Optical properties of dissolved organic matter along a salinity gradient from a boreal river estuary to open coastal waters
Aleksandr Berezovski A * , Dag O. Hessen A , Hanne Halkjelsvik Børseth A and Tom Andersen A
A * , Dag O. Hessen A , Hanne Halkjelsvik Børseth A and Tom Andersen A
A
Abstract
Water darkening is an increasingly relevant problem affecting many coastal ecosystems, and it is exacerbated by human activity and climate change.
To determine the spectral, isotopic, and quantitative changes in coloured dissolved organic matter (CDOM) across a salinity gradient from the Glomma River to the outer Oslofjord.
CDOM absorbance was measured on a spectrophotometer and organic carbon in the water column was analysed using stable isotopes.
Spectral slope and CDOM absorption at 443 nm decreases linearly with an increasing salinity. CDOM absorption exhibited significant variations, with low-salinity samples showing higher light absorption per unit of carbon, but little seasonal variations. δ13C of TOC analysis showed a strong positive correlation with salinity.
Dissolved organic matter optical characteristics of coastal waters are tightly linked to CDOM. The distance to the river mouth and to the shore are some of the most important factors affecting water clarity. The δ13C of TOC analysis indicated a linear transition from terrestrial to marine carbon sources.
The study has shown how coastal darkening can be an important ecological issue mainly in near-coastal areas and describes valuable diagnostic tools for tracking it.
Keywords: CDOM absorption, coloured dissolved organic matter, δ13C of TOC, estuary, light attenuation, salinity gradient, spectral absorbance.
Introduction
Landscapes, freshwaters and coastal areas are tightly linked in seaward catchments, and catchment properties and terrestrial processes will thus ultimately affect marine systems, for example, in terms of export flux of substances and elements such as dissolved organic carbon (DOC) (Regnier et al. 2022; Crapart et al. 2023). The current ‘browning’, i.e. increased load of coloured, terrestrial matter into freshwaters, is typically observed in boreal areas (Monteith et al. 2007; Finstad et al. 2016; Creed et al. 2018). This is expected to have an impact on coastal systems as well, often here denoted ‘coastal darkening’ (Opdal et al. 2019; Frigstad et al. 2020; McGovern et al. 2020). Coastal darkening could be considered a form of optical deterioration of water quality, caused by coloured dissolved organic matter (CDOM) loading and resulting in increased light attenuation. The magnitude and ecological impact of coastal darkening is far from settled and will also have a pronounced spatial and temporal variability. The North Sea, Norwegian coastal waters and the Baltic Sea are all interconnected and influenced by the coloured organic matter coming from land. The quantity and quality of the organic matter entering marine waters is heavily affected both by catchment properties and climate. Typically, brown, DOC-rich water are prevalent in boreal system with coniferous forest and bogs as the main spatial determinants (Larsen et al. 2011; Finstad et al. 2016). These northern ecosystems are also disproportionately affected by climate warming (Cohen et al. 2014; Myers-Smith et al. 2020), and many regions also experience changes in snow cover and precipitation that may affect concentrations and runoff of DOC (de Wit et al. 2016).
Boreal regions are undergoing accelerated greening (increased vegetation cover and thus also carbon fixation) as a result of afforestation, as well as higher temperatures and elevated runoff. Increased terrestrial net primary production translates into larger loads of organic matter entering the water systems, and is one of the likely causes for the observed browning (Larsen et al. 2011; Finstad et al. 2016; Kritzberg 2017). Also, the reduced acid deposition promotes leaching and export of DOC from soils (Monteith et al. 2007). Dissolved organic matter (DOM) is the predominant form of organic carbon in aquatic systems (Hedges et al. 1997; Findlay and Parr 2017). As it travels with water masses from catchments and among bodies of water, DOM undergoes microbial and photochemical degradation (Cory et al. 2014), meaning that DOM cannot be considered a conservative substance. Also, exudates from phytoplankton and bacteria contribute to the total DOM pool and light attenuation; yet, this autochthonous DOM lacks the highly brown humic compounds (Jaffé et al. 2004). Humic substances in allochthonous DOM strongly promote light attenuation in the shorter wavelengths, because of high absorption potential of double bonds in their molecular structure (Martín et al. 2023). These double bond groups are known as chromophores, and they confer colour to humic compounds (Stevenson 1994). CDOM is the component of DOM that primarily absorbs light. Increasing concentrations of CDOM can affect aquatic ecosystems by diminishing light, where the organic molecules become effectively a ‘competitor’ to the light-harvesting pigments of phytoplankton. Thus, reducing photosynthetic activity in a water body, resulting in lower autotrophs production and biomass of phytoplankton or aquatic macrophytes, and shift the compensation depth upward (Karlsson et al. 2009; Thrane et al. 2014). Reduced primary production will subsequently limit biomass of higher trophic levels (du Pontavice et al. 2021). However, riverine exports will also promote phytoplankton productivity in coastal systems, owing to the transport of nutrients such as nitrogen (N), phosphorus (P), silicon (Si) and iron (Fe) (Deininger et al. 2016). Thus, the net effect on the productivity of coastal waters is not settled and will depend both on the flux and fate of DOM v. nutrient elements.
Phytoplankton blooms have been predicted to occur earlier as global warming progresses because of earlier and increased stratification (Desmit et al. 2020). Yet, for Norwegian coastal waters, peak spring phytoplankton blooms have been delayed by 22 days over the past century, which was attributed to coastal darkening (Opdal et al. 2019, 2023). Mustaffa et al. (2020) studied the relationship between light intensity and phytoplankton growth rates in the Sognefjord at the Norwegian western coast affected by freshwater inputs from several rivers. They found that increased light intensity led to higher phytoplankton growth rates in the central and outer fjord, but with no effect on phytoplankton in the open ocean, where the impact of terrestrial DOM is marginal. Besides impact on autotrophs, darkening and spectral changes may also affect higher trophic levels by favouring tactile predators (e.g. jellyfish) over visual predators such as fish, potentially causing an ecosystem regime shift (Eiane et al. 1997; Aksnes et al. 2009). DOC enrichment associated with darkening was also shown to lead to microbial community composition changes (Spilling et al. 2022; Lyche Solheim et al. 2024). Increased inputs of DOC will promote system heterotrophy by providing a substrate for heterotrophic bacteria while reducing primary production (Tranvik and Sieburth 1989). This shift can enhance microbial loop, altering the structure of the food web from traditional grazing chains to new microbial pathways.
Total organic carbon (TOC) is a sum of particulate organic carbon (POC) and DOC. DOC typically constitutes more than 90% of TOC in Norwegian coastal waters (Olsen et al. 2006). It is important to study TOC, when investigating light climate of aquatic environments, because organic carbon compounds are some of the most light-attenuating elements in the water. To separate the sources of TOC along this transect from the river mouth to open waters, we used the δ13C isotope signature of TOC as a marker. This is often used to discriminate between allochthonous and autochthonous TOC sources, because δ13C signature of terrestrial C3 plant material is between −23 and −34‰, whereas for marine phytoplankton it typically ranges from −18 to −22‰ (Lee et al. 2020). These differences in δ13C signature occur as a result of differences in photosynthetic pathways and in CO2 diffusion rates in air and water (O’Leary 1988). δ13C TOC thus serves as a proxy for how much of the organic carbon dissolved in a water system is of terrestrial origin at the moment, unlike measuring δ13C in sediments that gives a record of historical carbon data deposition. This sort of analysis is rarely conducted, owing to difficulties in methodology. However, as we show in this study, it can be effectively implemented for determining the source of organic carbon. The authors show that the use of stable isotope analysis of δ13C of TOC on a freshwater–marine gradient can be a valuable diagnostic tool for the origin of TOC.
The main aim of this study was to identify whether and how terrestrially derived DOM undergoes spectral, isotopic and quantitative changes along a spatial transect from land to open water. This, in turn, helps assess the extent of light attenuation in the coastal waters. To achieve this we measured light absorption by CDOM along a salinity gradient from the mouth of the Glomma (largest river in Norway) to the outer Oslofjord in different seasons. We hypothesised that DOM concentrations would follow passive mixing, and hence water transparency should be negatively correlated with salinity. We also used spectroscopy and stable isotope analysis to infer the endmembers of terrestrial and marine TOC. We further hypothesised that there would be seasonal variations in CDOM absorbance, with higher concentrations during high-runoff seasons.
