peñuelas and Llusià 1997; Llusià and Peñuelas 2000; Alessio et al. 2008b). A seasonal change in terpene concentrations can be of course linked to corresponding changes in temperature (Tingey et al. 1980), radiation or light (Tingey et al. 1980; Langenheim 1994; Loreto et al. 1996) and water availability (Bertin and Staudt 1996; Peñuelas and Llusià 1997; Llusià and Peñuelas 1998), but also to leaf development and activity (Llusià and Peñuelas 2000). The abovementioned abiotic factors need to be considered specially in Mediterranean conditions, which are characterised by marked seasonality and a long dry summer with low precipitation coinciding with high irradiance and high temperature (Di Castri 1973). The seasonal changes may also include different changes for each terpene compound as a result of their different physico-chemical traits (Llusià and Peñuelas 2000). Moreover, the temporal variation of terpene content we highlighted differed according to the level of terpene identification (i.e. subgroup and single compound) but was significant only in two species. Indeed, diterpene content was higher in winter and summer for C. leylandii and in winter for P. halepensis (which had higher monoterpene content in summer as well), which agrees with Alessio et al. (2008b). In contrast to our results regarding the latter species, Llusià and Peñuelas (2000) highlighted a drop in terpene content in spring, regardless of the compound; this difference could be due, among other factors, to a difference in seasonal weather conditions (for instance, in our study, spring was drier than usual) but also to plant genotype and soil differences among studies. Temporal variations in terpene content (and emission) have been reported in several other studies and have confirmed that the season of peak concentrations seems to be species-specific, e.g. in spring for Scots pine and some junipers (Adams 1970), in summer for Douglas-fir and other coniferous species (Wagner et al. 1990; Gambiel and Cates 1995; Zou and Cates 1995), in fall (autumn)–winter, but with larger terpene concentrations also observed at the end of summer (whereas the lowest content was highlighted during the hot season) for some Mediterranean species, including P. halepensis (Llusià and Peñuelas 2000; Peñuelas and Llusià 2001; Llusià et al. 2006). In Lluisà et al. (2006), P. halepensis’ terpene content seemed to follow the annual variation in temperature and soil moisture, with higher values in dry and hot conditions (therefore mainly in summer in Mediterranean areas, which was also the case in our work) in contrast to other Mediterranean species such as Rosmarinus officinalis. In the current study, when a variation of the terpene subgroup content was highlighted, it was due to the temporal variation of some individual terpene compounds. However, the number of compounds that varied throughout the year and therefore the terpene content were low and changed among species (none for C. sempervirens and H. arizonica, only one for P. halepensis and six for C. leylandii, all differing among species). For most compounds, only trends could be highlighted, mostly owing to the high inter-individual content variation within seasons (i.e. high standard deviation; see Table 2). Besides the low number of samples per season, this high variation in terpene content among trees sampled at the same site may indicate genotypic differences as already highlighted for P. halepensis’ serotiny, which was genotypically different according to the past fire history of the populations (Romero et al. 2023), for instance. This possible variation of traits among populations of a same species stresses the need to examine the role of intraspecific/genotypic variation in terpene production. Previous studies usually showed a spatial variation of terpene content resulting from differences in environmental conditions, such as drought stress, or soil fertility (Gilmore 1977; Muzica et al. 1989; Kainulainen et al. 1992; Owens et al. 1998; Ormeño et al. 2008). This could, however, explain the difference in terpene content obtained for some species compared with previous studies (e.g. between 3.6 and 10 mg g−1 in Llusià and Peñuelas (2000) vs between 2 and 2.5 mg g−1 in our work for P. halepensis). Regarding the variation of content according to the different terpenes, Llusià et al. (2006) highlighted a temporal variation in the content of the monoterpene myrcene for P. halepensis that differed from our results (the highest in summer instead of fall–winter) whereas the temporal variation of the sesquiterpene caryophyllene was in agreement with our results.
