Changes in systemic cancer therapy in Australia during the Delta and Omicron waves of the COVID-19 pandemic (2021–2022): a controlled interrupted time series analysis
Forrest Koch A , Monica Tang A , Ramesh Walpola C , Maria Aslam D , Sallie-Anne Pearson G # * and Benjamin Daniels A # *A
B
C
D
E
F
G
H
I
# SAP and BD were responsible for equal contributions as senior authors.
Abstract
Objectives:Australian lockdowns in response to the initial coronavirus disease 2019 (COVID-19) outbreak in 2020 were associated with small and transient changes in the use of systemic cancer therapy. We aimed to investigate the impacts of the longer and more restrictive lockdowns in the Australian states of New South Wales (NSW) and Victoria during both the Delta subvariant lockdowns in mid-2021 and the Omicron subvariant outbreak in late 2021/early 2022. Study type: Population-based, controlled interrupted time series analysis. Methods: We conducted a national observational study using de-identified records of government-subsidised cancer medicines dispensed to a random 10% sample of Australians between July 2018 and July 2022. We used controlled interrupted time series analysis to investigate changes in the dispensing, initiation and discontinuation of all cancer medicines dispensed to residents of NSW and Victoria, using the rest of Australia as a control series. We used quasi-Poisson regression to model weekly counts and estimate incidence rate ratios (IRRs) for the effect of (each) the Delta phase lockdown and the Omicron outbreak on our systemic cancer therapy outcomes. Results: Between July 2018 and July 2022, cancer medicines were dispensed 592 141 times to 33 198 people in NSW and Victoria. Overall, there were no changes to the rates of dispensing, initiation or discontinuation of antineoplastics during the Delta phase lockdowns. In both states during the Omicron outbreak, there were significant decreases in the dispensing of antineoplastics (NSW IRR 0.89; 95% confidence interval [CI] 0.84, 0.93. Victoria IRR 0.92; 95% CI 0.88, 0.96) and in the initiation of endocrine therapy (NSW IRR 0.85; 95% CI 0.74, 0.99. Victoria IRR 0.78; 95% CI 0.65, 0.94), and no changes in the discontinuation of any systemic cancer therapy. Conclusions: The 2021 lockdowns and 2021/2022 Omicron outbreaks in NSW and Victoria had significant impacts on the dispensing, initiation and discontinuation of systemic cancer therapies, however, the overall effects were minimal. The impacts of lockdowns were less significant than the Omicron outbreaks, suggesting COVID-19 infection, health system capacity, and patient and community concerns were important factors for treatment changes.Introduction
The coronavirus disease 2019 (COVID-19) pandemic has significantly disrupted cancer care around the world. Since the initial outbreak in March 2020, there have been numerous reports of interruptions to screening programs1-3, declines in cancer diagnoses1, delayed surgeries4, and patient hesitancy to engage with the health system.5-7 Many jurisdictions and cancer organisations implemented policies designed to adapt to, and/or mitigate the effects of COVID-19, such as stay-at-home orders, elective surgery bans and revised clinical guidelines, that had further impacts on patient care.1
The Australian experience of the COVID-19 pandemic during 2020 differed from other developed nations in that COVID-19 infections, hospitalisations and deaths were comparatively low.8 However, the outbreak of the Delta COVID-19 variant in June 2021 resulted in increased case numbers, and the two most populous Australian states, New South Wales (NSW) and Victoria, entered lockdowns in late June and early July, respectively.9 As many Australian jurisdictions began removing restrictions in December 2021, the Omicron COVID-19 variant became prevalent and case numbers dramatically increased – by orders of magnitude greater than those reported during the Delta outbreak – into early 2022.
Our previous study found that the impacts of the COVID-19 lockdowns on the use of systemic cancer treatment in Australia during 2020 were small and transient.10 However, the impacts of the longer, more restrictive lockdowns during 2021 and the more virulent Omicron outbreak are not yet known. The aim of our study was to investigate and quantify changes in systemic cancer therapy during the Delta and Omicron phases of the COVID-19 pandemic in Australia. We examine changes in the dispensing, initiation and discontinuation of systemic cancer medicines and supportive medicines in NSW and Victoria.
Methods
Study setting and data
Our population-based, observational study used records of all cancer medicines and supportive cancer medicines dispensed to a random 10% sample of Pharmaceutical Benefits Scheme (PBS)-eligible Australian residents between July 2018 and July 2022. PBS dispensing claims are processed and maintained by Services Australia. The de-identified, individual-level data included the name of the dispensed medicine, the date of dispensing, and the state in which the medicine was dispensed.