Materials and methods
Study site and sampling methods
The Glomma Estuary and the transect in the outer Oslofjord in south-eastern Norway was chosen as a study site because of its location; it is a region where the DOC-rich water from the Glomma mixes with Baltic Sea water carried by the Skagerrak Coastal Current and seawater coming from the North Sea via the Atlantic Current (Sætre 2007). The Glomma River is the largest river in Norway, with a mean daily discharge of 684 m3 s−1 (since 1964; Norges vassdrags- og energidirektorat 2023) and a total catchment area of 41,970 km2 (NVE Atlas, see https://atlas.nve.no/html5Viewer/?viewer=nveatlas, accessed 7 June 2023). It drains large parts of alpine landscapes and boreal forest with bogs and wetlands, before entering the Oslofjord (Fig. 1). Flow regime in the sampling area is dependent on the Baltic outflow and most importantly on the discharge from the Glomma.
Sampling area with six sampling stations and the location of the sampling area on a map of Scandinavia (bottom-right corner). The image was created in Stadia Maps (see https://stadiamaps.com/).
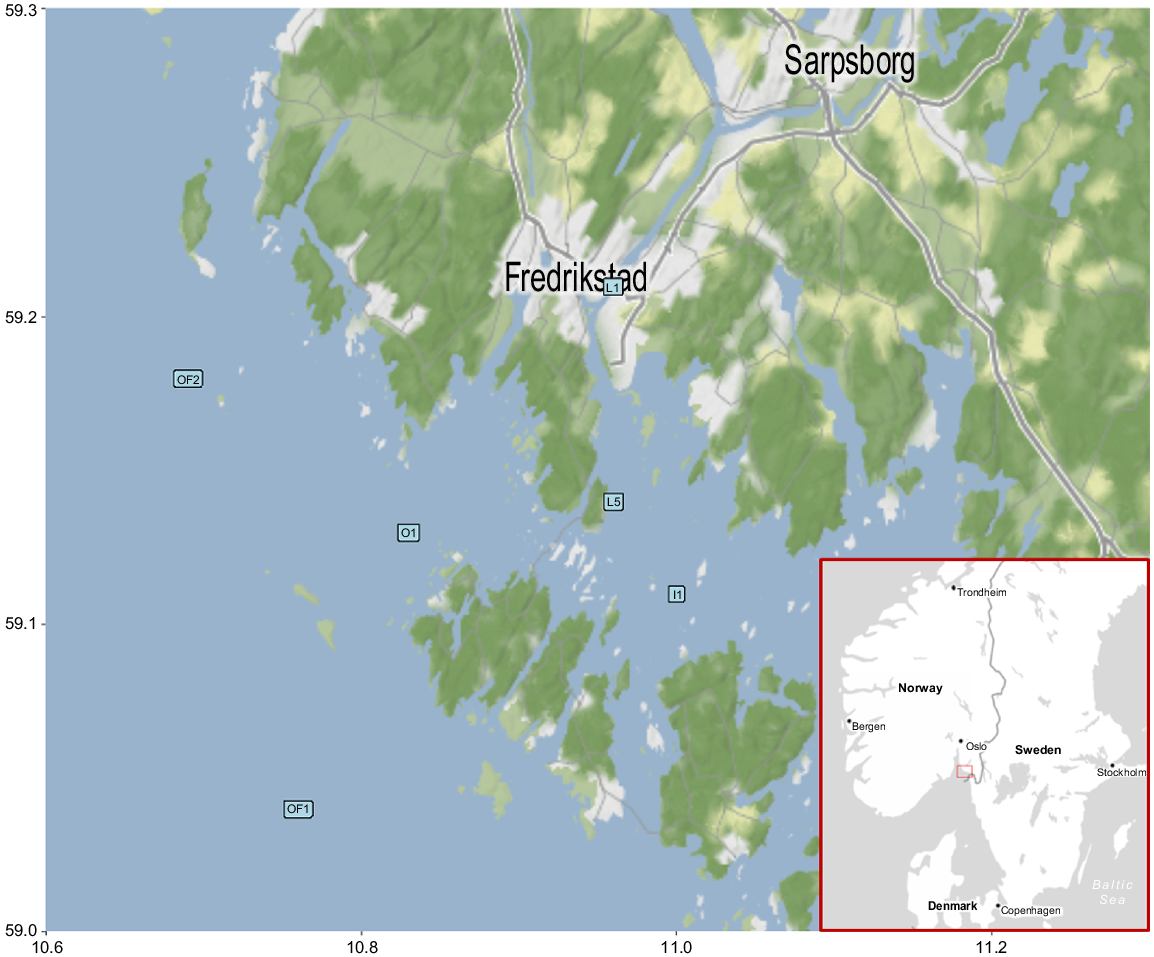
Six sampling cruises took place between June 2020 and June 2021 (4 June 2020, 8 September 2020, 26 November 2020, 23 February 2021, 25 March 2021 and 17 June 2021). Three additional sampling cruises were conducted to obtain more data on DOM (12 May 2022, 2 June 2022 and 29 June 2022) (Børseth 2022). A usual sampling route encompassed six sampling stations (marked OF2, OF1, O1, I1, L5 and L1 in Fig. 1). The coordinates and other information regarding the sampling stations can be found in Supplementary Table S1. The same stations were visited at approximately the same times of day (12:00 hours ± 120 min) on all sampling days, meaning the differences of physicochemical properties of water owing to the tide level were minimal (biggest tidal range among all six sampling dates was 40 cm; Kartverket 2024). The stations represented a salinity gradient with the highest salinity at Station OF2 and lowest at Station L1. The salinities of each station show seasonal variation, yet at very different levels. For the open-sea site, salinity ranged between 19 and 29 PSU, whereas the inner, riverine site had salinities ranging from 0 to 5 PSU. The total transect length was 56 km. Conductivity, temperature and oxygen concentrations were recorded in situ by using a CTD profiler (Sea-Bird Model SB9).
Absorbance measurements
Water samples were collected at each sampling site with a Rosette sampler. Water samples were extracted from 3–4-m depths, representing the mixed layer in most cases. Mixed layer depth was, on average, the shallowest at L5 (5 m) and the deepest at OF1 (10 m). The samples were brought to the laboratory where the absorbance by CDOM was measured on a double beam Shimadzu UV-2550 UV-VIS Spectrophotometer (Shimadzu Scientific Instruments, Columbia, MD, USA). The definition of ‘dissolved’ organic matter is somewhat arbitrary, Xu and Guo (2017) described DOM as any molecules that pass through a <0.7-μm pore-sized filter. In this study, 0.7-μm pore GF/F Whatman glass-fibre filters were used to separate the ‘dissolved’ part of organic matter from larger detritus.
Absorbance of the filtrate (Aλ) was measured in 5-cm (L) quartz cuvettes over the 250–750-nm range with 1-nm increments and referenced to Milli-Q water. The resulting CDOM absorbance (log10 scale of absorbance per centimetre of path length) was subsequently transformed into base-e absorption coefficients per metre (aλ, as seen in Eqn 1). All absorbance values were corrected for baseline offset by subtracting average absorbance between 740 and 750 nm (Green and Blough 1994). The 740–750-nm range was chosen because absorbance by CDOM is expected to be negligible here.
Organic carbon concentrations
TOC concentrations were also analysed by high-temperature catalytic oxidation and non-dispersive infra-red detection on a Shimadzu TOC-VCPH Analyzer (Shimadzu Scientific Instruments). The water samples were heated to 680°C with platinum catalyst and subsequently converted into CO2 gas and measured.
POC concentrations were measured by filtering sample water through a 0.7-μm pore GF/F glass-fibre filter and measured on a FlashEA 1112 Nitrogen and Carbon Analyzer (ThermoFisher Scientific), where filters were combusted and the produced gas was analysed on elemental composition by gas chromatography. DOC concentrations were calculated by subtracting POC from TOC concentrations.
Stable isotope analysis
δ13C of TOC (both dissolved plus particulate organic carbon) was measured with an isotope ratio mass spectrometry (IRMS). The reason for analysing TOC as a whole was not to prioritise organic molecules of certain sizes over the others and also to lower risk of contamination. Analysis of δ13C of TOC in marine water poses problems due to salt content. Salt combusted at high temperatures deteriorates quartz tubing inside the elemental analyser, as well as decreases the efficiency of combustion. Also, combusted salt may ‘compete’ with CO2 molecules in the analyser, leading to incorrect readings (Lalonde et al. 2014). Because of these complications, data for TOC δ13C in salt water are rarely measured. Our δ13C analysis was performed by Jan Veizer Stable Isotope Laboratory (Ottawa, ON, Canada). A predetermined quantity of water was deposited in a reaction chamber of an OI Analytical Aurora Model 1030W TOC Analyser, where it was mixed with hydrochloric acid to remove inorganic carbon. The water containing organic carbon was combusted several times and passed through chemical and Nafion traps to remove traces of water. CO2 was trapped by GD-100 trap, where the peak was rinsed with ultrapure helium gas before the analysis in IRMS (Lalonde et al. 2014). Organic carbon was converted into CO2 gas as a result of combustion, which is then passed into a Finnigan Mat DeltaPlusXP IRMS. Data were normalised using two different internal organic standards and the analytical precision was ±0.2‰.
