52 EFFECT OF TRICHOSTATIN A ON DEVELOPMENTAL POTENTIAL OF INTER-SPECIES CLONED GAUR (BOS GAURUS) EMBRYOS
K. Srirattana A , C. Laowtammathron A , R. Devahudi A , S. Imsoonthornruksa A , A. Sangmalee A , W. Tunwattana B , C. Lorthongpanich A , N. Sripunya A , K. Keawmungkun A , W. Phewsoi A , M. Ketudat-Cairns A and R. Parnpai AA Embryo Technology and Stem Cell Research Center, Suranaree University of Technology, Nakhon Ratchasima, Thailand;
B Khao Kaew Open Zoo, Chonburi, Thailand
Reproduction, Fertility and Development 21(1) 126-126 https://doi.org/10.1071/RDv21n1Ab52
Published: 9 December 2008
Abstract
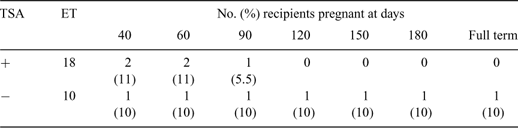
This study was carried out to investigate the effect of trichostatin A (TSA) treatment on interspecies cloned gaur (Bos gaurus) embryos development and implantation rate after transfer to bovine (Bos taurus) recipients. The bovine (Bos taurus) enucleated oocytes were used as recipient cytoplasm for male and female gaur fibroblasts. After electrical fusion, oocytes were separated into two groups, TSA treatment and control. For the TSA group, the oocytes were placed in EmCare (ICPbio, Ltd., Auckland, New Zealand) holding medium + 50 nm TSA for 1 h. The fused oocytes were activated by 7% ethanol + 50 nm TSA for 5 min at room temperature and 10 μg mL–1 cycloheximide + 1.25 μg mL–1 cytochalasin D + 50 nm TSA at 38.5°C under 5% CO2 in air for 5 h. Then the embryos were cultured in mSOFaa medium + 3 mg mL–1 bovine serum albumin (BSA) + 50 nm TSA up to 10 h. After 10 h, the reconstructed embryos were transferred to embryo culture medium without TSA and culture for 2 days at 38.5°C under 5% CO2, 5% O2, 90% N2. The control embryos were cultured with the same culture system without TSA supplementation. Eight-cell stage embryos were selected and co-cultured with bovine oviductal epithelial cells in culture medium at 38.5°C under 5% CO2 in air for 5 days. Half volume of the culture medium was replaced daily. Two blastocysts at days 7 or 8 derived from male fibroblasts of treated and non-treated TSA were non-surgically transferred to each synchronized estrous bovine recipients. The statistical analysis was done by ANOVA and the comparison of means by Duncan’s Multiple Range Test (DMRT). The development to blastocyst stage was not different among male and female, treated and non-treated TSA embryos which range between 34.8 to 39.3%. The pregnancy rate at 40 days after recipients received cloned embryos derived from male fibroblasts treated v. non-treated TSA was 11% (2/18) v. 10% (1/10) (Table 1). One recipient which received a non-treated embryo gave birth by C-section on March 4, 2008. The male gaur calf died from respiratory problem at 12 h after birth. Eight bovine microsatellite markers analysis confirmed that the newborn gaur was derived from the donor gaur fibroblast. In this study, TSA has no effect on pre-implantation cloned gaur embryos development either derived from male or female gaur fibroblasts. Cloned gaur calves could be produced by interspecies cloning using bovine oocytes as recipient cytoplasm.
|
This study was supported by National Center for Genetic Engineering and Biotechnology (BIOTEC) and Suranaree University of Technology.