All medicines dispensed in the community, private hospitals, and to outpatients in public hospitals are captured in PBS data. The PBS does not subsidise medicines dispensed within public hospitals to public hospital inpatients. Because of this arrangement, most systemic cancer treatments are administered in the outpatient setting in Australia. Once listed on the PBS, the Australian Government covers the cost of the medicine less standard PBS co-payment amounts. Private insurance does not provide reimbursement for medicines already subsidised through public programs, and, due to their high cost, it is unlikely that patients would access these medicines through other avenues. Consequently, PBS data capture the majority cancer medicines dispensed in Australia.
COVID-19 in Australia during 2021–2022
Figure 1 and Supplementary Figure A illustrate the Australian context of the COVID-19 pandemic. The Delta outbreak began in June 2021, and Australia’s two–most populous states – NSW and Victoria – enacted a range of policy responses, including stay-at-home orders (lockdowns), from 25 June (NSW) and 15 July (Victoria) 2021 (Supplementary Table A, available from: doi.org/10.6084/m9.figshare.c.7040453). These lockdowns persisted until 11 October (NSW) and 21 October (Victoria) 2021, when fully vaccinated residents were permitted to circulate in the community. Between December 2021 and January 2022, the Omicron variant became the prevalent COVID-19 strain. NSW and Victoria did not reimpose lockdowns in response. However, mobility in the metropolitan areas surrounding the capital cities of both states (Sydney and Melbourne) decreased significantly as many residents limited their movements.11
Daily number of new COVID-19 cases, deaths, hospitalisations and ICU admissions in Australia between January 2021 and June 2022
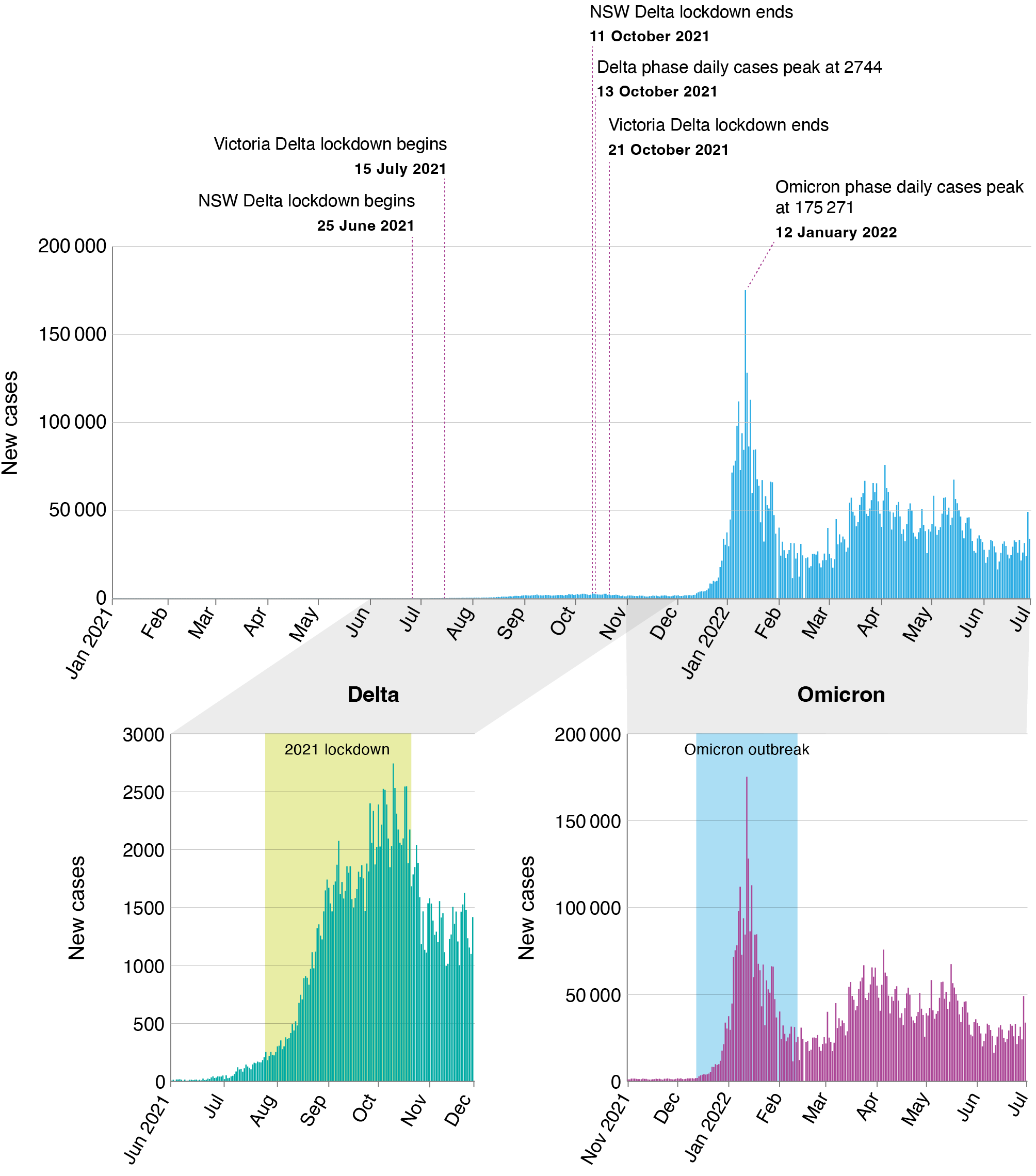
NSW: New South Wales.