Modelling
Salinity dilution curves were analysed by simple or additive regression models. For linear mixed models we used the lme4 package (ver. 1.1-37, see https://CRAN.R-project.org/package=lme4; Bates et al. 2015), whereas we used the mgcv package (ver. 1.9-3, S. Wood, see https://cran.r-project.org/package=mgcv/; Wood 2017) for additive models. Hierarchical generalised additive models (HGAMs) were used to estimate smooth function relationships between dissolved substances and salinity, grouped by sampling date (Pedersen et al. 2019). HGAMs can represent non-conservative mixing as departures from linearity in the salinity relationship.
CDOM absorption coefficients (aλ) as a function of wavelength was modelled as an exponential function with an offset (Eqn 2), as suggested by Stedmon et al. (2000). The model has the following three parameters: the absorption coefficient a443 at the reference wavelength (443 nm), the spectral slope (S) and the background absorbance (K). S represents relative slope of the spectrum independent of wavelength () and shows the rate of change of absorption. The wavelengths were rescaled from nanometres to micrometres following Stedmon et al. (2000), such that the unit of S will be per micrometre. The wavelength 443 nm (0.443 μm) was used as the reference wavelength because it is around blue absorbance peak of chlorophyll-a (Kirk 2011), and is often used in remote-sensing models of ocean colour. All CDOM spectra were fitted simultaneously to a non-linear model by using the saemix package (ver. 3.3, see https://cran.r-project.org/package=saemix; Comets et al. 2017) in RStudio (ver. 2023.06.1+524, Posit Software, PBC, Boston, MA, USA, see https://posit.co/products/open-source/rstudio/).
CDOM absorption coefficient at 443 nm (a443) for every sample was estimated according to Stedmon et al. (2000) model. Specific absorption coefficient at 443 nm (sa443) was calculated as CDOM absorption coefficient at 443 nm (a443) divided by DOC concentration of the water sample (). Resulting units were square metres per gram. The wavelength of 443 nm was once again chosen as a reference wavelength. sa443 is used to estimate DOC from remote sensing (Kutser et al. 2015).
Statistical analyses
We conducted several statistical analyses to assess relationships and variations of selected parameters. δ13C, TOC concentrations, S, a443 and sa443 were analysed using linear regression models, where both salinity (as a continuous predictor) and sampling date (as a categorical predictor representing seasonality) were included as explanatory variables. The significance of each predictor in explaining variance within the response variables was then assessed using ANOVA.
Results
Water samples had a good spread of salinities, ranging from 0.03 in the inner station (L1) to 29.50 PSU at the outer (OF2) (Fig. 2, x-axis). The six sampling cruises covered different seasons, reflecting that the discharge volumes from the Glomma River varied with season and precipitation. Cruises 2, 4, 5, 7 and 8 (Fig. S2) took place during low-discharge periods (<600 m3 s−1), whereas Cruises 1 and 9 saw medium discharge levels (<1000 m3 s−1). Cruises 3 and 6 occurred after heavy discharge periods (<1300 m3 s−1). Salinity changed strongly between the sampling seasons in shallow water layers at every sampling station.
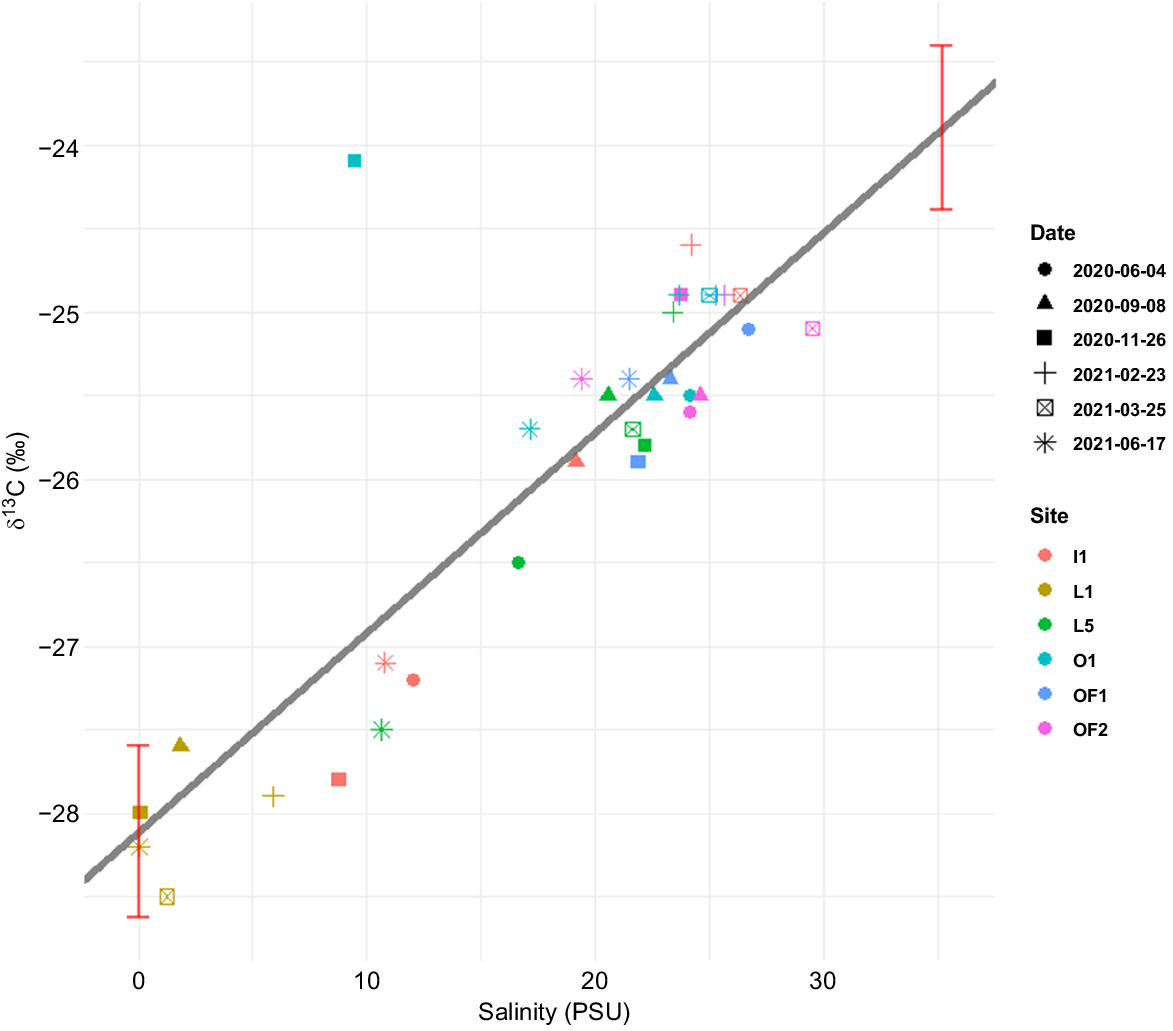
Relationship between δ13C (‰) of TOC in water samples and salinity (PSU). Line represents simple regression. Red bars represent 95% confidence intervals of the freshwater and marine endmembers based linear regression model. Shapes of data points correspond to the dates of sampling and colours correspond to the sampling stations.
