
Australian Journal of Chemistry
Volume 68 Number 1 2015
RESEARCH FRONT: Fluorine Chemistry
The Editor-in-Chief gives an overview of publishing in 2014 and wishes the readers a very happy, enjoyable, and successful new year.
Fluorine chemistry is an important discipline in the Australian research landscape, because it interfaces with many other research areas. This foreword introduces a collection of original research papers and review articles on diverse aspects of fluorine chemistry, with contributing authors from around Australia and overseas.
CH14198Bioisosteric Fluorine in the Clandestine Design of Synthetic Cannabinoids
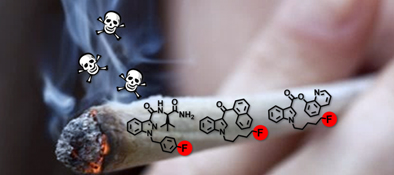
Over the past decade, a plethora of indole-based synthetic cannabinoids (SCs) have emerged with structures inspired by scientific literature. The latest generation of SCs frequently use bioisosteric fluorine-for-hydrogen replacement. This highlight explores the evolution of SCs to this latest generation of fluorinated SCs, where accumulating evidence indicates increased health concerns.
CH14356Biosynthetic Incorporation of Fluorinated Amino Acids into Peptides and Proteins
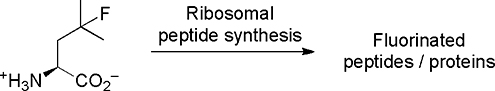
Native and engineered protein biosynthetic machinery has been used to incorporate many fluorinated amino acids into peptides, conferring them with a diverse variety of interesting properties. This biochemical synthetic approach should routinely be assessed as an alternative to traditional solid- and solution-phase peptide synthesis.
CH14165Fluoropolymers: Origin, Production, and Industrial and Commercial Applications

Fluoropolymers have had a major industrial and commercial impact since their discovery almost 80 years ago. From development during the Manhattan project and the introduction of Teflon, to today’s modern polymers, these unique materials have found use in almost every area of society. This review summarises the history of fluoropolymers, their production, and modern day applications.
CH14456Probing the Mode of Neurotransmitter Binding to GABA Receptors Using Selectively Fluorinated GABA Analogues
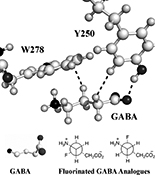
Fluorinated analogues of GABA have been developed to probe the conformation of GABA that activates GABA receptors. We have benchmarked the pharmacology of these analogues with the crystal structure of GABA bound to the GABAB receptor to develop an accurate pharmacophore comparing neurotransmitter binding to individual GABA receptors.
CH14117Effect of Modifying the Anion of an Ionic Liquid on the Outcome of an SN2 Process
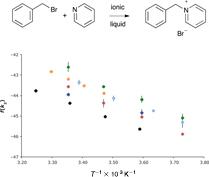
A general ionic liquid effect is observed for the substitution reaction shown above for ionic liquids containing different anions, including [N(CF3SO2)2]–, [PF6]–, and [BF4]–. In all cases, an increase in rate constant is seen relative to acetonitrile though the microscopic origin of these changes differs with the anion used.
CH14097Trifluoromethylphenylcarbenes. Carbene-Carbene Interconversion on the Singlet Energy Surface and Rearrangement to Trifluorobenzocyclobutene, Trifluorostyrene, and Trifluoromethylfulvenallenes
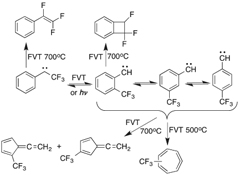
The four isomeric α-, ortho-, meta-, and para-trifluoromethylphenylcarbenes interconvert thermally and in part photochemically. Electron spin resonance investigations indicate that carbene-carbene rearrangement takes place on the singlet energy surface and is competitive with intersystem crossing to the triplet state. Flash vacuum thermolysis of the α-trifluoromethylphenylcarbene results in rearrangement to trifluorobenzocyclobutene and trifluorostyrene.
CH14129Investigation of cis- and trans-4-Fluoroprolines as Enantioselective Catalysts in a Variety of Organic Transformations
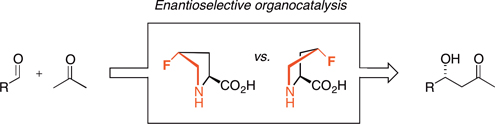
Cis- and trans-4-fluoroprolines are investigated as catalysts in five different organic transformations. Significant differences in enantioselectivity are observed between the two fluorinated catalysts, confirming that the ring pucker is important.
CH14256Cooperative Conformational Regulation in N-Heterocyclic Fluorohydrins
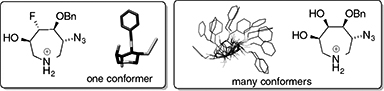
The conformation control of flexible seven-membered N-heterocycle ring structures is important for their bioactivity. Fluorine substitution in the appropriate relative configuration significantly reduces conformational disorder in highly substituted azepanes.
