Successful artificial incubation and juvenile-rearing of dropped eggs of a critically endangered freshwater crayfish (Cherax tenuimanus)
Rodney E. Duffy A * , Blaine Hodgson A , Amber Quinn A and Marcus Newman A
A * , Blaine Hodgson A , Amber Quinn A and Marcus Newman A
A
Abstract
One-third of the world’s freshwater crayfish species face extinction risk, necessitating extreme and urgent conservation measures such as captive rearing. Little information exists on these measures for most threatened species. The hairy marron, Cherax tenuimanus (Parastacidae), is a critically endangered freshwater crayfish endemic to the Margaret River in south-western Western Australia. C. tenuimanus is being rapidly replaced by the smooth marron, Cherax cainii, a translocated congener, causing its progressive decline in range, and now it faces imminent extinction in the wild. Attempts at captive breeding have resulted in few occurrences of egg laying of C. tenuimanus, which ended in complete loss of the clutch as eggs were progressively dropped by the female. To assess the potential of dropped eggs in captive rearing, we trialled ex situ artificial incubation techniques by using eggs dropped from a captively held female. Of 41 dropped eggs, 85% were successfully hatched in suspension upwellers, and 74% of the subsequent juveniles were reared successfully for 111 days in a purpose-built recirculating aquaculture system. We describe our protocols and facilities in detail because captive rearing of dropped eggs was demonstrated to be a viable option for improved success of captive reproduction. These approaches may be applicable for conserving similar crayfish species.
Keywords: Astacidea, captive breeding, conservation, egg disinfection, hairy marron, introduced species, Margaret River, Western Australia.
Introduction
One in three freshwater crayfish species worldwide is at risk of extinction from threats such as water extraction, pollution, habitat loss and fragmentation, and disease (Richman et al. 2015; O’Hea Miller et al. 2024). In Australia, the main threats are climate change, harvesting, agriculture, invasive species and fire (Richman et al. 2015; Kirsch et al. 2024). Although among the most threatened species, Australian freshwater crayfish are seldom adequately conserved in protected areas (Coughran and Furse 2012), in part owing to endemism to small regions and even single rivers (De Graaf et al. 2009; McCormack 2013). The increasing pressure of multiple threatening processes on Australian freshwater crayfish, combined with the lack of adequate protection of most wild populations, means that alternative approaches to long-term conservation are required (Richman et al. 2015).
One alternative approach that has attracted attention for some freshwater crayfish species is captive breeding and ex situ artificial incubation of eggs to increase populations for conservation (Nakata et al. 2004) or to limit transmission of pathogens (Edgerton and Owens 1997; Kozák et al. 2023). Ex situ artificial incubation techniques have been developed for commercial aquaculture of Cherax species, such as yabbies (C. destructor) (Leonard et al. 2001), redclaw (C. quadricarinatus) (King 1993; Shun et al. 2020; Cheng et al. 2022) and smooth marron (C. cainii) (Henryon and Purvis 2000) with promising results. Various bespoke systems and prophylactic treatments have been used and summarised in Table 1, but there is no consensus on a single suitable approach. Furthermore, these studies have little information on post-hatch handling and early-stage juvenile rearing, limiting their value for captive breeding through to reproductively mature adults.