The expected variation in FMC across seasons, as already highlighted in previous studies carried out on Mediterranean species (e.g. Alessio et al. 2008b), differed according to the species. Indeed, this variation was only significant for two out of the four species studied (C. sempervirens and P. halepensis), showing higher FMC in winter in contrast to spring and summer. This trend differs from that observed on other coniferous species in northwestern America, for instance, with higher FMC in spring (Van Wagner 1967; Rice and Martin 1985; Chuvieco et al. 1999; Agee et al. 2002). Usually, for tree species, the decrease in FMC is not pronounced in the driest season (as found for P. halepensis in Viegas et al. 2001) in contrast to shrubs (Pellizzaro et al. 2007), owing to the deeper root systems of most tree species (Kummerow 1981; Correia et al. 1992; Alessio et al. 2004). This lack of significant temporal variation was what we observed for H. arizonica and C. leylandii. It is worth noting that C. leylandii presented the lowest FMC in winter (in contrast to the other species) despite the fact that the species were all sampled at the same location and at the same time in each season, and therefore, subject to the same weather conditions throughout the year.
Relationships between flammability, terpenes and FMC across seasons
The negative effect of FMC on flammability (but, surprisingly, on only one variable) was significant in one season alone, which differed according to the species. In some cases, FMC showed temporal patterns that differed from those of flammability, meaning that some species (e.g. C. sempervirens) were more flammable during the wet season (winter, in our work). This finding agreed with those of Boving et al. (2023), who showed that leaves of two chaparral shrubs were more flammable during the wet growing season. The authors suggest that seasonal growth traits or drought-related tissue characteristics, such as lignin or chemical content, are critical for determining flammability. However, according to several studies (e.g. Peñuelas and Llusià 1997; Massari and Leopaldi 1998; Owens et al. 1998; McKenzie et al. 2004; Alessio et al. 2008a), the temporal pattern of flammability is usually consistent with the progressive increase in temperatures and water demand, agreeing with the results obtained for P. halepensis.
Terpene content and diversity also presented temporal variations with patterns that differed from those of flammability according to the species. Indeed, total terpene diversity was higher in summer for C. leylandii (owing to higher diversity in diterpenes) and H. arizonica (owing to higher diversity in sesquiterpenes) while both species were more flammable in spring. In contrast, P. halepensis and C. sempervirens presented the same terpene temporal pattern as for flammability (high terpene diversity and flammability in spring and winter, respectively). Regarding the temporal variation of the effect of terpenes on flammability, the number of significant results increased when correlations were performed at the single terpene compound level (compared with subgroup, as already highlighted in Ganteaume et al. 2021), regardless of the season. Some antagonist effects on flammability have also been highlighted between the different levels of terpene identification. For instance, at the terpene subgroup level, diterpenes positively affected C. sempervirens’ flammability (in summer) whereas, at the molecular level, two diterpene compounds showed a negative effect on flammability (out of the four significant in this season). This reinforces the importance of searching for terpene effects at a more refined level than the subgroup, as already highlighted by Ganteaume et al. (2021). The inclusion of minor compounds (i.e. content <0.1 mg g−1) in more explanatory models of flammability indicates that flammability is influenced by more than just the abundance of a particular terpene compound. It is worth noting that, in contrast to the results of Alessio et al. (2008a, 2008b) but in agreement with Guerrero et al. (2024), species with higher total content of terpene compounds were not the most flammable. Indeed, P. halepensis, the most flammable species in the current work, did not present the highest terpene content (which was obtained with C. leylandii). This lack of correlation could be due to interactions between FMC and terpene content on flammability and to the storage of other flammability-related phytochemicals, such as aldehydes, ketones and aromatic compounds, within the burning biomass (Guerrero et al. 2024).