δ13C of TOC
The δ13C of TOC data were predicted using HGAM as well as linear mixed-effect models with or without random slopes, but all of them showed very small variance among the seasons. Therefore, a simple regression model without grouping by seasons was chosen as a preferable model to describe decreasing δ13C of TOC along the salinity gradient (Fig. 2). There was a strong positive linear correlation between δ13C and salinity (F = 89.16, P < 0.01, R2 = 0.736) and insignificant seasonal variations in δ13C values (F = 0.57, P = 0.72). The boundaries for δ13C of freshwater and marine (0 and 35.2 PSU accordingly) endmembers were estimated from the regression model using the salinity range in Norwegian coastal waters reported by Aksnes (2015). The δ13C of TOC at the riverine station is constrained within freshwater boundaries, suggesting an entirely freshwater origin of TOC. From 10 PSU onward, TOC begins to display δ13C values higher than −27.5‰, indicative of a mix of marine and terrestrially derived carbon in the middle of the salinity transect. The outermost sampled stations, located at ~25–30 PSU, still exhibited some freshwater influence owing to the shallow sampling depth. As a result, the δ13C values did not reach strictly marine levels, instead varying between −25.5 and −24.5‰. This effectively means that there are lighter carbon isotopes in the freshwater-affected inner estuary and that the proportion of carbon shifts to heavier carbon further out in the open sea. This strong and linear relationship implies that δ13C can serve as a good diagnostic tool for the fraction of terrestrial (allochthonous) v. marine (autochthonous) DOM.
Knowing the marine and freshwater endmembers allows for calculating of the fraction of terrestrially derived organic carbon (Faust and Knies 2019). For example, the central data point (L5, 4 June 2020) has a δ13C value of −25.7‰, corresponding to a terrestrially derived organic carbon fraction of 43%. The highest terrestrial fraction (100%) was observed at L1 on 25 March 2021, whereas the lowest fraction (1%) was unexpectedly recorded at O1 on 26 November 2020.
TOC concentration
DOC was a major component of TOC (89% average; Table S2). DOC:TOC ratios varied with season; June 2020 had the lowest mean DOC:TOC ratio of all the sampled seasons (71%; Fig. S1), September saw higher average DOC:POC (84%), and November 2020 and February, March and June 2021 all had DOC constituting over 90% of TOC in the water column. We found no systematic change in dissolved fraction over salinity range; DOC:TOC fractions stayed mostly the same throughout the salinity gradient, except in June 2020, where a decline of DOC fraction at increased salinity was observed (Fig. S1).
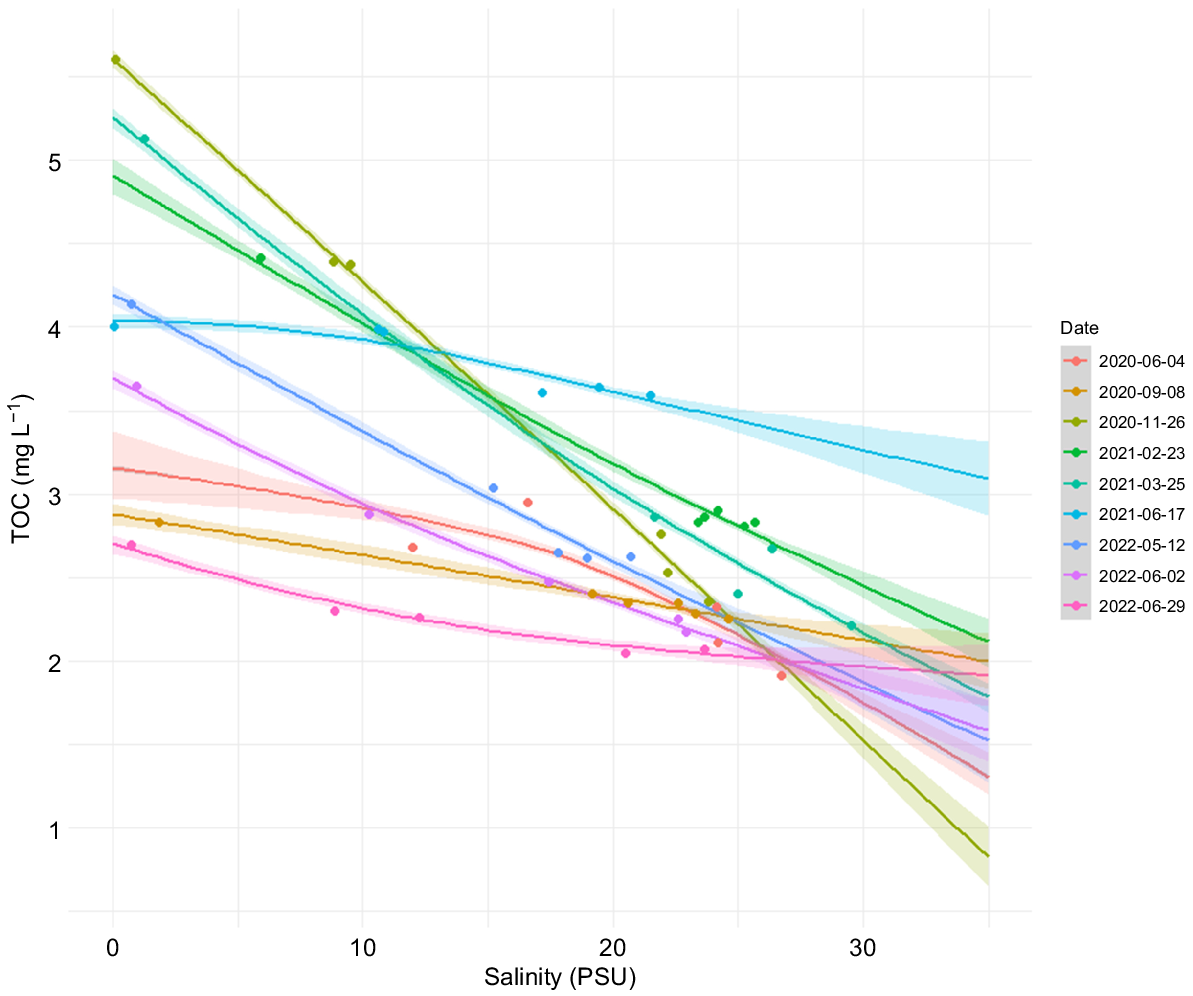
HGAMs were used to fit TOC concentration over 0–35 salinity range grouped by sampling date (Fig. 3). AICs of HGAM and linear model were compared (8 and 64 accordingly), indicating non-linear relationship and thus a better fit from HGAMs. As a general trend, TOC concentrations decreased nearly linearly with an increasing salinity in colder seasons (Novembers–May, and to a lesser extent September), whereas summer seasons showed indications of non-conservative mixing, especially in the freshwater end of the gradient, where TOC concentrations are more varied. The outer sampling sites were more uniform with less seasonal and site-by-site fluctuations of TOC concentrations. According to an ANOVA analysis, there were significant variations in TOC concentrations among different seasons (F = 115.06, P < 0.01). A linear regression was used to relate TOC concentrations to mean discharge volume (time lag of 14 days) at the river mouth site throughout 2 years of sampling. The variations in the TOC concentration at the river mouth station were not related to river flow (b = 74.70 ± 137.88, P = 0.61).