| Species and context | Result | References | |
|---|---|---|---|
| Cherax cainii – artificial incubation potential | A shaking apparatus and penicillin treatment was used to incubate fertilised eggs removed from females, there was low survivorship, owing to unstable power supply for equipment in a remote location. The high attention needed for this method was unsuitable for large-scale production | Morrissy (1976) | |
| Cherax quadricarinatus – artificial incubation potential | Hatching occurred only between 22 and 32°C. Surface treatment of eggs was necessary | King (1993) | |
| Cherax tenuimanus – artificial incubation potential | An artificial incubator was a suitable replacement for maternal care, with 89% of eggs hatching and developing to juveniles | Henryon and Purvis (2000) | |
| Cherax destructor – effectiveness of artificial incubation v. maternal brooding | Using a suspension upweller achieved survival rates averaging 93%, which was equal to or better than from maternal incubation | Leonard et al. (2001) | |
| Cherax quadricarinatus – addressing low egg hatching rate and asynchronous hatching | Alcohol as a disinfectant promoted hatching rate, densities of 300 and 600 eggs per incubator resulted in higher hatching and survival rates than 900 eggs per incubator; 600 eggs per incubator resulted in 82.05% hatching rate, 55.12% survival rate | Shun et al. (2020) | |
| Cherax quadricarinatus – developed a recirculating mechanical pulling device to explore egg stages, egg densities, attachments and incubator sizes | Egg development was sorted into three colour stages, the groups with seven pairs of appendages (orange egg) and well-formed eye pigment (orange egg with eye pigments) were significantly higher than egg-nauplius (yellow egg) in hatching and survival rates. Hatching rates of 300 eggs per incubator were significantly higher than 700 eggs per incubator. Survival rates of 300 or 500 eggs per incubator were significantly higher than 700 eggs per incubator. Sponge attachment significantly increased survival rate. Survival rate of groups with 2.5- or 3-cm width of the upper end of boxes were significantly higher than 4 cm | Cheng et al. (2022) | |
| Cherax quadricarinatus – the effects of water parameters on eggs during artificial incubation | Hatching rate less affected by nitrite, pH and ammonia nitrogen, survival rates were affected by nitrite, ammonia nitrogen and pH | Yong-chun et al. (2022) | |
| Cherax quadricarinatus – effects of disinfectants, sponge densities, water circulation rates, and vibration frequency on hatching % and survival % of embryos | A disinfectant solution comprising an essential oil base solution, Zn-superoxide dismutase, trehalose, bovine serum protein, and astaxanthin can be used instead of formaldehyde. Medium-density sponges of a length of 4.5 cm improved survival percentage. No significant difference in hatching percentage between water circulation rates. Survival percentage of group exposed to 11 vibrations min−1 was significantly higher than that of groups exposed to two other frequencies | Shun et al. (2023) |
Six species of Cherax are endemic to southern Western Australia (De Graaf et al. 2009). The critically endangered (Austin and Bunn 2010) hairy marron, Cherax tenuimanus Smith has a restricted distribution, being endemic to a single river, the Margaret River (Austin and Ryan 2002). The more widely distributed congener, the smooth marron (C. cainii), is a popular recreationally fished and aquaculture species. It does not naturally occur in Margaret River (Austin and Ryan 2002); however, its presence was detected in samples collected in 1985, which was attributed to its introduction by one or more translocation events (Austin and Ryan 2002; Duffy et al. 2014). Following this introduction, C. tenuimanus has been rapidly replaced by C. cainii throughout Margaret River, owing to reproductive interference and competition (Bunn 2004; De Graaf et al. 2009). Hybridisation and introgression between the two species has caused genetically pure C. tenuimanus to be rare in a mixed population (Bunn et al. 2008; Guildea et al. 2015). A small population persists in the wild (DPIRD, unpubl. data) and a very small captive population has been held at various sites since the early 2000s. Captive breeding programs have had varying levels of success. Semi-intensive production techniques developed for C. cainii have not produced juvenile C. tenuimanus, as a result, the captive population has gradually declined because of natural mortality (DPIRD, unpubl. data). Even though intensive efforts at breeding in recirculating systems have yielded clutches of eggs, no juveniles have been produced because the eggs have consistently been dropped by the ovigerous females (Lette 2020; DPIRD, unpubl. data) and die without maternal care.
Perth zoo holds a small population of C. tenuimanus that have been confirmed, through genetic testing, to be pure C. tenuimanus. In December 2022, a captive female C. tenuimanus held at the zoo mated and produced a clutch of fertilised eggs. When some of these eggs were dropped, we opportunistically recovered the eggs, determined that they were viable and attempted artificial incubation, followed by juvenile rearing, by using techniques applied to other Cherax species (Henryon and Purvis 2000; Cheng et al. 2022; Table 1). Our objective was to develop practical protocols for incubating dropped eggs and then rearing hatchlings beyond a 100-day grow-out period to assess the potential for this approach to conserving C. tenuimanus.
