Assessment of multiple paternity in spinner shark (Carcharhinus brevipinna) litters from eastern Australian waters
Alicia K. Linn A * , Alexis L. Levengood A , Christine L. Dudgeon B , Johan A. Gustafson A C , Julia L. Smith
A * , Alexis L. Levengood A , Christine L. Dudgeon B , Johan A. Gustafson A C , Julia L. Smith  D and Bonnie J. Holmes A
D and Bonnie J. Holmes A
A
B
C
D
Abstract
The spinner shark (Carcharhinus brevipinna) is a medium-sized, coastal species distributed across the warm waters of all ocean basins. Knowledge of this species’ reproductive biology is limited to general parameters such as reproductive mode and average litter sizes, and very little is known about their reproductive strategy.
Here, we provide an assessment of multiple paternity, using single nucleotide polymorphisms.
DNA was extracted from 2 gravid females and the 18 pups between their 2 litters captured off the Australian eastern coast. Sibship analyses were conducted to assess multiple paternity.
For the first time globally, multiple paternity was confirmed in this species in both litters examined.
This study confirms that C. brevipinna employs some degree of polyandry as part of its mating system.
This research contributes to the growing number of studies regarding multiple paternity in elasmobranchs and provides new information on the reproductive biology of this species.
Keywords: Carcharhinus brevipinna, elasmobranch, genetics, mating system, multiple sires, polyandry, reproductive biology, shark, single nucleotide polymorphisms, spinner shark.
Introduction
All elasmobranchs exhibit internal fertilisation, but their reproductive modes (characterised by embryo development) range from oviparity (egg cases laid externally) to a variety of forms of viviparity (birth of live offspring). The structure and function of these have been well documented (Conrath and Musick 2012; Klimley 2013); however, the relationship among ecology, reproductive mode and reproductive strategy for most species is less understood (Katona et al. 2023; Deeken et al. 2024; Mull et al. 2024). Reproductive strategies reflect morphological, physiological and behavioural adaptations that can serve to enhance reproductive success (Coleman and Jones 2011). Further, a species’ capacity to respond to selection pressures, such as overfishing, may be dependent on the genetic diversity of the population exposed to those extractive pressures (Frankham 2005; Rossouw et al. 2016). Knowledge of the intricacies of mating systems and reproductive strategies can be useful in effectively understanding potential risk factors and may be applied in population assessments (e.g. Traylor-Holzer 2021). For example, in smaller populations, reproductive parameters and mating systems can be expected to influence overall population viability (Legendre 2004; Daly-Engel et al. 2006; Byrne and Avise 2012). Therefore, gaining an understanding of the reproductive strategies across the mating system spectrum, including polyandry (the act of mating with multiple males), sperm storage and paternity of single litters by multiple sires (hereafter referred to as multiple paternity) is needed (Daly-Engel et al. 2006, 2010; Conrath and Musick 2012).
Multiple paternity appears to be prevalent among elasmobranchs, having been reported in both viviparous (placental and aplacental) and oviparous species (reviewed in Fitzpatrick et al. 2012, Rossouw et al. 2016 and Lamarca et al. 2020). It has been suggested that multiple paternity may be an ancestral trait among elasmobranchs (Lamarca et al. 2020), facilitated by female sperm storage in the oviducal gland (Klimley 2013). Sperm storage is common among elasmobranchs and can persist from several weeks to years (Conrath and Musick 2012; Fitzpatrick et al. 2012). Although polyandry can be observed behaviourally, multiple paternity can be confirmed only by analysing the genotypes from mother and offspring to infer the minimum number of sires per brood (Daly-Engel et al. 2010). Both interspecific and intraspecific variation in the frequency of multiple paternity has been documented among elasmobranchs, albeit the mechanisms and potential evolutionary benefits of multiple paternity as a reproductive strategy remain unclear (reviewed in Byrne and Avise 2012, Fitzpatrick et al. 2012, Rossouw et al. 2016 and Lamarca et al. 2020).