Notes: Top graph represents January 2021–July 2022 and includes both the Delta and Omicron phases.
Bottom left graph represents the Delta phase (note different y-axis scale).
Bottom right graph represents the Omicron phase.
The yellow shaded regions indicate the 2021 lockdown and the blue shaded regions indicate the Omicron outbreak.
Other Australian states did not experience significant outbreaks of the Delta variant during 2021. These jurisdictions had disparate, short-duration lockdowns – typically lasting 3–7 days – during both the Delta and Omicron phases of the pandemic. By mid-2022, while reported case numbers and COVID-19-related deaths remained high, almost all COVID-19 restrictions in Australia were ended. We focused our study on NSW and Victoria because the residents of these states experienced the most restrictive policies in response to the Delta outbreak in Australia.
Medicines of interest
We included all publicly-subsidised systemic cancer therapies in our study, comprising antineoplastic agents (World Health Organization Anatomical Therapeutic Chemical classification L01), endocrine therapies, and medicines typically prescribed to support patients undergoing cancer treatment. We excluded oral formulations of methotrexate, cyclophosphamide and mercaptopurine because these are not typically used in cancer therapy (see Supplementary Table B for a complete list of the medicines examined, available from: doi.org/10.6084/m9.figshare.c.7040453).
Outcome measures and statistical analyses
We examined three medicine utilisation measures – weekly dispensing, initiation and discontinuation – stratified by medicine class (chemotherapy, immunotherapy, targeted therapy), route of administration (oral, parenteral), and state (NSW, Victoria). We defined treatment initiation as the dispensing of a medicine with no dispensing of the same medicine or medicine class during the preceding 365 days. We defined discontinuation as a gap of ≥ 90 days between the dispensing of medicines or following the last observed dispensing. We estimated the date of discontinuation as the date of the last dispensing before a ≥ 90-day gap, plus 30 days. As a sensitivity analysis, we also used 60-day and 120-day periods to define discontinuation.
We used controlled interrupted time series analysis (CITS) to quantify changes for each combination of medicine class, outcome measure and state (NSW, Victoria) during the lockdown period and Omicron outbreak, using the remaining Australian states (combined) as the control series. By using an appropriate control series, CITS is less prone to time-varying and cointervention confounding, which are the primary threats to internal validity in single interrupted time series.12,13 We selected the other Australian states and territories as a control because these jurisdictions did not experience the more restrictive policy responses to the Delta outbreak that both NSW and Victoria experienced. These other jurisdictions also experienced smaller and later Omicron outbreaks.
To account for overdispersion present in the data, we used a quasi-Poisson regression model with a log-link function to model weekly counts and estimate the incidence rate ratio (IRR) for the effect of each lockdown and outbreak event on our systemic cancer therapy outcomes. We included log-transformed counts for the control series as an offset and indicator variables for the 2020 lockdowns, 2021 lockdowns and Omicron outbreak. We assumed global trends to be linear with respect to log-transformed counts and accounted for them in the models. We defined the:
NSW 2020 lockdown period as 15 March 2020–1 July 2020
Victoria 2020 lockdown periods as 30 March 2020–12 May 2020 and 8 July 2020–27 October 2020
NSW 2021 lockdown period as 25 June 2021–11 October 2021
Victoria 2021 lockdown period as 15 July 2021–21 October 2021
Omicron outbreak period for both states as 12 December 2021–12 February 2022.