CH14458Fluorous 1,2,3-Triazol-4-ylmethyl Amines and Amine Derivatives for Novel Surfactant Applications
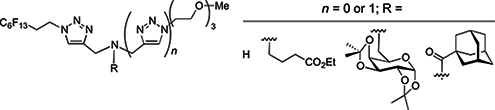
Mono- and bis(triazolylmethyl)amines and their derivatives have potential as functional surfactants. Building blocks have been prepared and strategies developed for the construction in good yields of a range of fluorous tertiary amine and amide derivatives with polyfluoroalkyl and oligoether substituents for enhanced surfactant activity.
CH14130[18F]Fluorination Optimisation and the Fully Automated Production of [18F]MEL050 Using a Microfluidic System
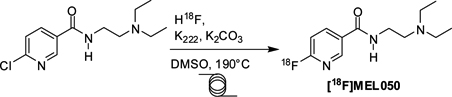
The [18F]radiolabelling optimisation of [18F]MEL050 was performed using a microfluidic system. The radiochemical production was then fully automated and performed on a larger scale, in quantities sufficient for preclinical studies.
CH14298Fluorovinyl Thioethers as Putative Steric and Electronic Thioester Enolate Mimetics: Chemoselective HF Addition to Acetylene Thioethers
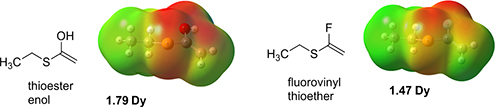
Fluorovinyl thioethers are presented as a putative biomimetic surrogate for the enol/ate of a thioester. A method is explored for the preparation of fluorovinyl thioethers by treatment of acetylene thioethers with pyridinium (poly)-hydrogen fluoride. A fluorovinyl thioether representing the terminus of the pantothenoyl chain of acetyl-CoA is prepared as a relevant biomimetic example.
CH14110Synthesis, Structures, and Properties of Four Novel HgII Complexes Based on Pyridine Acylamide Ligands
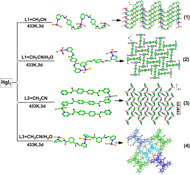
We present here four novel HgII complexes. 1 and 2 are supramolecular isomers, which have one-dimensional (1D) helical chain or 1D meso-helical chain, respectively. Complex 3 shows a novel Z-shaped zero-dimensional structure. In complex 4, flexible L3 ligands link HgI2 units to construct a 1D helical chain.
CH14033Evaluation of Antiradical Activity and Reducing Capacity of Synthesised Bispyridinium Dibromides Obtained by Quaternisation of 4-Pyridyl-1,4-dihydropyridines with Propargyl Bromide
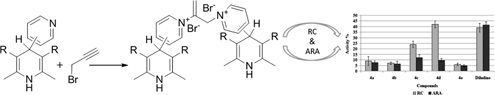
New bispyridinium dibromides on a 1,4-dihydropyridine (1,4-DHP) core were synthesised in the reaction between propargyl bromide and 4-pyridyl-1,4-DHP derivatives with variation of electronic nature of the 1,4-DHP cycle. The antiradical activity and reducing capacity of the newly synthesised compounds have been evaluated.
CH13693Synthesis, Photo-physical and Electrochemical Properties of Dendrimers with (S)-BINOL Core and Benzothiazole Surface Unit
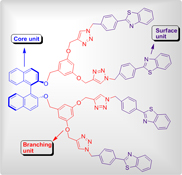
Chiral dendritic architectures with benzothiazole as surface group, 1,2,3-triazole as bridging unit, and (S)-BINOL as core unit were synthesised in good yields via a convergent synthetic strategy. All the dendrimers showed excellent optical and electrochemical response and hence would find application in dye sensitised solar cells.
CH13679Characterization and Acidic Properties of AlMCM-41 Prepared by Conventional and Post-Synthesis Alumination

The acidic properties of MCM-41 hexagonally ordered mesoporous silica were created by an isomorphous substitution of Si by a trivalent cation Al3+, generating catalysts of the AlMCM-41 type prepared by conventional and post-synthesis alumination.
CH14028Dawson-type Polyoxometalate Covalently Linked to Naphthalene: Synthesis, Characterisation and Material Properties
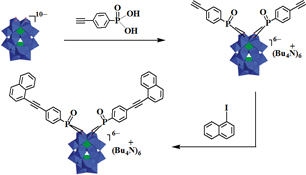
A novel covalently linked organic–polyoxometalate hybrid compound was prepared and characterised. It displayed interesting electrical conductivity behaviours, photovoltage response, and photocatalytic activity.