Spectral slopes and CDOM absorbance
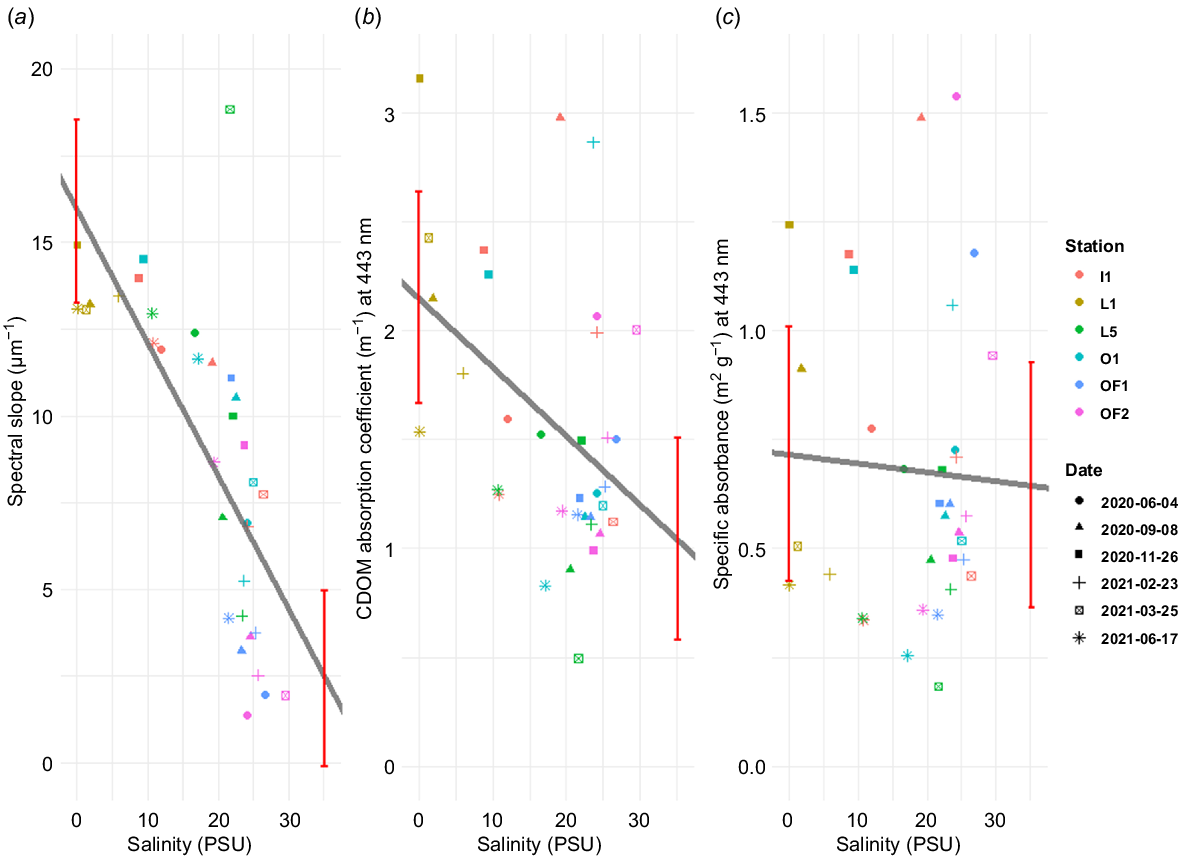
a443 was linearly related to salinity in the Glomma Estuary (Fig. 4b). There were significant differences when comparing a443 to salinity (F = 10.40, P < 0.01), but without a significant variation among the seasons (F = 1.16, P = 0.35). Spectral slope (S) followed a similar trend, but the decrease over salinity was even sharper (F = 27.32, P < 0.01; Fig. 4a). Seasonal changes in S were also found to be significant (F = 3.30, P = 0.02). sa443 values displayed a weak trend of decreased sa443 at higher salinities (Fig. 4c). However, there was a strong scatter in sa443 across the salinity gradient, and the confidence intervals of the endmembers overlapped. This suggests that sa443 does not differ significantly between freshwater and marine water samples, which was also supported by ANOVA showing no significant change of sa443 along the salinity gradient (F = 0.76, P = 0.39). Because S is variable across the salinity gradient, whereas sa443 is not, it can be deduced that S changes the most at a pivot point somewhere below 443 nm. Values for S, sa443 and sa443 can be found in Table S3.
Relationship between salinity (PSU) and saemix (Comets et al. 2017) model parameter estimates: (a) spectral slope (S, μm−1), (b) absorption coefficient at the reference wavelength (a443, m−1) and (c) specific absorption coefficient (sa443, m2 g−1) at reference wavelength. Grey line represents simple linear regression, whereas red bars are its confidence intervals for freshwater and marine endmembers. Shapes of datapoints correspond to sampling dates and colours represent sampling station.
Discussion
DOM dynamics
DOM undergoes qualitative and quantitative changes along the salinity gradient. The estuarine waters have higher DOM concentrations and thus also higher light absorption than do more offshore waters. Seasonality played a major role in changes in TOC concentration, especially in freshwater end of the gradient. The seasonal fluctuation declined in the outer sea, although, still, with significant seasonal changes in TOC concentration (Fig. 3) and insignificant variations in absorption coefficients (Fig. S3). Spring snowmelt, which occurs in May to June in this region (Poste et al. 2021), seems to add non-linearity to TOC mixing across the salinity gradient. All the summer samplings differ greatly in their observed TOC concentrations; however, the common trend is that summer HGAMs show curvature, whereas samplings from colder seasons (including early May sampling) show more linear patterns of TOC mixing. Overall, TOC concentrations are higher at the winter seasons, which is consistent with previous studies (Poste et al. 2021). Element fluxes from Glomma are more evenly scattered throughout a year because of the combined effect of hydrology and agricultural runoff (Poste et al. 2021).
TOC concentrations correlated weakly with discharge volumes in the days leading to the sampling days (Fig. S2). For example, discharge volume during the November sampling was two-fold higher than in February and March; however, the TOC concentrations across the three seasons were similar (Fig. 3). In fact, TOC concentrations at lower discharge seasons even exceeded November concentrations at the more saline part of the gradient. Conversely, the contrast between June 2020 and 2021 TOC concentrations shows that discharge volumes could indeed be used to predict TOC input in the area. Observations from June 2020 capture the beginning of summer flood, whereas those from June 2021 reflect the period after the major summer flood. This affected TOC concentrations in the way that there are generally lower TOC concentrations throughout the whole salinity gradient in the 2020 season. Overall, seasonal differences in TOC concentrations do not perfectly mirror seasonality in river discharge, in support of the findings from other boreal rivers (Lepistö et al. 2008; Schultze et al. 2022) and temperate estuaries (García-Martín et al. 2021).
Frigstad et al. (2023) reported an increase of TOC transport to the Skagerrak area from major rivers, including the Glomma, from 1990 to 2020. This coincided with an increase in discharge volumes and a decrease in surface salinity.
Allochthonous v. autochthonous organic carbon fraction
δ13C of TOC describes the relative contribution of terrestrial organic matter in the Oslofjord fairly well (Fig. 2). Carbon isotope ratios changed linearly with salinity, consistent with the findings of Osburn and Stedmon (2011). Peterson et al. (1994) estimated the range for marine endmember in US estuaries to be between −22 and −25‰, whereas the freshwater endmembers were between −26 and −28‰. The majority of measured isotope ratios in this study lie well between these boundaries. The linear transition from terrestrially derived DOM in the inner estuary to marine DOM in the open fjord indicates a conservative mixing with salinity, as was also confirmed by the linear response of CDOM absorption coefficients along the transect (Fig. 4b). A study on isotopic composition of DOC in American estuaries reported the biggest seasonal variations in the Altamaha Estuary, with average carbon isotope signatures deviating between −19.9‰ in summer–autumn to −22.3‰ in spring (Otero et al. 2003). However, we found minimal fluctuations in δ13C among all six sampling cruises, suggesting that the terrestrial and marine endmembers for DOM have consistent isotopic composition throughout the year (minimal and maximal average carbon isotopic composition are −25.3‰ in June and −24.1‰ in February). δ13C of TOC analysis of seawater is an underutilised method in scientific literature, owing to complications associated with salt combustion. Salts interfere with traditional IRMSs, meaning that only a few laboratories are equipped to run such analysis; hence, the results from a freshwater–marine gradient are rare (e.g. Sampedro-Avila et al. 2024). We think that showing a progressively smaller allochthonous signal throughout the salinity gradient (Fig. 2) is a visually demonstrative answer to the set hypothesis that CDOM absorbance reduces along the salinity gradient. Although δ13C of TOC suspended in water column gives us good understanding of current isotopic composition of organic carbon in the area, application of δ13C analysis on sediment profiles may also give a proxy of the relative contribution of allochthonous v. autochthonous C over time.
The main source of freshwater and terrestrial DOM to the surveyed transect is the Glomma river. The catchment of this largest Norwegian river is located in the boreal and heavily forested central-eastern Norway, providing a large export of terrestrial organic matter. Seasonal fluctuations in TOC concentrations were prevalent in fresher near-shore sites, reflecting seasonality and variations in discharge, while being more constant (i.e. more tightly clustered) in the outer sea environments, where TOC had time to mix in the water column (Fig. 3). This is consistent with Schultze et al. (2022), who reported more seasonal variability in organic carbon in a boreal river than in an open fjord where hardly any seasonal variations were encountered.
CDOM absorbance changes and along the salinity gradient
There were also striking changes in DOM quality and optical properties along the salinity gradient, as evident from the CDOM absorption (Fig. S3), where low-salinity samples absorbed more light per unit of carbon. Judging from spectral slope (Fig. 4a), CDOM is following conservative mixing. Endmembers of spectral slope are significantly different, suggesting that the shape of CDOM spectrum changes as it gets further from the shore. a443 (Fig. 4b) was less consistent, with many samples having disproportionately high absorption coefficients all throughout the salinity gradient. Nevertheless, the boundaries of endmembers are far from overlapping, indicating that there is significant difference in how much light is absorbed by CDOM at shorter wavelengths in fresh v. marine waters, which is also supported by an ANOVA test. However, sa443 (Fig. 4c) values suggest that the variation in CDOM across the gradient is more quantitative than qualitative, because the endmember margins overlap. The fact that S changes, whereas sa443 stays mostly constant, indicates that the qualitative changes in the CDOM spectrum are mainly below 443 nm.