Models were assessed for autocorrelation by inspecting the autocorrelation function and model fit plots.
The regression model can be summarised by the following formula
Where λt is the expected or mean rate of dispensing, initiation or discontinuation per population at time for the time series being modelled; β0 is the model intercept; is the global trend; β2, β3 and β4 are the level change effects of the 2020 lockdown periods, 2021 lockdown periods and Omicron outbreak, respectively; IA(t), IB(t) and IC(t) are indicator functions for the 2020 lockdown periods, 2021 lockdown periods and Omicron outbreak, respectively, and take a value of 1 during the event and 1 otherwise; and is an offset for the expected count of dispensing, initiation or discontinuation at time in the control states.
All analyses were performed using R statistical software (v4.0.4).14
Results
Between July 2018 and July 2022, cancer medicines were dispensed 592 141 times (337 024 for antineoplastic medicines and 255 117 for endocrine therapies) to 33 198 people in NSW and Victoria in the PBS 10% sample data. Most people (55%) who were dispensed a cancer medicine were aged >65 years. The most frequently dispensed antineoplastic medicines were fluorouracil (13% of dispensed antineoplastic medicines), paclitaxel (7%) and carboplatin (4%). The most frequently dispensed endocrine therapies were anastrozole (28% of dispensed endocrine therapies), letrozole (27%) and tamoxifen (18%).
Antineoplastic medicines
Dispensing
During the 2021 lockdowns, estimated IRRs were similar for all dispensing of antineoplastic medicines in NSW and Victoria: 1.03 (95% confidence interval [CI] 0.99, 1.06) and 1.04 (95% CI 1.00, 1.08), respectively (Figures 2 and 3). We also observed a significant increase in the dispensing rate of parenteral chemotherapy in Victoria (1.07; 95% CI 1.02, 1.12), and a similar increase in NSW (1.03; 95% CI 0.99, 1.09). We did not observe any other significant changes in antineoplastic medicine dispensing rates during the 2021 lockdowns for either NSW or Victoria.
IRR estimates and 95% CI for the impacts of the Delta lockdowns and Omicron outbreaks on systemic cancer medicines in NSW and Victoria
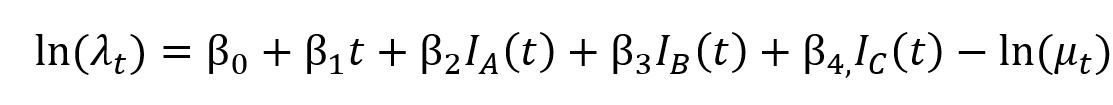
NSW: New South Wales
Notes: The points represent IRR estimates and the lines represent 95% CI limits.
The IRRs compare the post-intervention rate (numerator) to the pre-intervention rate (denominator) while adjusting for co-occurring effects observed in the control series.
NSW: New South Wales
Notes: The yellow shaded regions indicate the 2021 Delta lockdowns, the blue shaded regions indicate the Omicron outbreak and the grey shaded regions indicate the 2020 lockdowns.
During the Omicron outbreak, both NSW and Victoria experienced significant reductions in the dispensing rates of therapies including: antineoplastic medicines (NSW IRR 0.89; 95% CI 0.84, 0.93. Victoria IRR 0.92; 95% CI 0.88, 0.96); parenteral chemotherapies (NSW IRR 0.88; 95% CI 0.82, 0.94. Victoria IRR 0.91; 95% CI 0.85, 0.97); parenteral immunotherapies (NSW IRR 0.88; 95% CI 0.79, 0.97. Victoria IRR 0.88; 95% CI 0.78, 0.99); and parenteral targeted therapies (NSW IRR 0.84; 95% CI 0.75, 0.93. Victoria IRR 0.83; 95% CI 0.75, 0.91). (see Figures 2 and 3)
Initiation
In NSW during the 2021 lockdown, there was a significant increase in the initiation rate of parenteral immunotherapies (1.49; 95% CI 1.11, 2.00) and a decrease in the initiation rate of oral targeted therapies (0.50; 95% CI 0.32, 0.77) (Figures 2 and 4). We did not observe any other significant changes to the initiation rate of antineoplastic medicines in NSW or Victoria.