Regarding the possible temporal variation of the combined effect of single terpene compounds and FMC on flammability, our results showed that the composition of terpene compounds significantly explaining flammability varied throughout the year. The effects (positive or negative) of these compounds contrasted according to the species and flammability index taken into account. Only a few compounds (rarely among the most concentrated molecules) were significant in several seasons (except for C. sempervirens), sometimes presenting the opposite effect on flammability among seasons (e.g. diterpene sempervirol for H. arizonica). Besides, possible opposite effects also occurred between flammability indexes (e.g. sesquiterpene cedrol for C. leylandii) and among different compounds of the same terpene subgroup in the same season (e.g. sesquiterpenes cubebol and cadinene in summer for P. halepensis). Previous studies have already shown that monoterpene content, for instance, presented the opposite effect on flammability according to the compound considered (Owens et al. 1998) but none showed that this effect varied throughout the year.
Among the significant terpene compounds, the best drivers of each model, mostly diterpenes, changed among seasons, according to the flammability index considered. The highest proportion of flammability (R2) explained by terpene compounds varied temporally according to the species (e.g. in summer for H. arizonica and C. sempervirens regarding FI1 or in winter for C. leylandii regarding FI2). It is worth noting that some very high values of R2 obtained in the analyses could be partly due to the low number of data points used as input in the analyses. Regarding FMC, previous laboratory-scale studies identified leaf moisture content as a key flammability variable (Pausas et al. 2016) that interacted with the terpenes contained in the leaves (Alessio et al. 2008a; De Lillis et al. 2009). However, Della Rocca et al. (2017) and Ganteaume et al. (2021) confirmed that flammability was not always driven by FMC but also by the terpene content and that, in some cases, there was an interaction between both leaf traits. In the current study, we also highlighted that each species presents its own temporal pattern regarding the different traits, even within a same family (Cupressaceae). Adding FMC improved the fit of half of the models (especially for H. arizonica and P. halepensis) but this parameter was often not significant (in half of the models), regardless of the season. In the rest of the models, the lack of improvement when adding FMC would further underline the fact that other fuel parameters could play a more important role in flammability. It is worth noting that the experimental set-up (epiradiator) only allowed radiant and conductive heat transfers. There could be distinct influences when convection is considered that could reflect on results. Moreover, the PLS regression analyses were performed on a small sample size (N = 5 per season, for each species), which could decrease their statistical power but, however, these analyses allowed deviance from normality and multicollinearity.
Conclusions
The aim of this work was to assess what part terpene content and FMC play on the temporal variation of flammability of different conifer species found at the WUI of SE France (Pinus halepensis, Cupressus sempervirens, Cupressocyparis leylandii and Hesperocyparis arizonica). We highlighted that the temporal patterns of FMC and terpenes (for both composition and content) were highly variable, contributing to high variation in seasonal patterns of flammability. Moreover, we found that the terpene content at the compound level was a more important driver of seasonal flammability than FMC. However, the patterns varied according to the species, even within a same family (Cupressaceae), which highlighted the complex nature of flammability within and among species.
Even though laboratory experiments cannot be used for the prediction or the assessment of fuel flammability under natural conditions (i.e. in situ fire behaviouras opposed to fire behaviour in laboratory conditions), they can help to improve our knowledge of the effects of fuel properties on flammability and represent basic information for assessing fire risk as well as provide valuable input data in fire behaviour models. Currently, the absence of chemical compounds such as terpenes in the models may also be due to our relatively poor understanding of foliar chemistry and its interactions with moisture content. This could also lead to a lack of consensus about the importance of foliar moisture content in the prediction of crown fire behaviour.
The flammability of the highly flammable Aleppo pine, as that of the other species studied in the current work, could also be enhanced by some single terpene compounds, especially in spring. This means that these species could affect fire hazard not just in summer, as it is commonly assumed in the Mediterranean regions. We recommend that fire prevention strategies in the wildland–urban interface and in forest areas consider the potential that fire hazard may not be lower during non-summer months as often expected. This distinction is particularly important considering that climate change is steadly increasing the fire season length and the frequency of drought events outside of the summer season.