Harvey et al. (2015) studied light attenuation in the Baltic Sea by using absorption at 440 nm as a reference wavelength, which closely aligns with our absorption measurements at 443 nm. Whereas waters in the Baltic Proper contain much less CDOM, resulting in weak absorption (~0.5 m−1 at 440 nm), the Gulf of Bothnia exhibited significantly higher absorption than what we measured in the Norwegian coastal waters. Our a₄₄₃ values are mostly confined to 3 m−1, whereas Harvey et al. (2015) recorded absorption as high as 8 m−1 during summer. Their May sampling showed the highest CDOM absorption, consistently reaching 3–7 m−1. These findings suggest that even Norwegian coastal waters, including the Glomma Estuary, are not as CDOM-rich as those in the Baltic Sea.
CDOM absorption and S in the Skagerrak and Kattegat regions were previously studied by Stedmon et al. (2000). Their sampling transects extended farther from the coast, resulting in lower riverine influence. Consequently, their CDOM absorption coefficients were lower than those observed in this study, ranging from 0.091 to 0.915 m−1, despite using a shorter reference wavelength of 375 nm. By contrast, their S values (17.6–23.4 μm−1) were higher but still comparable to ours (1.4–18.8 μm−1). Given the influence of outflows from the Skagerrak, Kattegat and Baltic Sea, it is likely that water clarity in these four interconnected systems is mutually affected to some extent.
Even though DOM is persistent in aquatic environments, it is being continuously removed through means of microbial degradation and photodegradation. Degradability of DOC is also dependent on the environment it is found in; ocean DOC is less degradable than inland water DOC (Catalán et al. 2016). Allochtonous DOM consists mainly of proteins (10%), carbohydrates (30–50%), lignin (15–25%) and other biomacromolecules (Nebbioso and Piccolo 2013). Whereas proteins and carbohydrates are susceptible to biodegradation, lignin is chemically stable and tends to stay in water for longer (Opsahl and Benner 1997; Nebbioso and Piccolo 2013; Baltar et al. 2021). Additionally, photochemically reactive molecules in DOM are degraded into lower molecular weight when exposed to sunlight (Bertilsson and Tranvik 2000; Ogawa and Tanoue 2003). Once DOM is at a low molecular weight (<1 kDa), it becomes resistant to microbial oxidation, which prevents it from further rapid degradation (Ogawa and Tanoue 2003; Xu and Guo 2018). At the same time, DOM needs to be small enough for transport across bacterial membranes to occur in the first place. Photodegradation also was shown to reduce the colour of DOM (Dempsey et al. 2020). This means that once DOM is out of groundwater and in open water systems, its attenuation potential is progressively lost.
A gradual decrease in Secchi depth (a measure of water clarity) was recorded in the North and Baltic Seas over the past century (Dupont and Aksnes 2013). CDOM enrichments was assigned as the most important contributor to the observed darkening; however, there are also other factors influencing the clarity of water. Some elements, such as iron (Fe), have additive effects with CDOM on light attenuation. This effect has been studied in the boreal headwaters and rivers, where Fe constitutes up to 25% of total light absorbance (Kritzberg and Ekström 2012). However, the interactions of Fe and CDOM are still understudied in marine environments and therefore require more research. Another factor influencing water clarity is resuspension of bottom sediments driven by wind. Wilson and Heath (2019) reported that change in wave climate in the North Sea over the past century may have played a big role in reduction of water clarity.
DOC mixing behaviour along the land–ocean transect
Flocculation potential of DOM changes with salinity and pH. DOM molecules become more stable when in seawater (Lasareva et al. 2019) and the optimal pH for flocculation is 6–7 (Asmala et al. 2014). Flocculation reduces DOM export from estuaries and promotes retention by sedimentation (Asmala et al. 2014). Our findings suggest generally a conservative DOC mixing (Fig. S4) and minimal flocculation (Table S2). Asmala et al. (2014) found that DOC tends to flocculate at lower salinities (0–7 PSU) into aggregates. Flocculation may therefore cause a transition of some DOC into POC as well as altering the spectral properties of DOC (Asmala et al. 2014). In our study, most of the flocculation possibly occurred in the river upstream from the first sampled location. We did not find evidence for flocculation along the transect, with a possible exception for June 2020, when DOC:TOC fraction was reasonably small to begin with and was decreasing in the more saline part of the transect. Otherwise, the DOC:TOC fraction stays mostly the same across the whole gradient and absolute DOC concentrations perpetually decrease. This is similar to observed monotonous decrease of DOC withan increase in salinity at other boreal estuaries (Hessen et al. 2010; García-Martín et al. 2021; Kleven 2021; Schultze et al. 2022).
Broader ecological impacts
Loads of DOM coming from land to sea could lead to darkening of coastal areas. This affects aquatic life in multiple ways; reduced transparency not only implies lower photosynthetic potential for phototrophs, but it also promotes heterotrophic bacteria that utilise this large supply of organic carbon. Visual predators are also affected by the change in optical light field, being disadvantaged by darker waters, whereas tactile predators are getting a competitive edge (Aksnes et al. 2004). There are thus potentially far-reaching and cascading ecosystem impacts of darkening, such as phenological shifts, destabilising whole ecosystems by mismatch between the spring bloom of primary producers and spawning seasons for consumers (Opdal et al. 2019, 2023). Because coastal darkening can result from factors such as climate change, land-use change, forestry and afforestation, sediment resuspension, nutrient enrichment and reduced acid deposition, it highlights the need for a holistic perspective on ecosystem connectivity. Changes in coastal environments should be understood in direct relation to upstream influences from climate, terrestrial and freshwater systems.
Conclusions
DOM optical characteristics of coastal waters are tightly linked to terrestrial DOM. Salinity, and consequently the distance from the river mouth, are among the key factors influencing water clarity along an estuarine transect. The δ13C analysis of TOC indicates a linear transition from terrestrial to marine, marking it as an effective and accurate method of tracking the fate of terrestrially derived organic matter in an estuary. Endmembers derived from the δ13C model enable the calculation of the terrestrial organic carbon fraction in both water and sediments, providing a valuable tool for ecosystem management and research. Spectral slope, CDOM absorption and specific absorption at 443 nm were used to describe qualitative changes in CDOM across the salinity transect. The observed seasonal and spatial patterns in TOC concentrations and CDOM absorbance provide insights into the biogeochemical processes governing carbon dynamics in estuarine systems. Understanding these dynamics is crucial for predicting the responses of estuarine ecosystems to changing environmental conditions and for managing these critical coastal zones.
Data availability
All data generated or analysed during this study are included in this article and accompanying supplementary material. A preprint version of this article is available (Berezovski et al. 2024).
Declaration of funding
This work was supported by Norges Forskningsråd. Grant number TerraCoast 287490, which was awarded to A. Berezovski, D. O. Hessen and T. Andersen.
Author contributions
A. Berezovski provided conceptualisation, data curation, formal analysis, investigation, visualisation, and writing of the original draft. D. O. Hessen contributed conceptualisation, supervision, and review and editing. H. H. Børseth undertook investigation. T. Andersen provided conceptualisation, supervision, formal analysis, and review and editing.
References
Aksnes DL (2015) Sverdrup critical depth and the role of water clarity in Norwegian coastal water. ICES Journal of Marine Science 72(6), 2041-2050.
| Crossref | Google Scholar |
Aksnes DL, Nejstgaard J, Sædberg E, Sørnes T (2004) Optical control of fish and zooplankton populations. Limnology and Oceanography 49(1), 233-238.
| Crossref | Google Scholar |
Aksnes DL, Dupont N, Staby A, Fiksen Ø, Kaartvedt S, Aure J (2009) Coastal water darkening and implications for mesopelagic regime shifts in Norwegian fjords. Marine Ecology Progress Series 387, 39-49.
| Crossref | Google Scholar |
Asmala E, Bowers DG, Autio R, Kaartokallio H, Thomas DN (2014) Qualitative changes of riverine dissolved organic matter at low salinities due to flocculation. Journal of Geophysical Research: Biogeosciences 119(10), 1919-1933.
| Crossref | Google Scholar |
Baltar F, Alvarez-Salgado XA, Arístegui J, Benner R, Hansell DA, Herndl GJ, Lønborg C (2021) What is refractory organic matter in the ocean? Frontiers in Marine Science 8, 642637.
| Crossref | Google Scholar |
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. Journal of Statistical Software 67(1), 1-48.