NSW: New South Wales
Yellow shaded regions indicate the 2021 Delta lockdown; blue shaded regions indicate the Omicron outbreak; and grey shaded regions indicate the 2020 lockdowns.
During the Omicron outbreak, there were no significant changes in the overall initiation rate of antineoplastic medicines in either NSW (0.87; 95% CI 0.74, 1.02) or Victoria (0.87; 95% CI 0.73, 1.04) (Figures 2 and 4). We observed a marginally significant reduction in the initiation rate of parenteral chemotherapy in NSW (0.76; 95% CI 0.57, 1.00), and in the initiation rate of oral targeted therapy in Victoria (0.50; 95% CI 0.25, 1.01).
Discontinuation
We observed a significant decrease in the discontinuation of endocrine therapy during the Omicron outbreak in Victoria (0.88; 95% CI 0.78, 0.98). We did not observe significant changes to the discontinuation rates of any other antineoplastic medicine classes in NSW or Victoria for either the 2021 lockdowns or the Omicron outbreak (Figure 2; Supplementary Figure B, available from: doi.org/10.6084/m9.figshare.c.7040453). Sensitivity analyses (not included in this manuscript) did not yield any meaningful differences from the primary analyses.
Endocrine therapies
During the 2021 lockdowns, there were no significant changes to the rates of dispensing, initiation or discontinuation of endocrine therapy for either NSW or Victoria (Figure 2). Estimated IRRs indicated a slight reduction in the rate of dispensing during the Omicron outbreak in both NSW (0.96; 95% CI 0.93, 1.00) and Victoria (0.96; 95% CI 0.92, 1.00), and significant reductions in endocrine therapy initiation in both states (NSW IRR 0.85; 95% CI 0.74, 0.99. Victoria IRR 0.78; 95% CI 0.65, 0.94) (Figure 2, Supplementary Figure C, available from: doi.org/10.6084/m9.figshare.c.7040453). There was no change to the discontinuation rate in NSW during the Omicron outbreak, but we observed a significant reduction in Victoria (0.88; 95% CI 0.78, 0.98).
Supportive medicines
In NSW during the 2021 lockdowns, there was a trend towards a reduced dispensing rate for supportive cancer medicines (0.97; 95% CI 0.93, 1.00) (Figure 2, Supplementary Figure C, available from: doi.org/10.6084/m9.figshare.c.7040453), and significant reductions to the rates of initiation (0.90; 95% CI 0.85, 0.95) and discontinuation (0.88; 95% CI 0.86, 0.96). In both NSW and Victoria during the 2021 lockdowns, we observed significant reductions in the dispensing rates of ondansetron and other serotonin antagonists (used for chemotherapy- and radiotherapy-related nausea), and significant reductions in the initiation and discontinuation rates of ondansetron (Figure 2). During the Omicron outbreak, there was a significant increase in the dispensing rate of ondansetron in Victoria (1.11; 95% CI 1.01, 1.23), and significant decreases in the rates of initiation (0.81; 95% CI 0.69, 0.95) and discontinuation (0.83; 95% CI 0.72, 0.96) of ondansetron in NSW.
Discussion
Our population-based, controlled interrupted time series study showed that the 2021 lockdowns and 2021/22 Omicron outbreaks in NSW and Victoria resulted in significant changes to the dispensing, initiation and discontinuation of systemic cancer therapies in those states relative to other states that did not experience prolonged periods of lockdown. However, the overall impacts of these changes, which were primarily reductions, were minimal. Systemic cancer treatment in Australia during the COVID-19 pandemic (2020–early 2022) does not appear to have been dramatically impacted by the virus or the policy responses to the pandemic.10
The reasons for the minimal impacts of the 2021 lockdowns and Omicron outbreaks are not clear but may be related to each states’ experiences of the 2020 COVID-19 outbreaks and lockdowns. During 2020, strategies were established to mitigate the effects of both the pandemic and the policy responses to the pandemic.10 This may have resulted in both patients and clinicians feeling more comfortable continuing and initiating treatment during 2021. While we found that initiation rates of oral targeted therapies decreased by 50% in NSW during the 2021 lockdown, in absolute terms this decrease represents a change of just 0.8 people/100 000/week. Decreases in rates of ondansetron discontinuation were more likely related to the reductions in rates of dispensing. These reductions, in turn, were likely due to a reduction in gastroenteritis cases resulting from reduced community circulation during lockdown.10 Ondansetron is PBS-listed for chemotherapy- and radiotherapy-related nausea, however, it is often accessed via the PBS for gastroenteritis.