Several hypotheses are circulated in the literature regarding the adaptive significance of multiple paternity in elasmobranchs (Fitzpatrick et al. 2012). First, polyandry is suggested to be beneficial to males because their reproductive success may be dependent on the number of inseminations (Byrne and Avise 2012). However, the cost of polyandry is more skewed towards females, because they are susceptible to injuries, infections and decreased fitness from copulation (Daly-Engel et al. 2006, 2007, 2010), because males will typically seize the flanks and pectoral fins of females with their teeth (Klimley 2013). Therefore, it has been hypothesised that female elasmobranchs may engage in what is referred to as ‘convenience polyandry’ (Daly-Engel et al. 2010); suggesting that multiple paternity may arise as a result of sexual conflict avoidance, rather than benefitting the females directly (Daly-Engel et al. 2010). This theory may also account for variation in frequency of multiple paternity observed between conspecifics (Daly-Engel et al. 2010). Female choice has been suggested as a mechanism of multiple paternity, both pre- and post-copulation, as females may engage in polyandry to increase the chance of fertilisation by a viable male, or the storage of sperm from multiple males may promote competition of sperm within the oviducal gland (Byrne and Avise 2012). Finally, it has also been proposed that the frequency of multiple paternity increases with the rate of encounters between male and female conspecifics (the ‘encounter rate theory’) (Daly-Engel et al. 2007, 2010). Hence, assuming no direct or indirect benefits to the sexes, higher rates of multiple paternity would be more likely among schooling species or species that aggregate during the mating season. Whereas the adaptive significance of multiple paternity is still deliberated, the number of studies documenting multiple paternity in elasmobranchs continues to grow (reviewed in Byrne and Avise 2012, Fitzpatrick et al. 2012, Rossouw et al. 2016 and Lamarca et al. 2020).
The spinner shark (Carcharhinus brevipinna) is a cosmopolitan coastal and pelagic schooling species of carcharhinid, predominantly found in warm–temperate and tropical waters near continental shelves (Capapé et al. 2003; Geraghty et al. 2013; Rigby et al. 2020). Like many carcharhinids, C. brevipinna is viviparous, and produces litters of between 3 and 14 pups every 2 years following a lengthy gestation period of 12 months (Allen and Cliff 2000; Joung et al. 2005; Geraghty et al. 2016; Deeken et al. 2024). However, little is known about its reproductive strategy, such as whether the species can store sperm and produce litters sired by multiple males, similarly to other carcharhinids (Rossouw et al. 2016). Here, we provide the first assessment of multiple paternity in C. brevipinna globally by genotyping two mother–litter groups from the Australian eastern coast, through the application of single nucleotide polymorphisms (SNPs). As a commercially harvested species throughout much of its range, gaining a more robust understanding of this species’ reproductive biology is prudent.
Materials and methods
Study locations and sampling
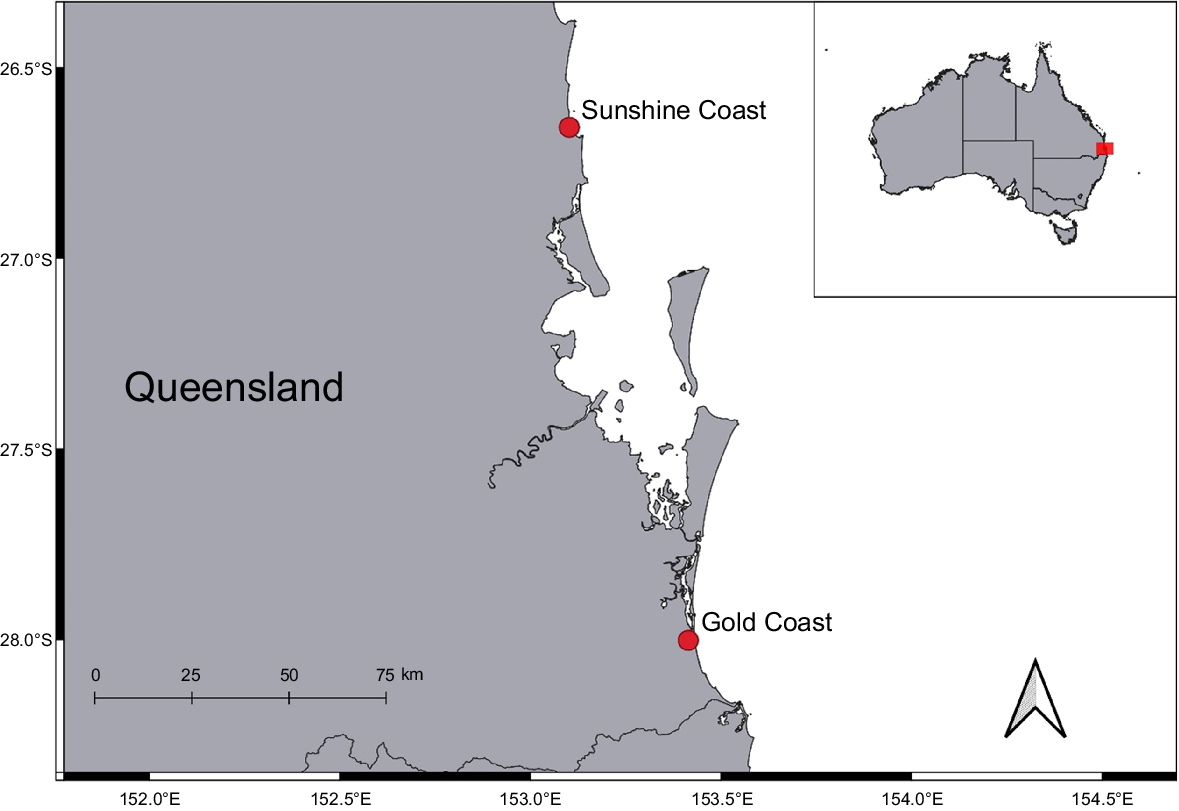
Tissue samples (fin clips and liver tissue) were obtained from two mother–litter groups of C. brevipinna to be assessed for multiple paternity. The two gravid females were caught in the Queensland Shark Control Program (QSCP) between 2020 and 2021. The pregnant females were captured in shark nets (gillnets) off the Gold Coast and the Sunshine Coast in south-eastern Queensland, Australia (Fig. 1). Full litters were made available, except for a single female pup retained by the Gold Coast Shark Control contractors (Table 1). Thus, genotypes were obtained for a total of 18 embryos (12 from the Gold Coast and 6 from the Sunshine Coast), as well as from the 2 mothers. All samples were stored in 80% ethanol and frozen at −80°C until laboratory processing. Specimens were retained under Queensland General Fisheries permit number 208761, and the University of the Sunshine Coast’s Animal Ethics Committee approval number ANE2161.