Data availability
Data for this manuscript is accessible at the Dryad Digital Repository. A preprint version of this article is available at: https://assets-eu.researchsquare.com/files/rs-634012/v1/fb6a05fd-64b5-48b4-9c7c-da1872b2039a.pdf?c=1631885068.
Acknowledgements
We gratefully thank Amélie Saunier (Institut Méditerranéen de Biodiversité et d’Ecologie) for her help in the chemical analyses as well as Fabien Guerra, Ugo Furet, Christian Travaglini and Denis Morge (INRAE) for their help during the burning experiments. The authors also sincerely thank Aimee MacCormack for English revision.
References
Adams RP (1970) Seasonal variation of terpenoid constituents in natural populations of Juniperus pinchotii Sudw. Phytochemistry 9, 397-402.
| Crossref | Google Scholar |
Adams RP, Bartel JA, Price RA (2009) A new genus, Hesperocyparis, for the cypresses of the western hemisphere. Phytologia 91(1), 160-185.
| Google Scholar |
Agee JK, Wright CS, Williamson N, Huff MH (2002) Foliar moisture content of Pacific Northwest vegetation and its relation to wildland fire behavior. Forest Ecology and Management 167, 57-66.
| Crossref | Google Scholar |
Alessio G, De Lillis M, Brugnoli E, Lauteri M (2004) Water sources and water-use efficiency in Mediterranean coastal dune vegetation. Plant Biology 6, 350-357.
| Crossref | Google Scholar | PubMed |
Alessio G, Peñuelas J, De Lillis M, Llusià J (2008a) Implications of foliar terpene content and hydration on leaf flammability of Quercus ilex and Pinus halepensis. Plant Biology 10, 123-128.
| Crossref | Google Scholar |
Alessio G, Peñuelas J, Llusià J, Ogaya R, Estiarte M, De Lillis M (2008b) Influence of water and terpenes on flammability in some dominant Mediterranean species. International Journal of Wildland Fire 17, 274-286.
| Crossref | Google Scholar |
Anderson HE (1970) Forest fuel ignitibility. Fire Technology 6, 312-319.
| Crossref | Google Scholar |
Bertin N, Staudt M (1996) Effect of water stress on monoterpene emissions from young potted holm oak (Quercus ilex L.) trees. Oecologia 107, 456-462.
| Crossref | Google Scholar | PubMed |
Blanch J-S, Peñuelas J, Sardans J, Llusià J (2009) Drought, warming and soil fertilization effects on leaf volatile terpene concentrations in Pinus halepensis and Quercus ilex. Acta Physiologiae Plantarum 31, 207-218.
| Crossref | Google Scholar |
Bloomfield KJ, Cernusak L, Eamus D, Ellsworth DS, Prentice IC, Wright IJ, Boer MM, Bradford M, Cale P, Cleverly JR, Egerton J, Evans B, Hayes L, Hutchinson M, Liddell MJ, Macfarlane C, Meyer WS, Prober SM, Togashi FH, Wardlaw T, Zhu L, Atkin O (2018) A continental-scale assessment of variability in leaf traits: within species, across sites and between seasons. Functional Ecology 32, 1492-1506.
| Crossref | Google Scholar |
Boving I, Celebrezze J, Salladay R, Ramirez A, Anderegg LDL, Moritz M (2023) Live fuel moisture and water potential exhibit differing relationships with leaf‐level flammability thresholds. Functional Ecology 37, 2770-2785.
| Crossref | Google Scholar |
Brofas G, Karetsos G, Dimopoulos P, Tsagari C (2006) The natural environment of Cupressus sempervirens in Greece as a basis for its use in the Mediterranean region. Land Degradation and Development 17, 645-659.
| Crossref | Google Scholar |
Cappelli M, Bonani S, Conci I (1983) Sul grado d’infiammabilità di alcune specie della macchia Mediterranea. Collana Verde 62, 1-52 [In Italian].
| Google Scholar |
Cawson JG, Burton JE, Pickering BJ, Demetriou V, Filkov AI (2023) Quantifying the flammability of living plants at the branch scale: which metrics to use? International Journal of Wildland Fire 32(10), 1404-1421.