Materials and methods
Obtaining, disinfecting and assessing the dropped eggs and larvae
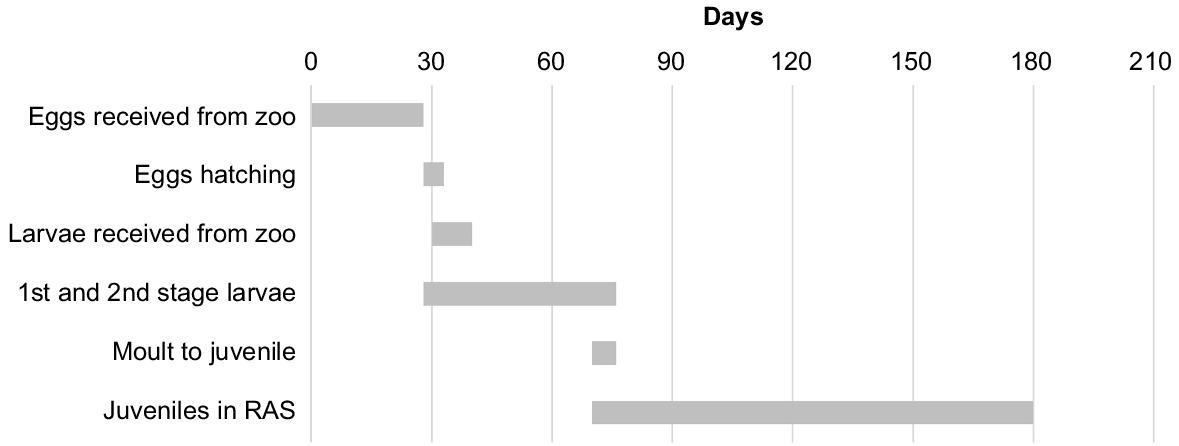
From the date a clutch was observed (27 November 2022) through to the final release of juveniles from her pleopods (24 March 2023), the berried female C. tenuimanus, originating from the wild in 2016 and currently located at Perth Zoo, was checked daily. Dropped eggs and larvae (Fig. 1a, b) were detected between 29 December 2022 and 10 February 2023 (Fig. 2) and were transferred within 24 h to the West Australian Fisheries and Marine Research Laboratories. During transport, eggs or larvae were placed in a closed 250-mL glass Schott jar containing source water and placed in a small cooler with a frozen ice brick. Temperature of the water ranged between 16 and 20°C.
Stages of C. tenuimanus during artificial incubation (a) egg, (b) first-stage larva with egg casing, (c) second-stage larva, and (d) juvenile.

Gantt chart of dropped C. tenuimanus egg development. Day 0 is 29 December 2022. The study was terminated after 180 days. RAS, recirculating aquaculture system.
On arrival at the research laboratory, eggs were disinfected with 75% ethanol solution for 60 s. This was repeated every second day for 21 days. This procedure was then changed to a salt water bath (100 g L−1 solution, API aquarium salt) for a period of 5 min every second day for 14 days. During each disinfection event, the egg was observed under a stereo microscope (Nikon SMZ 745T) to determine egg development. Each egg was tracked with a development schedule reported for C. destructor (Sandeman and Sandeman 1991), with particular focus on the changing proportion of yolk and presence of a heartbeat observed in the egg. Dead eggs were removed using forceps, preserved in formalin solution and sent off for disease diagnostic testing. The disinfection process was continued for the first and second larval stages, through which Cherax spp. progress prior to reaching the first juvenile instar.
Artificial incubation of eggs
We incubated eggs by using Ziss Aqua Egg Tumbler suspension upwellers (ZET-55, Fig. 3) because these had been trialled successfully by the authors with Cherax cainii eggs, as well as C. quadricarinatus by Shun et al. (2020). A maximum of five eggs were placed in each of the 12 incubators used. Eggs were incubated with those dropped on the same or a similar date. Artificial incubators were distributed among four 16-L glass aquaria (36 × 26 × 22 cm), aerated at a rate sufficient to keep the eggs continuously but gently moving. Incubators and aquaria were disinfected with a 12.5% sodium hypochlorite solution prior to use.
The trial was undertaken in a climate-controlled room set at 18–20°C, with a photoperiod that changed to replicate the natural diel cycle at that time of year. For biosecurity, the room was restricted access, with a Virkon footbath and handwashing facilities on entry; laboratory coats were mandatory. Aquaria were filled with tap water that had been filtered through a carbon filter (Puretec EC011) and aerated for at least 24 h prior to use. This tap water did not contain chloramines, as confirmed by test strips (AquaChek). Water quality was monitored daily for pH, ammonia, nitrate, nitrite and hardness (API test kits). If ammonia or nitrite concentrations exceeded 0.1 mg L−1 or nitrate concentrations exceeded 10 mg L−1, half the water in the aquaria was changed. Salinity was also tested daily by using a Eutech EC-PCSTestr35 Multi-Purpose Meter and maintained close to 1 g L−1 by using aquarium salt (API).