Capture locations of spinner shark (Carcharhinus brevipinna) mother–litter groups (n = 2) caught in the Queensland Shark Control Program (QSCP) in south-eastern Queensland, Australia, between February 2020 and February 2021.
| Item | Litter 1 | Litter 2 | |
|---|---|---|---|
| Capture date | 4 February 2020 | 19 February 2021 | |
| Capture location | Gold Coast | Sunshine Coast | |
| Number of pups in litter | 13 | 6 | |
| Sex ratio, F:M | 2.25 | 0.2 | |
| Mother size (cm TL) | 289 | 200 | |
| Mean pup size (cm TL) s.e. | 80.09 0.2 | 13.13 0.2 |
TL, total length.
DNA extraction and SNP genotyping
Genomic DNA extraction was conducted using QIAGEN DNeasy Blood and Tissue extraction kits following standard protocols (QIAGEN 2020). DNA was successfully extracted from the 20 individuals (18 embryos and 2 mothers) and sent to Diversity Arrays Technology (DArT, Canberra, ACT, Australia) for genotyping. DArT processed the DNA isolates by using their trademarked DArTseq protocol to generate SNP data. This is a genotyping-by-sequencing approach that involves reducing complexity of the genome through digestion using refined restriction enzymes (PstI and SphI), and next generation sequencing of selected size fragments (Sansaloni et al. 2011; Kilian et al. 2012).
SNP filtering
A preliminary SNP dataset received from DArT was imported into R (ver. 4.2.1, R Foundation for Statistical Computing, Vienna, Austria, see https://www.r-project.org/) as a genlight object using the dartR package (ver. 2.0.4, see https://cran.r-project.org/web/packages/dartR; Gruber et al. 2018). To mitigate errors and maintain a dataset with minimal missing data, stringent filtering criteria were applied. The filtering pipeline proceeded with the removal of monomorphic loci, retaining loci with a repeatability of >0.9, retaining loci with a call rate threshold of 0.9, removal of secondaries, and retaining loci with a read depth between 5 and 50. The dataset was then filtered at a moderate minor allele frequency (MAF) threshold of 0.3 (e.g. Premachandra et al. 2019). Alternative filtering stringencies were also tested showing the same results and, thus, the aforementioned filtering pipeline was considered to be most appropriate to obtain a final dataset with a robust number of SNPs and minimal missing data. Finally, loci showing a significant departure from Hardy–Weinberg equilibrium (HWE) proportions were filtered at two significance levels (0.01 and 0.001) to assess variation in stringency and ensure the loci segregated according to Mendelian expectations.