| Crossref | Google Scholar |
Centritto M, Brilli F, Fodale R, Loreto F (2011) Different sensitivity of isoprene emission, respiration and photosynthesis to high growth temperature coupled with drought stress in black poplar (Populus nigra) saplings. Tree Physiology 31, 275-286.
| Crossref | Google Scholar | PubMed |
Chetehouna K, Barboni T, Zarguili E, Simeoni A, Fernandez-Pello A-C (2009) Investigation on the emission of volatile organic compounds from heated vegetation and their potential to cause an accelerating forest fire. Combustion Science and Technology 181(10), 1273-1288.
| Crossref | Google Scholar |
Clarke PJ, Prior LD, French BJ, Vincent B, Knox KJE, Bowman DMJS (2014) Using a rainforest-flame forest mosaic to test the hypothesis that leaf and litter fuel flammability is under natural selection. Oecologia 176, 1123-1133.
| Crossref | Google Scholar | PubMed |
Cornelissen JHC, Lavorel S, Garnier E, Diaz S, Buchmann N, Gurvich DE, Reich PB, Ter Steege H, Morgan HD, Van der Heijden MGA, Pausas JG, Poorter H (2003) ) Handbook of protocols for standardized and easy measurement of plant functional traits worldwide. Australian Journal of Botany 51, 335-380.
| Crossref | Google Scholar |
Cornwell WK, Elvira A, van Kempen L, Van Logtestijn RSP, Aptroot A, Cornelissen JHC (2015) Flammability across the gymnosperm phylogeny: the importance of litter particle size. New Phytologist 206, 672-681.
| Crossref | Google Scholar | PubMed |
Correia OA, Oliveira G, Martins-Loucão MA, Catarino FM (1992) Effects of bark stripping on the water relations of Quercus suber L. Scientia Gerundensis 18, 195-204.
| Google Scholar |
Courty L, Chetehouna K, Halter F, Foucher F, Garo J-P, Mounaïm-Rousselle C (2012) Flame speeds of α-pinene/air and limonene/air mixtures involved in accelerating forest fires. Combustion Science and Technology 184(10–11), 1397-1411.
| Crossref | Google Scholar |
De Lillis M, Bianco PM, Loreto F (2009) The influence of leaf water content and terpenoids on flammability of some Mediterranean woody species. International Journal of Wildland Fire 18, 203-212.
| Crossref | Google Scholar |
Della Rocca G, Madrigal J, Marchi E, Michelozzi M, Moya B, Danti R (2017) Relevance of terpenoids on flammability of Mediterranean species: an experimental approach at a low radiant heat flux. iForest 10, 766-775.
| Crossref | Google Scholar |
de Magalhães RMQ, Schwilk DW (2012) Leaf traits and litter flammability: evidence for non-additive mixture effects in a temperate forest. Journal of Ecology 100, 1153-1163.
| Crossref | Google Scholar |
Engber EA, Varner JM (2012) Patterns of flammability of the California oaks: the role of leaf traits. Canadian Journal of Forest Research 42, 1965-1975.
| Crossref | Google Scholar |
Fajardo A, Siefert A (2016) Phenological variation of leaf functional traits within species. Oecologia 180, 951-959.
| Crossref | Google Scholar | PubMed |
Gambiel HA, Cates RG (1995) Terpene changes due to maturation and canopy level in Douglas fir (Pseudotsuga menziesii) flush needle oil. Biochemical Systematics and Ecology 5, 469-476.
| Crossref | Google Scholar |
Ganteaume A (2018) Does plant flammability differ between leaf and litter bed scale? Role of fuel characteristics and consequences for flammability assessment. International Journal of Wildland Fire 27, 342-352.
| Crossref | Google Scholar |
Ganteaume A, Jappiot M, Lampin C (2013) Assessing the flammability of surface fuels beneath ornamental vegetation in wildland–urban interfaces in Provence (south-eastern France). International Journal of Wildland Fire 22, 333-342.