Rearing juveniles (hatchlings) in a recirculating aquaculture system
As soon as second-stage larvae had moulted to the first juvenile instar (Fig. 1c, d), they were placed into one of two purpose-built recirculating aquaculture systems (RAS, Fig. 4a). Each RAS consisted of a 200-L sump, Fluval FX6 canister filter, ZKSJ Blue70-12000 DC pump and shelving, each with fifty-four 5-L clear plastic tubs. Juveniles were randomly assigned to a tub and housed individually with a ‘hide’ (Fig. 4b) made of 12-mm diameter black plastic hose, which was enlarged as the animals grew. Each system was filled with carbon-filtered (PureTec EC011) tap water and a probiotic (0.25 g, Sanolife MIC) was added biweekly. The system was cycled for 14 days to establish suitable biological filtration capacity prior to addition of animals. Nitrate, nitrite, ammonium, carbonate hardness, general hardness, pH, calcium, free chlorine, chlorine and salinity were tested daily (Table 2), and salinity was maintained close to 1 g L−1 by using aquarium salt (API).
| Ammonia (mg L−1) | General hardness (mg L−1) | Carbonate hardness (mg L−1) | pH | Nitrite (mg L−1) | Nitrate (mg L−1) | Salinity (g L−1) | Calcium (mg L−1) | Chlorine (mg L−1) | Free chlorine (mg L−1) | |
|---|---|---|---|---|---|---|---|---|---|---|
| 0–0.25 | 125.3–214.8 | 107.4–161.1 | 7.8–8.2 | 0–0.25 | 0–10 | 0.81–1.17 | 60–120 | 0 | 0 |
Juveniles were randomly assigned feed types and fed either frozen peas, corn or crushed lupin, renewed as needed. Sinking spirulina algae wafers, Aquatopia freshwater crayfish pellets, Repashy gel premix and AquaOne frozen bloodworms were also provided on different days, according to a weekly schedule. Each morning, faeces and other waste were manually siphoned from the tubs, and water was changed twice weekly. Filters and pumps were cleaned periodically to maintain suitable water quality.
Results
Survival of eggs
Of 41 eggs collected from a single berried Cherax tenuimanus, 35 (85%) were successfully artificially incubated and hatched. Five eggs died during the experiment, on Days 15, 17, 23 (two eggs) and 25. Disease diagnostic investigations showed bacterial infection or physical damage in the handling protocol as the likely cause of death. One egg was dead on arrival at the research laboratory.
Development time of eggs
Mating and egg extrusion by the female occurred on 27 November 2022. The first egg received at the research laboratory (29 December 2022, Fig. 2) was staged at 35–40% according to the embryo development of C. destructor, defined by the size of the antennae and antennules, appearance of maxilla and maxilliped buds and the presence of a heartbeat (Sandeman and Sandeman 1991). Eggs hatched between 25 January 2023 and 31 January 2023 (Fig. 2), between 59 and 65 days post-laying.
Survival and growth of hatchlings and juveniles
There was high survival of first-stage (98%) and second-stage (90%) larvae in the incubator. The two stages were separated by a decrease in dorsal hunchback and store of yolk, stalked eyes and elongation of antennules, antennae and abdomen (Fig. 1b, c; Sandeman and Sandeman 1991). After transferring the juveniles (immature miniature adult crayfish; Fig. 1d) to the RAS (Fig. 2), 31 (84%) of them survived the final grow-out phase. After 180 days, the study was terminated, and the 31 animals were taken offsite for further rearing. The final mean weight of the juveniles was 2.71 g (±0.718 s.d.), with a range of 1.57–4.16 g. On the basis of survival, all diets (i.e. frozen peas, corn or crushed lupin) were deemed suitable for juvenile rearing.
Discussion
Our work indicated that dropped eggs of the critically endangered Cherax tenuimanus can be successfully artificially incubated with a high level of survival. From 41 eggs dropped by a single berried C. tenuimanus, 35 (85%) were hatched using ZISS incubators. Of 42 larvae (DPIRD hatched animals and a subsequent 7 larvae from the zoo), 31 (74%) were reared through first and second larval stages in the same ZISS incubators and then survived as juveniles in a RAS.