Sibship analysis
To determine the most likely number of paternal genotypes for each litter, a sibship analysis was performed using likelihood assignment methods, implemented in COLONY (ver. 2.0.6.8, see https://www.zsl.org/about-zsl/resources/software/colony; Jones and Wang 2010; Wang 2018). To address the potential genotyping errors in the SNP data, two allelic dropout rates were tested (0.25 and 0), which approximately correspond to sequencing coverages of 4 and 10 (Wang 2019). Additionally, two false allele error rates were tested (0.02 and 0.1). Therefore, the combination of significance thresholds for HWE, allelic dropout rates and false allele error rates resulted in eight COLONY input files that were analysed for each litter (16 total). The two litters were analysed separately, and the genotypes of the pups and their known mother were included in the sibship analysis, with this known maternal relationship being specified in the input file. Within all input files, the following parameters remained consistent. The full-likelihood method was selected, with medium precision, and medium run length. The dioecious and diploid options were selected, and a polygamous mating system was selected for both males and females. Allele frequencies were not updated, no sibship prior was selected, sibship scaling was set to the default, the seed for the random number generator was set to 1234 and markers were noted as co-dominant. COLONY reports two likelihood probabilities to determine whether full-sibling groups have not been over- or underestimated in the analysis. The inclusion probability (Prob(Inc.)) describes the probability that individuals assigned to an inferred sire are actual full-siblings, and the group is not under-split. The exclusion probability (Prob(Exc.)) describes the probability that all individuals assigned to an inferred sire are full-siblings and the group has not been over-split. Reporting higher values for both metrics indicates that the number of full-sibling groupings (i.e. the number of inferred sires) is not over- or under-split.
Results
SNP filtering
In total, 30,569 SNP loci were derived from the DArTseq analysis. Stepwise filtering, with the additional filtering for departure from HWE at significance thresholds of 0.01 and 0.001 resulted in final datasets of 377 and 567 loci respectively. These datasets consisted of only 1.62 and 3.26% missing data. Most loci were excluded owing to departure from HWE (Supplementary Table S1).
Sibship analysis
The sibship analyses conducted in COLONY confirmed the presence of multiple paternity in both litters of C. brevipinna assessed in this study. Of the 16 input files analysed, the results remained consistent, yielding the same five full-sibling groupings for the Gold Coast litter from all corresponding input files (Tables 2, S2) with no evidence of over- or under-splitting. Of the eight input files corresponding to the Sunshine Coast litter, five produced the same three full-sibling groupings, with the remaining analyses producing two (Table S3). Lower exclusion probabilities were reported in the outputs indicative of three full-sibling groupings and, thus, the overall consensus is that there are two clear full-sibling groups in the Sunshine Coast litter (Table 2). Hence, reported here are the results obtained with the application of lower error rates for allelic dropout (0), and false alleles (0.1) to the less conservative dataset of 567 loci (HWE = 0.001).
| Litter | Inferred sire or full-sibling group | Prob (inc.) | Prob (exc.) | Pups | |
|---|---|---|---|---|---|
| Litter 1 | S1 | 0.9988 | 0.9988 | P1(F), P5(F), P6(M) | |
| S2 | 1.0000 | 1.0000 | P10(M), P4(F) | ||
| S3 | 1.0000 | 0.9988 | P11(F) | ||
| S4 | 0.9636 | 0.9636 | P12(M), P2(F), P3(F) | ||
| S5 | 1.0000 | 1.0000 | P7(F), P8(M), P9(F) | ||
| Litter 2 | S1 | 0.9707 | 0.9707 | P1(M), P2 (M), P4(M), P6(M) | |
| S2 | 1.0000 | 1.0000 | P3(M), P5(F) |
The inferred sire, inclusion probability (Prob (inc.)), exclusion probability (Prob (exc.)), and assigned names and sexes (M, male; F, female) of the pups clustered in each full-sibling group are presented.
Discussion
The results of this study showed novel insights into the reproductive biology of C. brevipinna. Through the application of SNP genotyping and subsequent sibship analyses, we have provided the first verification of multiple paternity in this species and can, thus, infer that C. brevipinna employs polyandry as part of its mating system. Although our results show clear evidence of multiple paternity in the two litters examined, we could not determine the frequency at which polyandry occurs in this species because of the small sample size. Given that intraspecific variation in frequency of multiple paternity has been observed in other carcharhinids (Byrne and Avise 2012; Lamarca et al. 2020), and the potential associated population-level implications (Hoekert et al. 2002; Byrne and Avise 2012), further sampling and examination of this strategy for C. brevipinna across populations is prudent.
Of the 13 carcharhinid species examined, 11 have been reported to exhibit some degree of multiple paternity (Lamarca et al. 2020; Lyons et al. 2021; Nash et al. 2021; Armada-Tapia et al. 2023), with the exceptions being the Galapagos shark (Carcharhinus galapagensis; Daly-Engel et al. 2006) and the tiger shark (G. cuvier; Holmes et al. 2018; Pirog et al. 2020). The lack of multiple paternity observed for C. galapagensis comes with the caveat that only a single litter was assessed. However, the lack of multiple paternity in G. cuvier remained consistent across the eight litters examined between the two studies conducted in different ocean basins. This observation may be tied to the species’ reproductive mode (aplacental viviparity) and semi-solitary nature, which are traits that are relatively atypical of the Carcharhinidae. Hence, the detection of multiple sires in C. brevipinna litters here is consistent with existing literature on multiple paternity in most carcharhinids.