Patients and the general community might have also been less fearful of medicine shortages or significant disruptions to treatment compared to 2020. For instance, in March 2020, many Australian patients filled endocrine therapy prescriptions in classic stockpiling behaviour10,15, but the dispensing and initiation of endocrine therapies were unaffected by the 2021 NSW and Victoria lockdowns. Following the experience of 2020, the level of concern associated with lockdowns might not have been strong enough to provoke the same behaviour in 2021.
We observed more changes to antineoplastic dispensing rates during the Omicron outbreak than the 2021 lockdowns. Extrapolating our results from a 10% sample to the wider Australian population, during the first week of 2022, there were approximately 600 and 1500 fewer parenteral chemotherapies dispensed than expected in Victoria and NSW, respectively. These changes might have been due to increased COVID-19 infections among Australians undergoing or beginning cancer treatment. Infections, both symptomatic and asymptomatic, were far more widespread during the Omicron outbreak than during earlier stages of the pandemic, and it is common clinical practice to delay cancer treatment until recovery from COVID-19 infection.16 Moreover, widespread infection through the general community led to high rates of furloughing of healthcare workers, which might have affected the capacity of the health system to deliver cancer treatment as scheduled.
It is also possible that a level of fear among patients and/or the general community might have been a factor affecting changes to systemic cancer therapy during the Omicron outbreak. Fear has long been acknowledged as an important factor in motivating public behaviour changes in response to public health crises17,18, and there is precedent in Australia for media-spurred concerns changing prescription medicine use.19 When the Omicron outbreak began at the end of 2021 – coinciding with the end of lockdowns in both NSW and Victoria – the case numbers reported in the media daily were orders of magnitude larger than those that had been reported to that point. There were many calls in the media to reimpose lockdowns20, mobility in the community abruptly declined11, and, as we found here, initiation of endocrine therapies also declined in NSW and Victoria.
The end of the calendar year/beginning of the new year coincides with several public holidays and the start of the Australian summer. It is a time when cancer treatment is often reduced or postponed, so we should expect reductions in our study outcomes during this time. However, the controlled time series analyses we employed – using as a control series the Australian states that did not experience restrictive lockdowns or large Omicron outbreaks in December 2021 – highlights that the reductions in dispensing and treatment initiation we observed during this time were beyond what was expected.
During 2020, many cancer screening programs were shut down1,21, and cancer diagnoses subsequently fell below levels from previous years.22 We did not find evidence of increased levels of systemic cancer therapy during 2021 as a result of diagnoses delayed from 2020, although the decreases in endocrine therapy initiation rates during the Omicron outbreak may reflect reduced breast cancer diagnoses. In response to the 2021 lockdowns in NSW and Victoria, cancer screening programs were again closed, and it is likely that there are a considerable number of cancer cases yet to be diagnosed. These will likely be cancers diagnosed at later stages, where systemic therapies are likely to be used as treatments, so we expect treatment numbers to increase from late 2022 onwards.
While the impacts of cancer screening closures remain to be seen, at a population level, the COVID-19 pandemic had little impact on systemic cancer treatment in Australia. Most of the changes detected in our study, while statistically significant, at most affected only a handful of patients on a weekly basis. The largest change we observed was the stockpiling of endocrine therapies during the initial outbreak of COVID-19 in March 2020. This event was transitory, but health systems planners and policy makers should consider the potential for shortages of highly specialised and critical medicines in future pandemic response planning. We saw a similarly transitory effect with the substantial decrease in antineoplastic medicine dispensing that occurred at the onset of the Omicron outbreak. While our dispensing data do not allow us to fully know what caused this sharp decline, pandemic response planning should consider second-order impacts in critical health areas. Striking a balance between maintaining an appropriate level of public concern, protecting patients and healthcare professionals from immediate risks, and ensuring essential treatment continues will always be challenging. Robust information channels between public health officials, healthcare professionals and patients are essential, as are policies that facilitate changed modes of care. The rapid rollout of Medicare Benefit Schedule (MBS) item numbers to enable government-subsidised telephone and video consultations reduced infection risk for patients and healthcare staff while allowing continuity of care, and planners and policymakers should consider embedding telehealth provisions to improve ongoing access and facilitate essential cancer care in future pandemics.