| Crossref | Google Scholar |
Berezovski A, Hessen DO, Børseth HH, Andersen T (2024) Optical properties of dissolved organic matter along a salinity gradient from a boreal river estuary to open coastal waters. Research Square 2024, ver. 1 [Preprint, published 16 June 2024].
| Crossref | Google Scholar |
Bertilsson S, Tranvik LJ (2000) Photochemical transformation of dissolved organic matter in lakes. Limnology and Oceanography 45(4), 753-762.
| Crossref | Google Scholar |
Catalán N, Marcé R, Kothawala DN, Tranvik LJ (2016) Organic carbon decomposition rates controlled by water retention time across inland waters. Nature Geoscience 9, 501-504.
| Crossref | Google Scholar |
Cohen J, Screen JA, Furtado JC, Barlow M, Whittleston D, Coumou D, Francis J, Dethloff K, Entekhabi D, Overland J, Jones J (2014) Recent Arctic amplification and extreme mid-latitude weather. Nature Geoscience 7(9), 627-637.
| Crossref | Google Scholar |
Comets E, Lavenu A, Lavielle M (2017) Parameter estimation in nonlinear mixed effect models using saemix, an R implementation of the SAEM algorithm. Journal of Statistical Software 80, 1-41.
| Crossref | Google Scholar |
Cory RM, Ward CP, Crump BC, Kling GW (2014) Sunlight controls water column processing of carbon in arctic fresh waters. Science 345(6199), 925-928.
| Crossref | Google Scholar | PubMed |
Crapart C, Finstad AG, Hessen DO, Vogt RD, Andersen T (2023) Spatial predictors and temporal forecast of total organic carbon levels in boreal lakes. Science of The Total Environment 870, 161676.
| Crossref | Google Scholar |
Creed IF, Bergström A-K, Trick CG, Grimm NB, Hessen DO, Karlsson J, et al. (2018) Global change-driven effects on dissolved organic matter composition: implications for food webs of northern lakes. Global Change Biology 24(8), 3692-3714.
| Crossref | Google Scholar | PubMed |
de Wit HA, Valinia S, Weyhenmeyer GA, Futter MN, Kortelainen P, Austnes K, Hessen DO, Räike A, Laudon H, Vuorenmaa J (2016) Current browning of surface waters will be further promoted by wetter climate. Environmental Science & Technology Letters 3(12), 430-435.
| Crossref | Google Scholar |
Deininger A, Faithfull CL, Lange K, Bayer T, Vidussi F, Liess A (2016) Simulated terrestrial runoff triggered a phytoplankton succession and changed seston stoichiometry in coastal lagoon mesocosms. Marine Environmental Research 119, 40-50.
| Crossref | Google Scholar | PubMed |
Dempsey CM, Brentrup JA, Magyan S, Knoll LB, Swain HM, Gaiser EE, et al. (2020) The relative importance of photodegradation and biodegradation of terrestrially derived dissolved organic carbon across four lakes of differing trophic status. Biogeosciences 17(24), 6327-6340.
| Crossref | Google Scholar |
Desmit X, Nohe A, Borges AV, Prins T, De Cauwer K, Lagring R, Van der Zande D, Sabbe K (2020) Changes in chlorophyll concentration and phenology in the North Sea in relation to de-eutrophication and sea surface warming. Limnology and Oceanography 65(4), 828-847.
| Crossref | Google Scholar |
du Pontavice H, Gascuel D, Reygondeau G, Stock C, Cheung WWL (2021) Climate-induced decrease in biomass flow in marine food webs may severely affect predators and ecosystem production. Global Change Biology 27(11), 2608-2622.
| Crossref | Google Scholar |
Dupont N, Aksnes DL (2013) Centennial changes in water clarity of the Baltic Sea and the North Sea. Estuarine, Coastal and Shelf Science 131, 282-289.
| Crossref | Google Scholar |
Eiane K, Aksnes DL, Giske J (1997) The significance of optical properties in competition among visual and tactile planktivores: a theoretical study. Ecological Modelling 98(2-3), 123-136.
| Crossref | Google Scholar |
Faust JC, Knies J (2019) Organic matter sources in North Atlantic fjord sediments. Geochemistry, Geophysics, Geosystems 20(6), 2872-2885.
| Crossref | Google Scholar |
Findlay SEG, Parr TB (2017) Chapter 24. Dissolved organic matter. In ‘Methods in stream ecology’, 3rd edn. (Eds GA Lamberti, FR Hauer) pp. 21–36. (Academic Press) doi:10.1016/B978-0-12-813047-6.00002-4
Finstad AG, Andersen T, Larsen S, Tominaga K, Blumentrath S, De Wit HA, Tømmervik H, Hessen DO (2016) From greening to browning: catchment vegetation development and reduced S-deposition promote organic carbon load on decadal time scales in Nordic lakes. Scientific Reports 6(1), 31944.
| Crossref | Google Scholar |
Frigstad H, Andersen GS, Trannum HC, McGovern M, Naustvoll L-J, Kaste Ø, Deininger A, Hjermann DØ (2023) Three decades of change in the Skagerrak coastal ecosystem, shaped by eutrophication and coastal darkening. Estuarine, Coastal and Shelf Science 283, 108193.
| Crossref | Google Scholar |
García-Martín EE, Sanders R, Evans CD, Kitidis V, Lapworth DJ, Rees AP, et al. (2021) Contrasting estuarine processing of dissolved organic matter derived from natural and human-impacted landscapes. Global Biogeochemical Cycles 35(10), e2021GB007023.
| Crossref | Google Scholar |
Green SA, Blough NV (1994) Optical absorption and fluorescence properties of chromophoric dissolved organic matter in natural waters. Limnology and Oceanography 39(8), 1903-1916.
| Crossref | Google Scholar |
Harvey ET, Kratzer S, Andersson A (2015) Relationships between colored dissolved organic matter and dissolved organic carbon in different coastal gradients of the Baltic Sea. AMBIO 44(S3), 392-401.
| Crossref | Google Scholar |
Hedges JI, Keil RG, Benner R (1997) What happens to terrestrial organic matter in the ocean? Organic Geochemistry 27(5-6), 195-212.
| Crossref | Google Scholar |
Hessen DO, Carroll J, Kjeldstad B, Korosov AA, Pettersson LH, Pozdnyakov D, Sørensen K (2010) Input of organic carbon as determinant of nutrient fluxes, light climate and productivity in the Ob and Yenisey Estuaries. Estuarine, Coastal and Shelf Science 88(1), 53-62.
| Crossref | Google Scholar |
Jaffé R, Boyer JN, Lu X, Maie N, Yang C, Scully NM, Mock S (2004) Source characterization of dissolved organic matter in a subtropical mangrove-dominated estuary by fluorescence analysis. Marine Chemistry 84(3–4), 195-210.
| Crossref | Google Scholar |
Karlsson J, Byström P, Ask J, Ask P, Persson L, Jansson M (2009) Light limitation of nutrient-poor lake ecosystems. Nature 460(7254), 506-509.
| Crossref | Google Scholar | PubMed |
Kartverket (2024) Se havnivå, tidevann og vannstand. Available at https://www.kartverket.no/til-sjos/se-havniva/resultat?id=365324&location=Fredrikstad [In Norwegian, verified 25 January 2024]
Kleven A (2021) Distribution of metals and organic material along a Norwegian river–ocean transect. Fordeling av metaller og organisk materiale langs en norsk elv – hav transekt. MSc thesis, Norges miljø- og biovitenskapelige universitet, Ås, Norway. Available at https://hdl.handle.net/11250/2827337 [In English with title and abstract in English and Norwegian]
Kritzberg ES (2017) Centennial-long trends of lake browning show major effect of afforestation. Limnology and Oceanography Letters 2(4), 105-112.
| Crossref | Google Scholar |
Kritzberg ES, Ekström SM (2012) Increasing iron concentrations in surface waters – a factor behind brownification? Biogeosciences 9, 1465-1478.
| Crossref | Google Scholar |
Kutser T, Alikas K, Kothawala DN, Köhler SJ (2015) Impact of iron associated to organic matter on remote sensing estimates of lake carbon content. Remote Sensing of Environment 156, 109-116.
| Crossref | Google Scholar |
Lalonde K, Middlestead P, Gélinas Y (2014) Automation of 13C/12C ratio measurement for freshwater and seawater DOC using high temperature combustion. Limnology and Oceanography: Methods 12(12), 816-829.
| Crossref | Google Scholar |
Larsen S, Andersen T, Hessen DO (2011) Predicting organic carbon in lakes from climate drivers and catchment properties. Global Biogeochemical Cycles 25(3), GB3007.