| Crossref | Google Scholar |
Ganteaume A, Romero B, Fernandez C, Ormeño E, Lecareux C (2021) Volatile and semi-volatile terpenes impact leaf flammability: differences according to the level of terpene identification. Chemoecology 31, 259-275.
| Crossref | Google Scholar |
Gilmore AR (1977) Effects of soil moisture stress on monoterpenes in loblolly pine. Journal of Chemical Ecology 3, 667-676.
| Crossref | Google Scholar |
Grootemaat S, Wright I, Van Bodegom P, Cornwell W (2015) Burn or rot: leaf traits explain why flammability and decomposability are decoupled across species. Functional Ecology 29, 1486-1497.
| Crossref | Google Scholar |
Guerrero F, Carmona C, Hernández C, Toledo M, Arriagada A, Espinoza L, Bergmann J, Taborga L, Yañez K, Carrasco Y, Muñoz AA (2022) Drivers of flammability of Eucalyptus globulus Labill leaves: terpenes, essential oils, and moisture content. Forests 13, 908.
| Crossref | Google Scholar |
Guerrero F, Espinoza L, Carmona C, Blackhall M, Quintero C, Ocampo-Zuleta K, Paula S, Madrigal J, Guijarro M, Carrasco Y, Bustamante-Sánchez MA, Miranda A, Yáñez K, Bergmann J, Taborga L, Toledo M (2024) Unravelling the chemistry of plant flammability: exploring the role of volatile secondary metabolites beyond terpenes. Forest Ecology and Management 572, 122269.
| Crossref | Google Scholar |
Kainulainen P, Oksanen J, Palomäki V, Holopainen J, Holopainen T (1992) Effect of drought and waterlogging stress on needle monoterpenes of Picea abies. Canadian Journal of Botany 70, 1613-1616.
| Crossref | Google Scholar |
Langenheim JH (1994) Higher plant terpenoids: a phytocentric overview of their ecological roles. Journal of Chemical Ecology 20, 1223-1280.
| Crossref | Google Scholar | PubMed |
Llusià J, Peñuelas J (1998) Changes in terpene content and emission in potted Mediterranean woody plants under severe drought. Canadian Journal of Botany 76, 1366-1373.
| Crossref | Google Scholar |
Llusià J, Peñuelas J (2000) Seasonal patterns of terpene content and emission from seven Mediterranean woody species in field conditions. American Journal of Botany 87, 133-140.
| Google Scholar | PubMed |
Llusià J, Peñuelas J, Alessio GA, Estiarte M (2006) Seasonal contrasting changes of foliar concentrations of terpenes and other volatile organic compound in tour dominant species of a Mediterranean shrubland submitted to a field experimental drought and warming. Physiologioa Plantarrum 127, 632-649.
| Crossref | Google Scholar |
Loreto F, Ciccioli P, Cecinato A, Brancaleoni E, Frattoni M, Tricoli D (1996) Influence of environmental factors and air composition on the emission of α-pinene from Quercus ilex leaves. Plant Physiology 110, 267-275.
| Crossref | Google Scholar | PubMed |
Madrigal J, Hernando C, Guijarro M (2013) A new bench-scale methodology for evaluating flammability of live forest fuels. Journal of Fire Science 31(2), 131-142.
| Crossref | Google Scholar |
Marchese JA, Ferreira JFS, Rehder VLG, Rodrigues O (2010) Water deficit effect on the accumulation of biomass and artemisinin in annual wormwood (Artemisia annua L., Asteraceae). Brazilian Journal of Plant Physiology 22, 1-9.
| Crossref | Google Scholar |
Marino E, Guijarro M, Hernando C, Madrigal J, Díez C (2011) Fire hazard after prescribed burning in a gorse shrubland: implications for fuel management. Journal of Environmental Management 92, 1003-1011.