Artificial incubation of dropped eggs and rearing of juveniles in a RAS has the potential to increase captive populations of C. tenuimanus. In captivity, this species has demonstrated regular clutch failure and dropped eggs, severely hampering conservation efforts to build an adequate captive population (Lette 2020; DPIRD, unpubl. data). This method also offers a suitable alternative to maternal incubation, provided that eggs can be obtained from the female without negative effects. Freshwater crayfish embryos develop outside the females’ body and therefore the eggs are suited to artificial incubation (McLay and Van den Brink 2016). Suspension incubators can imitate the environment provided in a female’s brood chamber to provide conditions for successful development. The constant flow inside the incubators mimics the female’s oscillations of her pleopods to ‘fan’ her eggs, providing freshwater, oxygen and preventing microbial establishment (Leonard et al. 2001). Brooding females groom and clean eggs (Bauer 2013) and are known to discard unhealthy or dead embryos (Fernández et al. 2020).
During the incubation period, the issue of greatest concern was the presence of potentially undesirable organisms as freshwater crayfish eggs are susceptible to infection and disease (Romero and Jiménez 1997). Epistylis (caused by a ciliated protozoan) and temnocephalan flatworms were visible under the microscope on multiple eggs throughout the experiment, albeit at low levels. Various antifungal treatments have been previously used during artificial incubation of freshwater crayfish eggs (Henryon and Purvis 2000; Carral et al. 2009; Shun et al. 2020). However, effective agents such as formaldehyde and malachite green show concerning health impacts toward humans and the aquatic environment (Sapkota et al. 2008; Carral et al. 2009). Salt solution is a suitable non-toxic alternative, assisting the treatment of parasites and improving survival of early-stripped eggs (Seemann et al. 2014; Rida et al. 2021). Previous studies have used salt solution at concentrations of up to 15–16 g L−1 (Seemann et al. 2014). However, on the basis of advice from a commercial producer (Dr Lisa Elliott, pers. comm., Australian Redclaw Crayfish Hatchery), who successfully trialled the dosage on C. quadricarinatus, we demonstrated that C. tenuimanus eggs and first-stage larvae can be treated with 100 g L−1 salt water for up to 5 min, with 100% survival and highly effective results.
The previous lack of success in captive breeding of C. tenuimanus (Lette 2020; DPIRD, unpubl. data) begs the following question: should artificial incubation be prioritised over maternal care for conservation of the critically endangered C. tenuimanus? Our study was highly successful; however, we acknowledge the lack of replication of ovigerous females. We counter this criticism with the observations that this species is so rare that it is unethical to harvest females from the wild for a trial, nor could we access dropped eggs from other females because no other breeding in captivity occurred at the time of the study.
This study entailed intensive work on comparatively few eggs and hatchlings. To use our methods to incubate a whole clutch of potentially hundreds of eggs and yet maintain low densities of eggs per incubator would require substantial resources. This may be warranted for a species such as C. tenuimanus, where even a small increase in numbers represents a substantial increase in population. Future work should look at increasing densities in incubators (Shun et al. 2020; Cheng et al. 2022) as well as developing a protocol for successful grow-out to mature adults.
In conclusion, our work indicated that dropped eggs of this critically endangered species can be successfully artificially incubated and reared, and that the protocols and facilities described show promise for arresting the mortality of eggs dropped in captivity. Despite the success and potential benefits of this approach to ensure the persistence of this species, the main limiting factor remains the low number of successful pairings in captivity over multiple decades. Nonetheless, for threatened crayfish species such as C. tenuimanus where each egg or larvae represent a substantial addition to the population, captive breeding as described may provide a crucial tool for maintaining and enhancing the population.
Data availability
The data that support this study will be shared upon reasonable request to the corresponding author.
Declaration of funding
Funding was provided by the Department of Primary Industries and Regional Development, State Government of Western Australia.
Acknowledgements
The authors acknowledge the extensive collaboration with the staff at Perth Zoo required to successfully implement this research. We are grateful for the generous advice provided by Lisa Elliott of the Australian Redclaw Crayfish Hatchery that guided prophylactic treatment and rearing. We also thank staff at the Department of Primary Industries and Regional Development: Roger Barnard, Andrew Beer and the Aquaculture group for space and advice on design, construction and maintenance of systems; the Disease and Diagnostic Laboratories for disease testing and advice regarding biosecurity protocols; Sue Martin for enthusiasm and assistance in system construction, maintenance and rearing; and Andrew Beer, Druimé Nolan, Dan Yeoh and Andrew Boulton for comments on earlier drafts of this paper. Thanks go to the anonymous reviewers for helping strengthen the paper.