| Crossref | Google Scholar |
Lasareva EV, Parfenova AM, Romankevich EA, Lobus NV, Drozdova AN (2019) Organic matter and mineral interactions modulate flocculation across Arctic river mixing zones. Journal of Geophysical Research: Biogeosciences 124(6), 1651-1664.
| Crossref | Google Scholar |
Lee S-A, Kim T-H, Kim G (2020) Tracing terrestrial versus marine sources of dissolved organic carbon in a coastal bay using stable carbon isotopes. Biogeosciences 17(1), 135-144.
| Crossref | Google Scholar |
Lepistö A, Kortelainen P, Mattsson T (2008) Increased organic C and N leaching in a northern boreal river basin in Finland. Global Biogeochemical Cycles 22(3), GB3029.
| Crossref | Google Scholar |
Lyche Solheim A, Gundersen H, Mischke U, Skjelbred B, Nejstgaard JC, Guislain ALN, et al. (2024) Lake browning counteracts cyanobacteria responses to nutrients: evidence from phytoplankton dynamics in large enclosure experiments and comprehensive observational data. Global Change Biology 30(1), e17013.
| Crossref | Google Scholar |
Martín AP-S, Marhuenda-Egea FC, Bustamante MA, Curaqueo G (2023) Spectroscopy techniques for monitoring the composting process: a review. Agronomy 13, 2245.
| Crossref | Google Scholar |
McGovern M, Pavlov AK, Deininger A, Granskog MA, Leu E, Søreide JE, Poste AE (2020) Terrestrial inputs drive seasonality in organic matter and nutrient biogeochemistry in a high Arctic fjord system (Isfjorden, Svalbard). Frontiers in Marine Science 7, 542563.
| Crossref | Google Scholar |
Monteith DT, Stoddard JL, Evans CD, De Wit HA, Forsius M, Høgåsen T, Wilander A, Skjelkvåle BL, Jeffries DS, Vuorenmaa J, Keller B, Kopácek J, Vesely J (2007) Dissolved organic carbon trends resulting from changes in atmospheric deposition chemistry. Nature 450(7169), 537-540.
| Crossref | Google Scholar | PubMed |
Mustaffa NIH, Kallajoki L, Hillebrand H, Wurl O, Striebel M (2020) Sea surface phytoplankton community response to nutrient and light changes. Marine Biology 167(9), 123.
| Crossref | Google Scholar |
Myers-Smith IH, Kerby JT, Phoenix GK, Bjerke JW, Epstein HE, Assmann JJ, et al. (2020) Complexity revealed in the greening of the Arctic. Nature Climate Change 10(2), 106-117.
| Crossref | Google Scholar |
Nebbioso A, Piccolo A (2013) Molecular characterization of dissolved organic matter (DOM): a critical review. Analytical and Bioanalytical Chemistry 405(1), 109-124.
| Crossref | Google Scholar | PubMed |
Norges vassdrags- og energidirektorat (2023) Solbergfoss (2.605.0) measurement data. (NVE) Available at https://sildre.nve.no/station/2.605.0 [Verified 11 May 2023]
Ogawa H, Tanoue E (2003) Dissolved organic matter in oceanic waters. Journal of Oceanography 59(2), 129-147.
| Crossref | Google Scholar |
Olsen Y, Agustí S, Andersen T, Duarte CM, Gasol JM, Gismervik I, et al. (2006) A comparative study of responses in plankton food web structure and function in contrasting European coastal waters exposed to experimental nutrient addition. Limnology and Oceanography 51, 488-503.
| Crossref | Google Scholar |
Opdal AF, Lindemann C, Aksnes DL (2019) Centennial decline in North Sea water clarity causes strong delay in phytoplankton bloom timing. Global Change Biology 25(11), 3946-3953.
| Crossref | Google Scholar | PubMed |
Opdal AF, Andersen T, Hessen DO, Lindemann C, Aksnes DL (2023) Tracking freshwater browning and coastal water darkening from boreal forests to the Arctic Ocean. Limnology and Oceanography Letters 8, 611-619.
| Crossref | Google Scholar |
Opsahl S, Benner R (1997) Distribution and cycling of terrigenous dissolved organic matter in the ocean. Nature 386(6624), 480-482.
| Crossref | Google Scholar |
Osburn CL, Stedmon CA (2011) Linking the chemical and optical properties of dissolved organic matter in the Baltic–North Sea transition zone to differentiate three allochthonous inputs. Marine Chemistry 126(1–4), 281-294.
| Crossref | Google Scholar |
Otero E, Culp R, Noakes JE, Hodson RE (2003) The distribution and δ13C of dissolved organic carbon and its humic fraction in estuaries of southeastern USA. Estuarine, Coastal and Shelf Science 56(5–6), 1187-1194.
| Crossref | Google Scholar |
O’Leary MH (1988) Carbon isotopes in photosynthesis. BioScience 38(5), 328-336.
| Crossref | Google Scholar |
Pedersen EJ, Miller DL, Simpson GL, Ross N (2019) Hierarchical generalized additive models in ecology: an introduction with mgcv. PeerJ 7, e6876.
| Crossref | Google Scholar |
Peterson B, Fry B, Hullar M, Saupe S, Wright R (1994) The distribution and stable carbon isotopic composition of dissolved organic carbon in estuaries. Estuaries 17, 111-121.
| Crossref | Google Scholar |
Poste A, Kaste Ø, Frigstad H, de Wit H, Harvey T, Valestrand L, Deininger A, Bryntesen T, Delpech L, Christensen G (2021) The impact of the spring 2020 snowmelt floods on physicochemical conditions in three Norwegian river–fjord–coastal systems. NIVA Report number 7651-2021, Norsk institutt for vannforskning, Oslo, Norway.
Regnier P, Resplandy L, Najjar RG, Ciais P (2022) The land-to-ocean loops of the global carbon cycle. Nature 603(7901), 401-410.
| Crossref | Google Scholar | PubMed |
Sampedro-Avila JE, Lu K, Xue J, Liu Z, Maske H (2024) The chemical characteristics and mixing behaviors of particulate organic matter from small subtropical rivers in coastal Gulf of Mexico. Estuarine, Coastal and Shelf Science 299, 108664.
| Crossref | Google Scholar |
Schultze S, Andersen T, Hessen DO, Ruus A, Borgå K, Poste AE (2022) Land-cover, climate and fjord morphology drive differences in organic matter and nutrient dynamics in two contrasting northern river–fjord systems. Estuarine, Coastal and Shelf Science 270, 107831.
| Crossref | Google Scholar |
Spilling K, Asmala E, Haavisto N, Haraguchi L, Kraft K, Lehto A-M, Lewandowska AM, Norkko J, Piiparinen J, Seppälä J, Vanharanta M, Vehmaa A, Ylöstalo P, Tamminen T (2022) Brownification affects phytoplankton community composition but not primary productivity in eutrophic coastal waters: a mesocosm experiment in the Baltic Sea. Science of The Total Environment 841, 156510.
| Crossref | Google Scholar |
Stedmon CA, Markager S, Kaas H (2000) Optical properties and signatures of chromophoric dissolved organic matter (CDOM) in Danish coastal waters. Estuarine, Coastal and Shelf Science 51(2), 267-278.
| Crossref | Google Scholar |
Thrane J-E, Hessen DO, Andersen T (2014) The absorption of light in lakes: negative impact of dissolved organic carbon on primary productivity. Ecosystems 17(6), 1040-1052.
| Crossref | Google Scholar |
Tranvik LJ, Sieburth JM (1989) Effects of flocculated humic matter on free and attached pelagic microorganisms. Limnology and Oceanography 34(4), 688-699.
| Crossref | Google Scholar |
Wilson RJ, Heath MR (2019) Increasing turbidity in the North Sea during the 20th Century due to changing wave climate. Ocean Science 15(6), 1615-1625.
| Crossref | Google Scholar |
Wood SN (2017) ‘Generalized additive models: an introduction with R’, 2nd edn. (Chapman and Hall) 10.1201/9781315370279
Xu H, Guo L (2017) Molecular size-dependent abundance and composition of dissolved organic matter in river, lake and sea waters. Water Research 117, 115-126.
| Crossref | Google Scholar | PubMed |
Xu H, Guo L (2018) Intriguing changes in molecular size and composition of dissolved organic matter induced by microbial degradation and self-assembly. Water Research 135, 187-194.
| Crossref | Google Scholar | PubMed |