| Crossref | Google Scholar | PubMed |
Martin RE, Gordon DA, Gutierrez M, Lee DS, Molina DM, Schroeder RA, Sapsis DB, Stephens SL, Chambers M (1993) Assessing the flammability of domestic and wildland vegetation. In ‘Proceedings of the Proceedings of the 12th conference on fire and forest meteorology’. pp. 26–28. (Society of American Foresters Publication 94-02: Bethesda, MD, USA)
Massari G, Leopaldi A (1998) Leaf flammability in Mediterranean species. Plant Biosystems 132(1), 29-38.
| Crossref | Google Scholar |
McKenzie D, Gedalof ZE, Peterson DL, Mote P (2004) Climatic change, wildfire, and conservation. Conservation Biology 18(4), 890-902.
| Crossref | Google Scholar |
Murray BR, Hardstaff LK, Phillips ML (2013) Differences in leaf flammability, leaf traits and flammability-trait relationships between native and exotic plant species of dry sclerophyll forest. PLoS One 8, e79205.
| Crossref | Google Scholar | PubMed |
Muzica RM, Pregitzer KS, Hanover JW (1989) Changes in terpene production following nitrogen fertilization of grand fir [Abies grandis (Dougl.) Lindl.] seedlings. Oecologia 8, 485-489.
| Crossref | Google Scholar |
Ormeño E, Baldy V, Ballini C, Fernandez C (2008) Production and diversity of volatile terpenes from plants on calcareous and siliceous soils: effect of soil nutrients. Journal of Chemical Ecology 34, 1219-1229.
| Crossref | Google Scholar | PubMed |
Ormeño E, Cespedes B, Sanchez IA, Velasco-García A, Moreno JM, Fernandez C, Baldy V (2009) The relationship between terpenes and flammability of leaf litter. Forest Ecology and Management 257, 471-482.
| Crossref | Google Scholar |
Ormeño E, Ruffault J, Gutigny C, Madrigal J, Guijarro M, Hernando C, Ballini C (2020) Increasing cuticular wax concentrations in a drier climate promote litter flammability. Forest Ecology and Management 473, 118242.
| Crossref | Google Scholar |
Owens MK, Lin C-D, Taylor CA, Whisenant SG (1998) Seasonal patterns of plant flammability and monoterpenoid content in Juniperus ashei. Journal of Chemical Ecology 24, 2115-2129.
| Crossref | Google Scholar |
Page WG, Jenkins MJ, Runyon JB (2012) Mountain pine beetle attack alters the chemistry and flammability of lodgepole pine foliage. Canadian Journal of Forest Research 42, 1631-1647.
| Crossref | Google Scholar |
Pausas JG, Moreira B (2012) Flammability as a biological concept. New Phytologist 194, 610-613.
| Crossref | Google Scholar | PubMed |
Pausas JG, Alessio GA, Moreira B, Segarra-Moragues JG (2016) Secondary compounds enhance flammability in a Mediterranean plant. Oecologia 180, 103-110.
| Crossref | Google Scholar | PubMed |
Pellizzaro G, Duce P, Ventura A, Zara P (2007) Seasonal variations of live moisture content and ignitability in shrubs of the Mediterranean Basin. International Journal of Wildland Fire 16, 633-641.
| Crossref | Google Scholar |
Pellizzaro G, Ventura A, Arca B, Arca A, Duce P (2009) Weather seasonality and temporal pattern of live and dead fuel moisture content in Mediterranean shrubland. In ‘European Geosciences Union General Assembly 2009’, 19-24 April 2009, Vienna, Austria. p. 12100. Available at http://meetings.copernicus.org/egu2009
Peñuelas J, Llusià J (1997) Effects of carbon dioxide, water supply, and seasonality on terpene content and emission by Rosmarinus officinalis. Journal of Chemical Ecology 23, 979-993.
| Crossref | Google Scholar |
Peñuelas J, Llusià J (2001) The complexity of factors driving volatile organic compound emissions by plants. Biologia Plantarum 44, 481-487.