Convenience polyandry
As male reproductive success theoretically increases with the number of successful mating events, polyandrous mating may be male-mediated (Byrne and Avise 2012). However, matings imposed on females often result in physical damage to flanks, dorsal and pectoral fins as males attempt copulation (Klimley 2013). Coercive strategies such as mobbing and herding have been documented in nurse sharks (Ginglymostoma cirratum), where multiple males will cooperatively harass a female into submission (Pratt and Carrier 2001). The effects of this conflict may be compounded in species that may be sexually segregated, owing to coercive behaviours exhibited by male-biased aggregations. Owing to the increased physical costs of polyandry to females, the theory of convenience polyandry suggests that females may submit to the advances of multiple males to avoid the adverse outcomes of sexual conflict, such as injury, infection or decreased fitness (Daly-Engel et al. 2006, 2007, 2010). This idea has been corroborated in several studies that also documented female sharks’ induction into ‘tonic immobility’ during courtship, a reversible catatonic state characterised by a motionless posture and unresponsiveness to the external noxious stimuli or injury (Williamson et al. 2018). As wild observations of copulation and potential breeding aggregation behaviours are absent for C. brevipinna, we were unable to infer whether convenience polyandry may contribute to the occurrence of multiple paternity in this species at this time. As this is a consistent issue in attempting to understand the reproductive strategy of elasmobranchs, and especially the vagile carcharhinids, many studies instead have given increased thought to the post-copulatory mechanisms, which may potentially influence the manifestation of multiple paternity in these species (Daly-Engel et al. 2010; Fitzpatrick et al. 2012; Green et al. 2017; Lamarca et al. 2020; Lyons et al. 2021).
Conspecific encounters and female choice
A heavily cited hypothesis for the prevalence of multiple paternity in elasmobranchs is the ‘encounter rate theory’, which proposes that multiple paternity may be dependent on the number of potential mates a receptive female encounters during a breeding season (Daly-Engel et al. 2007, 2010). This theory suggests that increased rates of multiple paternity would be likely to occur in high-density populations, such as schooling or aggregating species. Under this idea, the detection of multiple paternity in C. brevipinna could simply be attributed to the species’ active schooling behaviour (Compagno 1984; Ebert et al. 2021). Similarly, considerably high rates of multiple paternity have been attributed to aggregatory behaviours in common smoothhound sharks (Mustelus mustelus) and finetooth sharks (Carcharhinus isodon) (Rossouw et al. 2016; Nash et al. 2021). The rate of encounter between females and males will invariably fluctuate in time and space, and between species and populations (Rossouw et al. 2016). Extraneous factors such as climate-driven shifts in habitat use and fisheries-related extractions may also affect population density and sex ratios, shifting the prevalence and detection rate of multiple paternity at different times.
Lengthy gestation periods, and the baring of large, live young, are energetically expensive to viviparous species such as C. brevipinna. Further, the species typically exhibits low fecundity, and the gestation period of 12 months results in offspring production once every 2 years (Allen and Cliff 2000; Joung et al. 2005; Geraghty et al. 2016). As such, it may be more beneficial for C. brevipinna females to engage in polyandrous mating, allowing them to optimise their potential reproductive output and minimise the chances of fertilisation by inadequate males (Jennions and Petrie 2000). The increased opportunity to mate with a viable male (or males) may outweigh the physical implications posed by multiple copulations, because the lifetime reproductive output potential of species with these life-history traits may be more constrained otherwise (Lyons et al. 2021). This notion may explain the prevalence of multiple paternity identified in species that exhibit life-history and reproductive parameters similar to those of C. brevipinna, such as C. plumbeus (Daly-Engel et al. 2006, 2007; Portnoy et al. 2007), C. isodon (Nash et al. 2021), bull shark (Carcharhinus leucas; Pirog et al. 2019) and blue shark (Prionace glauca; Armada-Tapia et al. 2023).
The notion of cryptic female choice has been discussed in some literature because post-copulatory investment is heavily skewed towards females (Conrath and Musick 2012; Fitzpatrick et al. 2012; Lyons et al. 2017, 2021). In instances where receptive females cannot evaluate the fitness or compatibility of potential mates, multiple maternity may be facilitated by some degree of female choice (Neff and Pitcher 2005). In C. brevipinna, we identified a considerable number of putative sires per litter in both the larger litter (Gold Coast, Npups = 12, Nsires = 5) and the smaller litter (Sunshine Coast, Npups = 6, Nsires = 2). Notably, the paternal skew in the Sunshine Coast litter, where one male sired four pups, and the other sired two, may be an indication of female selectivity; perhaps a result of biased sperm utilisation or the biochemical promotion of sperm competition within the oviducal glands (Jennions and Petrie 2000; Byrne and Avise 2012; Lyons et al. 2021).