Strengths and limitations
Our study used large, nationally representative data comprising dispensing records for 10% of the PBS-eligible Australian population. The PBS funds all government-subsidised medicines dispensed through community pharmacies and hospital outpatient oncology units, which is where the overwhelming majority of systemic cancer therapy is administered in Australia; this means that our data provide a comprehensive picture of treatment in NSW and Victoria. This is contrary to many jurisdictions with universal healthcare systems and is a strength of our study. These data contain medicines subsidised through the PBS – they do not contain medicines that patients may have received through compassionate access schemes or clinical trials, although we estimate the dispensing numbers for these medicines during the study period would have been very small. These data also do not contain clinical information, and we cannot comment on whether the changes we observed may be due to tumour type, stage or adverse events. Finally, our analyses comprise multiple statistical tests, and it is theoretically possible that some significant results were due to chance. While CITS is widely considered to be one of the strongest quasi-experimental designs12,13, it is only capable of controlling for time-varying and cointervention confounds that are observed in both the target and control series. There may be factors other than lockdown measures that affected the local populations of NSW and Victoria that were not reflected in the other states and that could have simultaneously impacted cancer treatment (e.g. media reports).
Conclusion
Our findings suggest that Australian systemic cancer treatment was robust in the face of a challenging COVID-19 year. Compared to states that did not experience long lockdown periods, the 2021 lockdowns and 2021/2022 Omicron outbreaks in NSW and Victoria resulted in significant changes to the dispensing, initiation and discontinuation of systemic cancer therapies and associated supportive medicines in those states. However, the overall impact of these periods on systemic cancer treatment in Australia was minimal. The public health policy of lockdowns appear to have had less significant impacts than the Omicron outbreaks, suggesting that COVID-19 infection, health system capacity, and general patient and community concerns are important factors for influencing treatment changes.
Acknowledgements
This research was supported by a UNSW Cancer Seed Grant and the National Health and Medical Research Council Centre of Research Excellence in Medicines Intelligence (ID: 1196900). BD is supported by a Cancer Institute NSW Early Career Fellowship (ECF1381). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
We acknowledge Services Australia for providing the PBS data we used in our study.
Author contributions
SAP and BD were responsible for equal contributions as senior authors All authors were involved in the study design. FK and BD were involved in data preparation and analysis. All authors were involved in writing and revising the manuscript. All authors read and approved the final manuscript.
Competing interests
FK has performed consulting work for BCAL Diagnostics within the past 3 years. BCAL Diagnostics did not have any knowledge of, or involvement in, the present study. SAP is a member of the Drug Utilisation Sub Committee of the Pharmaceutical Benefits Advisory Committee, an independent statutory body that advises the Australian Government on the PBS. The views expressed in this paper do not represent those of the Committee. All remaining authors have declared no conflicts of interest relevant to the submitted work. SAP is a member of the PHRP Editorial Board. She was not involved in reviewing, or in the decision to publish, this manuscript.
References
1 Fujisawa R. Impact of the COVID-19 pandemic on cancer care in OECD countries. US: OECS; 2022 [cited 2024 Jan 15]. Available from: www.oecd.org/els/impact-of-the-covid-19-pandemic-on-cancer-care-in-oecd-countries-c74a5899-en.htm
8 Australian Institute of Health and Welfare. The first year of COVID-19 in Australia: direct and indirect health effects. Canberra; AIHW; 2021 [cited 2024 Jan 15]. Available from: www.aihw.gov.au/getmedia/a69ee08a-857f-412b-b617-a29acb66a475/aihw-phe-287.pdf?v=20230605184353&inline=true
9 Knowlton C. A timeline of Covid-19 in Australia, two years on. Melbourne, Victoria: TimeOut; 2023 [cited 2024 Jan 15]. Available from: www.timeout.com/melbourne/things-to-do/a-timeline-of-covid-19-in-australia-two-years-on
11 Google Inc. COVID-19 Community mobility reports. US: Google; 2022 [cited 2023 19 Mar]. Available from: www.google.com/covid19/mobility
14 R Core Team. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2018 [cited 2024 Jan 30]. Available from: www.R-project.org/
16 Cancer Australia. Cancer care for cancer patients with COVID-19 NSW: Australian Government Cancer Australia; 2022 [cited 2023 Jun 06]. Available from: www.canceraustralia.gov.au/covid-19/guidance-health-professionals/cancer-care-cancer-patients-covid-19