| Crossref | Google Scholar |
Popović Z, Bojović S, Marković M, Cerdà A (2021) Tree species flammability based on plant traits: a synthesis. Science of The Total Environment 800, 149625.
| Crossref | Google Scholar | PubMed |
Rodriguez Añon JA, Fraga López F, Proupín Castiñeiras J, Palacios Ledo J, Núñez Regueira L (1995) Calorific values and flammability for forest wastes during the seasons of the year. Bioresource Technology 52, 269-274.
| Crossref | Google Scholar |
Romero B, Fernandez C, Lecareux C, Ormeño E, Ganteaume A (2019) How terpene content affects fuel flammability of wildland–urban interface vegetation. International Journal of Wildland Fire 28, 614-627.
| Crossref | Google Scholar |
Romero B, Scotti I, Fady B, Ganteaume A (2023) Fire frequency, as well as stress-response and developmental genes control serotiny level variation in a widespread pioneer Mediterranean conifer. Ecology and Evolution 13(3), e9919.
| Crossref | Google Scholar | PubMed |
Sampedro L, Moreira X, Zas R (2011) Costs of constitutive and herbivore-induced chemical defences in pine trees emerge only under low nutrient availability. Journal of Ecology 99, 818-827.
| Crossref | Google Scholar |
Santoni PA, Bartoli P, Simeoni A, Torero J (2014) Bulk and particle properties of pine needle fuel beds – influence on combustion. International Journal of Wildland Fire 23, 1076-1086.
| Crossref | Google Scholar |
Scarff FR, Westoby M (2006) Leaf litter flammability in some semi-arid Australian woodlands. Functional Ecology 20, 745-752.
| Crossref | Google Scholar |
Schwilk DW, Caprio AC (2011) Scaling from leaf traits to fire behaviour: community composition predicts fire severity in a temperate forest. Journal of Ecology 99, 970-980.
| Crossref | Google Scholar |
Tingey DT, Manning M, Grothaus LC, Burns WF (1980) Influence of light and temperature on monoterpene emission rates from slash pine. Plant Physiology 65, 797-801.
| Crossref | Google Scholar | PubMed |
Valette JC (1990) Inflammabilités des espèces forestières méditerranéennes. Revue Forestière Française 42, 76-92.
| Crossref | Google Scholar |
Varner JM, Kane JM, Kreye JK, Engber E (2015) The flammability of forest and woodland litter: a synthesis. Current Forestry Reports 1, 91-99.
| Crossref | Google Scholar |
Viegas DX, Piñol J, Viegas MT, Ogaya R (2001) Estimating live fine fuels moisture content using meteorologically based indices. International Journal of Wildland Fire 10, 223-240.
| Crossref | Google Scholar |
Wagner MR, Clancy KM, Tinus RW (1990) Seasonal patterns in the allelochemicals of Pseudotsuga menziesii, Picea engelmannii and Abies concolor. Biochemical Systematics and Ecology 18, 215-220.
| Crossref | Google Scholar |
Weise DR, White RH, Beall FC, Etlinger M (2005) Use of the cone calorimeter to detect seasonal differences in selected combustion characteristics of ornamental vegetation. International Journal of Wildland Fire 14, 321-338.
| Crossref | Google Scholar |
White CS (1994) Monoterpenes: their effects on ecosystem nutrient cycling. Journal of Chemical Ecology 20, 1381-1406.
| Crossref | Google Scholar | PubMed |
White RH, Zipperer WC (2010) Testing and classification of individual plants for fire behaviour: plant selection for the wildland–urban interface. International Journal of Wildland Fire 19, 213-227.
| Crossref | Google Scholar |
Zou JP, Cates RG (1995) Foliage constituents of Douglas fir (Pseudotsuga menziesii (Mirb) Franco (Pinaceae) - their seasonal resistance and silviculture management. Journal of Chemical Ecology 21, 387-402.
| Crossref | Google Scholar | PubMed |