Sperm storage
Whereas multiple paternity is thought to be facilitated by females’ capacity to store sperm and enact potential selection biases (Conrath and Musick 2012; Fitzpatrick et al. 2012; Lyons et al. 2021), sperm storage remains understudied in most carcharhinids, including C. brevipinna. One unpublished study conducted a histological evaluation of female C. brevipinna oviducal glands, and reported no evidence of sperm storage (Palmrose 2021). However, the study was limited to using historical tissues (preserved between 2003 and 2005), and potential degradation of the samples was not discussed. Notwithstanding, it is plausible that these findings may be attributed to the species’ schooling nature and implied access to conspecifics, because sperm storage may not be required as a pre-emptive measure for fertilisation in the presence of regular, repeated fresh inseminations. Lamarca et al. (2020) determined that the ability to store sperm in elasmobranchs was not a necessary pre-cursor for multiple paternity to occur in subsequent litters. In a more recent study, Lamarca et al. (2024) further suggested that an absence of the ability to store sperm may increase extinction rates in elasmobranchs, through a reduction in genetic diversity. Because our results confirmed multiple paternity as a mating strategy for C. brevipinna, the perceived lack of sperm storage in this species does not lend concern to these implied consequences. However, further research on sperm storge, and its function in C. brevipinna, is needed to resolve these ideas.
Conclusions
This study has demonstrated, for the first time, that C. brevipinna in eastern Australian waters employs multiple paternity as part of their mating system. Although these results are consistent with existing studies of multiple paternity in the Carcharhinidae, further research to determine the frequency of multiple paternity in this species, and subsequent fitness of those offspring, is necessary. Furthering our understand of the intricacies of mating systems and reproductive strategies employed by these carcharhinids enables us to ascertain a more robust understanding of potential impacts to their populations. Analyses of multiple paternity and the role of sperm storage among genetically differentiated populations would also be beneficial; facilitating an understanding of potential impacts on the species globally.
Data availability
The data that support this study can be made available upon reasonable request from the corresponding author.
Conflicts of interest
Christine Dudgeon is an Associate Editor for Marine and Freshwater Research. Despite this relationship, she took no part in the review and acceptance of this manuscript, in line with the publishing policy. The authors declare that they have no further conflicts of interest.
Declaration of funding
A. K. Linn received honours research funding support through the University of the Sunshine Coast. Additional funding was provided by Shark Ecology Australia for SNP genotyping.
Acknowledgements
The authors acknowledge the Gubbi Gubbi and Yugambeh Peoples; the Traditional Custodians of the lands and seas on which this study was conducted, and pay respect to Elders past, present and emerging. We extend sincerest thanks to current Queensland Shark Control Program (QSCP) contractors, P. Dimond and C. Newton, for their assistance in providing samples for this research. We also thank our anonymous reviewers for their feedback on the initial draft.
References
Allen BR, Cliff G (2000) Sharks caught in the protective gill nets off Kwazulu–Natal, South Africa. 9. The spinner shark Carcharhinus brevipinna (Müller and Henle). South African Journal of Marine Science 22, 199-215.
| Crossref | Google Scholar |
Armada-Tapia S, Castillo-Geniz JL, Victoria-Cota N, Arce-Valdés LR, Enríquez-Paredes LM (2023) First evidence of multiple paternity in the blue shark (Prionace glauca). Journal of Fish Biology 102, 528-531.
| Crossref | Google Scholar | PubMed |
Byrne RJ, Avise JC (2012) Genetic mating system of the brown smoothhound shark (Mustelus henlei), including a literature review of multiple paternity in other elasmobranch species. Marine Biology 159, 749-756.
| Crossref | Google Scholar |
Capapé C, HEMIDA F, Seck AA, Diatta Y, Guélorget O, Zaouali J (2003) Distribution and reproductive biology of the spinner shark, Carcharhinus brevipinna (Müller & Henle, 1841) (Condrichthyes: Carcharhinidae). Israel Journal of Zoology 49, 269-286.
| Crossref | Google Scholar |
Coleman SW, Jones AG (2011) Patterns of multiple paternity and maternity in fishes. Biological Journal of the Linnean Society 103, 735-760.
| Crossref | Google Scholar |
Daly-Engel TS, Grubbs RD, Holland KN, Toonen RJ, Bowen BW (2006) Assessment of multiple paternity in single litters from three species of carcharhinid sharks in Hawaii. Environmental Biology of Fishes 76, 419-424.
| Crossref | Google Scholar |
Daly-Engel TS, Grubbs RD, Bowen BW, Toonen RJ (2007) Frequency of multiple paternity in an unexploited tropical population of sandbar sharks (Carcharhinus plumbeus). Canadian Journal of Fisheries and Aquatic Sciences 64, 198-204.
