112 EFFECT OF EGG YOLK CONCENTRATION IN TEST BUFFERED EXTENDER ON SURVIVAL AND IN VITRO FUNCTIONALITY OF DOMESTIC CAT EPIDIDYMAL SPERMATOZOA FOLLOWING CRYOPRESERVATION
J. R. Saenz A , C. Dumas B , B. L. Dresser B , M. C. Gómez B , R. A. Godke A and C. E. Pope BA Louisiana State University Agriculture Center, Baton Rouge, LA, USA;
B Audubon Center for Research of Endangered Species, New Orleans, LA, USA;
C University of New Orleans, New Orleans, LA, USA
Reproduction, Fertility and Development 22(1) 214-215 https://doi.org/10.1071/RDv22n1Ab112
Published: 8 December 2009
Abstract
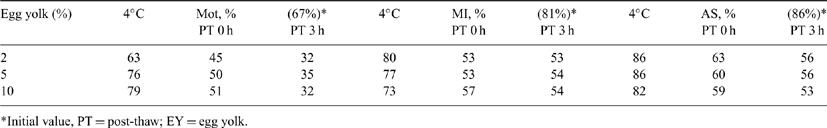
Our purpose was to examine the effect of egg yolk concentration (EY; 2, 5, or 10%) on in vitro survivability and functionality of cat epididymal spermatozoa cryopreserved in TEST-buffered extender (TYB). Testes were transported in HEPES saline; epididymes were dissected in HEPES 199 medium (He199) and repeatedly sliced. The sperm suspension was filtered (40 μ), layered onto a density gradient column (Isolate®, Irving Scientific, Santa Ana, CA, USA), and centrifuged at 650 g for 20 min. Aliquots of the sperm pellet were extended in TYB containing 2, 5, or 10% EY. After cooling to 4°C, samples were diluted 1:1 with TYB containing 2, 5, or 10% EY + 12% glycerol in 4 steps as modified from Gao DY et al. Hum. Reprod. 1995 10, 1109-1122. Then, samples were loaded into 0.25-mL straws, sealed, and frozen on a dry ice block (-80°C) for 20 min before storage in LN2. Straws were thawed by exposure to air (˜22°C) for 5 s and immersion in a 60°C water bath for 5 s. Samples were diluted by addition of He199 in 7 steps as modified from Gao DY et al. Hum. Reprod. 1995 10, 1109-1122 centrifuged at 200g for 10 min, and pellets resuspended in He199. Motility (Mot, phase contrast, 37°C), membrane integrity (M.I., SYBR 14-PI), and acrosomal status (A.S., FITC-PNA) were evaluated at 0 h, after gradual cooling to 4°C and after freezing at 0 and 3 h post-thaw (37°C). Ten replicates were done. Cumulus oocyte complexes (COC) were placed in modified TCM-199 and cultured for 24 h in 5% O2, 5% CO2, and 90% N2 at 38°C (IVM). For IVF, COC were co-incubated with spermatozoa frozen in TYB + 2% egg yolk or HSPM (no egg yolk) in droplets (1 million sperm/mL) of IVF medium under 5% CO2 in air at 38°C. After 18 h, oocytes were rinsed and cultured until blastocyst development was evaluated (Day 8). There were no treatment differences at any time or temperature point for the 3 sperm characteristics evaluated (one-way ANOVA; P > 0.05). As shown in the Table 1, at 0 h post-thawing, sperm in each group retained ˜70% of their initial pre-freeze motility. After 3 h of post-thaw incubation, motility decreased to ˜50% of the pre-freeze value. Cooling to 4°C did not affect membrane integrity or acrosomal status, but post-thaw values were reduced by 30-35% as compared with pre-freeze. Cleavage frequency and blastocyst development of 284 IVM oocytes after IVF using sperm frozen in TYB + 2% EY and HSPM were 53 v. 52% and 42 v. 38%, respectively (P > 0.05). In summary, we have shown that cat epididymal spermatozoa can be frozen successfully in a cryoprotectant solution containing minimal egg yolk (2%).
|