References
Austin CM, Ryan SG (2002) Allozyme evidence for a new species of freshwater crayfish of the genus Cherax Erichson (Decapoda: Parastacidae) from the south-west of Western Australia. Invertebrate Systematics 16(3), 357-367.
| Crossref | Google Scholar |
Austin CM, Bunn JJS (2010) Cherax tenuimanus. In ‘IUCN Red List of Threatened Species 2010’. e.T4618A11033949. (International Union for Conservation of Nature and Natural Resources) Available at https://www.iucnredlist.org/species/4618/11033949
Bunn JJS, Koenders A, Austin CM, Horwitz P (2008) Identification of hairy, smooth and hybrid marron (Decapoda: Parastacidae) in the Margaret River: morphology and allozymes. Freshwater Crayfish 16(1), 113-121.
| Crossref | Google Scholar |
Carral JM, González A, Celada JD, Sáez-Royuela M, Melendre PM, González R, Garcia V (2009) Antifungal treatments in artificial incubation of crayfish eggs (Pacifastacus leniusculus, Astacidae): searching for alternatives to formaldehyde. Knowledge and Management of Aquatic Ecosystems 16, 394-395.
| Google Scholar |
Cheng S, Wei Y-C, Chi M-L, Li F, Zheng J-B, Liu S-L, Jia Y-Y, Liu Y-N, Gu Z-M, Wang D-L, Sun L-H (2022) Mass artificial incubation of redclaw crayfish eggs in a recirculating mechanical pulling device. Aquaculture Research 53, 2923-2929.
| Crossref | Google Scholar |
Coughran J, Furse JM (2012) Conservation of freshwater crayfish in Australia. Crustacean Research Special(2012), 25-34.
| Crossref | Google Scholar |
De Graaf M, Lawrence C, Vercoe P (2009) Rapid replacement of the critically endangered hairy marron by the introduced smooth marron (Decapoda, Parastacidae) in the Margaret River (Western Australia). Crustaceana 82(11), 1469-1476.
| Crossref | Google Scholar |
Duffy R, Ledger J, Dias J, Snow M (2014) The critically endangered hairy marron, Cherax tenuimanus Smith, 1912: a review of current knowledge and actions required to prevent extinction of a species. Journal of the Royal Society of Western Australia 97, 297-306.
| Google Scholar |
Edgerton B, Owens L (1997) Age at first infection of Cherax quadricarinatus by Cherax quadricarinatus bacilliform virus and Cherax giardiavirus-like virus, and production of putative virus-free crayfish. Aquaculture 152(1–4), 1-12.
| Crossref | Google Scholar |
Guildea C, Hitchen Y, Duffy R, Dias PJ, Ledger JM, Snow M, Kennington WJ (2015) Introgression threatens the survival of the critically endangered freshwater crayfish Cherax tenuimanus (Decapoda: Parastacidae) in the wild. PLoS ONE 10(3), e0121075.
| Crossref | Google Scholar |
Henryon M, Purvis IW (2000) Eggs and hatchlings of the freshwater crayfish, marron (Cherax tenuimanus), can be successfully incubated artificially. Aquaculture 184(3–4), 247-254.
| Crossref | Google Scholar |
King CR (1993) Egg development time and storage for redclaw crayfish Cherax quadricarinatus von Martens. Aquaculture 109(3–4), 275-280.
| Crossref | Google Scholar |
Kirsch G, Anjos AG, Ruaro R, Abrantes N, Figueiredo BRS (2024) Ashes in freshwater ecosystems: a systematic review of their impacts on fish. Water, Air, & Soil Pollution 235(8), 521.
| Crossref | Google Scholar |
Kozák P, Erol KG, Uzunmehmetoğlu OY, Tangerman M, Mojžišová M, Özkök R, Kouba A, Çınar Ş, Petrusek A (2023) Short-term artificial incubation before hatching limits vertical transmission of Aphanomyces astaci from chronically infected females of a host species susceptible to crayfish plague. Aquaculture 569, 739373.
| Crossref | Google Scholar |
Leonard BV, Lennard WA, Kildea DG (2001) A method for testing the effectiveness of artificial incubation of eggs vs. maternal brooding in the freshwater crayfish Cherax destructor (Decapoda: Parastacidae). Aquaculture 195(3–4), 299-309.