| Crossref | Google Scholar |
Daly-Engel TS, Grubbs RD, Feldheim KA, Bowen BW, Toonen RJ (2010) Is multiple mating beneficial or unavoidable? Low multiple paternity and genetic diversity in the shortspine spurdog Squalus mitsukurii. Marine Ecology Progress Series 403, 255-267.
| Crossref | Google Scholar |
Deeken D, Macdonald C, Gainsbury A, Green ML, Cassill DL (2024) Maternal risk-management elucidates the evolution of reproductive adaptations in sharks by means of natural selection. Scientific Reports 14, 20088.
| Crossref | Google Scholar |
Fitzpatrick JL, Kempster RM, Daly-Engel TS, Collin SP, Evans JP (2012) Assessing the potential for post-copulatory sexual selection in elasmobranchs. Journal of Fish Biology 80, 1141-1158.
| Crossref | Google Scholar | PubMed |
Frankham R (2005) Genetics and extinction. Biological Conservation 126, 131-140.
| Crossref | Google Scholar |
Geraghty PT, Williamson JE, Macbeth WG, Wintner SP, Harry AV, Ovenden JR, Gillings MR (2013) Population expansion and genetic structure in Carcharhinus brevipinna in the southern Indo-Pacific. PLoS ONE 8, e75169.
| Crossref | Google Scholar |
Geraghty PT, Macbeth WG, Williamson JE (2016) Aspects of the reproductive biology of dusky, spinner and sandbar sharks (family Carcharhinidae) from the Tasman Sea. Marine and Freshwater Research 67, 513-525.
| Crossref | Google Scholar |
Green ME, Appleyard SA, White W, Tracey S, Ovenden J (2017) Variability in multiple paternity rates for grey reef sharks (Carcharhinus amblyrhynchos) and scalloped hammerheads (Sphyrna lewini). Scientific Reports 7, 1528.
| Crossref | Google Scholar |
Gruber B, Unmack PJ, Berry OF, Georges A (2018) dartr: an R package to facilitate analysis of SNP data generated from reduced representation genome sequencing. Molecular Ecology Resources 18, 691-699.
| Crossref | Google Scholar | PubMed |
Hoekert WEJ, Neuféglise H, Schouten AD, Menken SBJ (2002) Multiple paternity and female-biased mutation at a microsatellite locus in the olive ridley sea turtle (Lepidochelys olivacea). Heredity 89, 107-113.
| Crossref | Google Scholar | PubMed |
Holmes BJ, Pope LC, Williams SM, Tibbetts IR, Bennett MB, Ovenden JR (2018) Lack of multiple paternity in the oceanodromous tiger shark (Galeocerdo cuvier). Royal Society Open Science 5, 171385.
| Crossref | Google Scholar |
Jennions MD, Petrie M (2000) Why do females mate multiply? A review of the genetic benefits. Biological Reviews of The Cambridge Philosophical Society 75, 21-64.
| Crossref | Google Scholar | PubMed |
Jones OR, Wang J (2010) COLONY: a program for parentage and sibship inference from multilocus genotype data. Molecular Ecology Resources 10, 551-555.
| Crossref | Google Scholar | PubMed |
Joung S, Liao Y, Liu K, Chen C, Leu L (2005) Age, growth, and reproduction of the spinner shark, Carcharhinus brevipinna, in the northeastern waters of Taiwan. Zoological Studies 44(1), 102-110 Available at https://zoolstud.sinica.edu.tw/Journals/44.1/102.html.
| Google Scholar |
Katona G, Szabó F, Végvári Z, Székely T, Jr, Liker A, Freckleton RP, Vági B, Székely T (2023) Evolution of reproductive modes in sharks and rays. Journal of Evolutionary Biology 36, 1630-1640.
| Crossref | Google Scholar | PubMed |
Kilian A, Wenzl P, Huttner E, Carling J, Xia L, Blois H, Caig V, Heller-Uszynska K, Jaccoud D, Hopper C, Aschenbrenner-Kilian M, Evers M, Peng K, Cayla C, Hok P, Uszynski G (2012) Diversity arrays technology: a generic genome profiling technology on open platforms. In ‘Data production and analysis in population genomics: Methods and Protocols. Methods in Molecular Biology, Vol. 888’. (Eds F Pompanon, A Bonin) pp. 67–89. (Humana Press) doi:10.1007/978-1-61779-870-2_5
Lamarca F, Carvalho PH, Vilasboa A, Netto-Ferreira AL, Vianna M (2020) Is multiple paternity in elasmobranchs a plesiomorphic characteristic? Environmental Biology of Fishes 103, 1463-1470.
| Crossref | Google Scholar |
Lamarca F, Carvalho PH, Netto-Ferreira AL (2024) The loss of female sperm storage ability as a potential driver for increased extinction in Chondrichthyes. Evolutionary Ecology 38, 461-479.
| Crossref | Google Scholar |
Lyons K, Chabot CL, Mull CG, Paterson Holder CN, Lowe CG (2017) Who’s my daddy? Considerations for the influence of sexual selection on multiple paternity in elasmobranch mating systems. Ecology and Evolution 7, 5603-5612.
| Crossref | Google Scholar | PubMed |
Lyons K, Kacev D, Mull CG (2021) An inconvenient tooth: evaluating female choice in multiple paternity using an evolutionarily and ecologically important vertebrate clade. Molecular Ecology 30, 1574-1593.
| Crossref | Google Scholar | PubMed |
Mull CG, Pennell MW, Yopak KE, Dulvy NK (2024) Maternal investment evolves with larger body size and higher diversification rate in sharks and rays. Current Biology 34, 2773-2781.E3.
| Crossref | Google Scholar | PubMed |
Nash CS, Darby PC, Frazier BS, Hendon JM, Higgs JM, Hoffmayer ER, Daly-Engel TS (2021) Multiple paternity in two populations of finetooth sharks (Carcharhinus isodon) with varying reproductive periodicity. Ecology and Evolution 11, 11799-11807.
| Crossref | Google Scholar | PubMed |
Neff BD, Pitcher TE (2005) Genetic quality and sexual selection: an integrated framework for good genes and compatible genes. Molecular Ecology 14, 19-38.
| Crossref | Google Scholar | PubMed |
Pirog A, Magalon H, Poirout T, Jaquemet S (2019) Reproductive biology, multiple paternity and polyandry of the bull shark Carcharhinus leucas. Journal of Fish Biology 95, 1195-1206.
| Crossref | Google Scholar | PubMed |
Pirog A, Magalon H, Poirout T, Jaquemet S (2020) New insights into the reproductive biology of the tiger shark Galeocerdo cuvier and no detection of polyandry in Reunion Island, western Indian Ocean. Marine and Freshwater Research 71, 1301-1312.
| Crossref | Google Scholar |
Portnoy DS, Piercy AN, Musick JA, Burgess GH, Graves JE (2007) Genetic polyandry and sexual conflict in the sandbar shark, Carcharhinus plumbeus, in the western North Atlantic and Gulf of Mexico. Molecular Ecology 16, 187-197.
| Crossref | Google Scholar | PubMed |
Pratt HL, Jr, Carrier JC (2001) A review of elasmobranch reproductive behavior with a case study on the nurse shark, Ginglymostoma cirratum. Environmental Biology of Fishes 60, 157-188.
| Crossref | Google Scholar |
Premachandra HKA, Nguyen NH, Knibb W (2019) Effectiveness of SNPs for parentage and sibship assessment in polygamous yellowtail kingfish Seriola lalandi. Aquaculture 499, 24-31.
| Crossref | Google Scholar |
Rigby CL, Carlson J, Smart JJ, Pacoureau N, Herman K, Derrick D, Brown E (2020) Spinner shark Carcharhinus brevipinna. In ‘The IUCN Red List of Threatened Species 2020’. e.T39368A2908817. (International Union for Conservation of Nature and Natural Resources) Available at https://www.iucnredlist.org/species/39368/2908817
Rossouw C, Wintner SP, Bester-Van Der Merwe AE (2016) Assessing multiple paternity in three commercially exploited shark species: Mustelus mustelus, Carcharhinus obscurus and Sphyrna lewini. Journal of Fish Biology 89, 1125-1141.
| Crossref | Google Scholar | PubMed |
Sansaloni C, Petroli C, Jaccoud D, Carling J, Detering F, Grattapaglia D, Kilian A (2011) Diversity Arrays Technology (DArT) and next-generation sequencing combined: genome-wide, high throughput, highly informative genotyping for molecular breeding of Eucalyptus. BMC Proceedings 5, P54.
| Crossref | Google Scholar |
Wang J (2018) Estimating genotyping errors from genotype and reconstructed pedigree data. Methods in Ecology and Evolution 9, 109-120.
| Crossref | Google Scholar |
Wang J (2019) Pedigree reconstruction from poor quality genotype data. Heredity 122, 719-728.
| Crossref | Google Scholar | PubMed |
Williamson MJ, Dudgeon C, Slade R (2018) Tonic immobility in the zebra shark, Stegostoma fasciatum, and its use for capture methodology. Environmental Biology of Fishes 101, 741-748.
| Crossref | Google Scholar |