| Crossref | Google Scholar |
McCormack RB (2013) Conservation of imperiled crayfish, Euastacus dharawalus (Decapoda: Astacidea: Parastacidae), from the Southern Highlands of New South Wales, Australia. Journal of Crustacean Biology 33(3), 432-439.
| Crossref | Google Scholar |
Nakata K, Matsubara H, Goshima S (2004) Artificial incubation of Japanese crayfish (Cambaroides japonicus) eggs by using a simple, easy method with a microplate. Aquaculture 230(1–4), 273-279.
| Crossref | Google Scholar |
O’Hea Miller SB, Davis AR, Wong MYL (2024) The impacts of invasive crayfish and other non-native species on native freshwater crayfish: a review. Biology 13(8), 610.
| Crossref | Google Scholar | PubMed |
Richman NI, Böhm M, Adams SB, Alvarez F, Bergey EA, Bunn JJ, Burnham Q, Cordeiro J, Coughran J, Crandall KA, Dawkins KL, DiStefano RJ, Doran NE, Edsman L, Eversole AG, Füreder L, Furse JM, Gherardi F, Hamr P, Holdich DM, Horwitz P, Johnston K, Jones CM, Jones JPG, Jones RL, Jones TG, Kawai T, Lawler S, López-Mejía M, Miller RM, Pedraza-Lara C, Reynolds JD, Richardson AMM, Schultz MB, Schuster GA, Sibley PJ, Souty-Grosset C, Taylor CA, Thoma RF, Walls J, Walsh TS, Collen B (2015) Multiple drivers of decline in the global status of freshwater crayfish (Decapoda: Astacidea). Philosophical Transactions of the Royal Society of London – B. Biological Sciences 370, 20140060.
| Crossref | Google Scholar |
Rida R, Zein-Eddine R, Kreydiyyeh S, Garza de Yta A, Saoud IP (2021) Influence of salinity on survival, growth, hemolymph osmolality, gill sodium potassium ATPase activity, and sodium potassium chloride co-transporter expression in the redclaw crayfish Cherax quadricarinatus. Journal of the World Aquaculture Society 52(2), 466-474.
| Crossref | Google Scholar |
Romero X, Jiménez R (1997) Epistylis sp. (Ciliata: Peritrichida) infestation on the eggs of berried red claw crayfish Cherax quadricarinatus females in Ecuador. Journal of the World Aquaculture Society 28(4), 432-435.
| Crossref | Google Scholar |
Sandeman R, Sandeman D (1991) Stages in the development of the embryo of the fresh-water crayfish Cherax destructor. Roux’s Archives of Developmental Biology 200, 27-37.
| Crossref | Google Scholar | PubMed |
Sapkota A, Sapkota AR, Kucharski M, Burke J, McKenzie S, Walker P, Lawrence R (2008) Aquaculture practices and potential human health risks: current knowledge and future priorities. Environment International 34(8), 1215-1226.
| Crossref | Google Scholar | PubMed |
Seemann UB, Lorkowski K, Schiffer M, Hörterer C, Slater MJ, Buck BH (2014) Survival of early stripped eggs of the noble crayfish, Astacus astacus, and effects of saline solution during artificial incubation. Freshwater Crayfish 20(1), 1-10.
| Crossref | Google Scholar |
Shun C, Yong-Yi J, Mei-Li C, Shi-Li L, Jian-Bo Z, Dan-Li W, Zhi-Min G (2020) The exploration of artificial incubation of Cherax quadricarinatus eggs. Aquaculture 529, 735576.
| Crossref | Google Scholar | PubMed |
Shun C, Wen-Ping J, Shi-Li L, Mei-Li C, Jian-Bo Z, Yi-Nuo L, Xiao-Ying H, Miao P, Fei L, Dan-Li W (2023) Effects of disinfectants, sponge densities, water circulation rates, and vibration frequency on the artificial incubation of redclaw crayfish embryos. Aquaculture 570, 739374.
| Crossref | Google Scholar |
Yong-chun W, Shun C, Dan-Li W, Mei-Li C, Jian-Bo Z, Yong-Yi J, Fei L, Shi-Li L, Yi-Nuo L, Zhi-Min G (2022) The effect of ammonia nitrogen, nitrite and pH on artificial incubation of red claw crayfish Cherax quadricarinatus eggs and growth of juveniles. Aquaculture Research 53, 3788-3796.
| Crossref | Google Scholar |