CH14115Three ZnII Coordination Polymers Based on 1,2-Naphthalenedicarboxylate and Different 4,4′-Bipyridyl-like Bridging Co-ligands: Structural Regulation and Properties
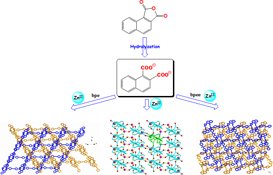
The construction of three luminescent ZnII coordination polymers has been successfully achieved based on 1,2-naphthalenedicarboxylate, and their diverse supramolecular structures can be well regulated by incorporating different auxiliary N-donor co-ligands.
CH14127Metal Ion-Driven Assembly of Two New Coordination Polymers Constructed by Asymmetric Tricarboxylate and Imidazole-Containing Ligands: Syntheses, Crystal Structures, and Luminescent Properties
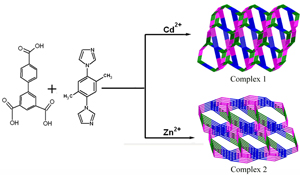
Two new coordination polymers have been obtained under hydrothermal conditions based on asymmetrical carboxylate ligands. It is shown that the asymmetrically tricarboxylate can bear diverse structures regulated by metal ions.
CH14078Functionalisation of Imidazolin-2-imine to Corresponding Phosphinamine, Chalcogenide (O, S, Se, Te), and Borane Compounds
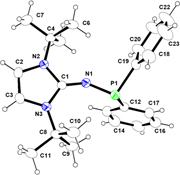
Imidazolin-2-imine was functionalised at the imine end by to synthesise imidazolin-2-ylidine-1,1-diphenylphosphinamine (2) and it was treated further with elemental sulfur, selenium, and tellurium to afford the corresponding chalcogenide derivatives.
CH14192Microwave-Assisted Synthesis of Ruthenium(II) Complexes with Trimethylsilylacetylene as Inhibitors against the Migration of Breast Cancer Cells
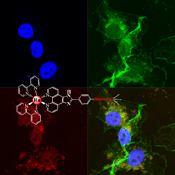
Two novel chiral ruthenium(ii) complexes with TMSA were synthesised by Songogashira coupling reaction under microwave irradiation, and can distribute and enrich in the nucleus of cancer cells and inhibit the migration of MDA-MB-231 cells.
CH14037Trichloroisocyanuric Acid/Triphenylphosphine-Mediated Synthesis of Benzimidazoles, Benzoxazoles, and Benzothiazoles
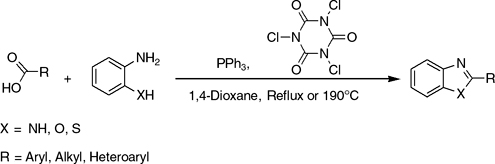
A new and efficient method for preparation of benzimidazoles, benzoxazoles, and benzothiazoles from reactions of different carboxylic acids with o-phenylenediamine, o-aminophenol, and o-aminothiophenol in the presence of triphenylphosphine/trichloroisocyanuric acid system is presented. The remarkable advantages are the inexpensive and readily available reagent, simple procedure, mild conditions, and good-to-excellent yields.
CH14079A Highly Selective and Sensitive Chemosensor for Colorimetric and Fluorescent Detection of Al3+ and Living Cell Imaging
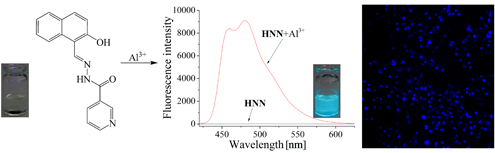
The colorimetric and fluorescent dual chemosensor, (E)-N'-((2-hydroxynaphthalen-1-yl)methylene)nicotinohydrazide (HNN), exhibits a specific response to Al3+.
CH14392A Novel (3,4,9)-Connected 3D Metal–Organic Framework Based on the Non-Planar Tricarboxyl Tecton and Zn5O4-Cluster SBU
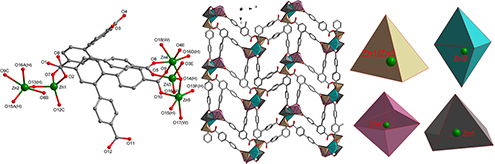
Reported here is an unusual three-dimensional ZnII (3,4,9)-connected (42.5)(3.43.52)(32.45.511.613.73.82) coordination framework constructed by a non-planar tricarboxyl tecton and Zn5O4-cluster secondary building units.
CH14412Pd-Catalyzed Dehydrogenative Cross-Coupling of 1,4-Quinones with N,N′-Dialkyluracils
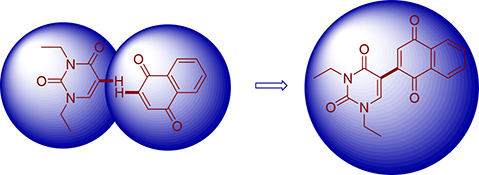
A straightforward and efficient method for the palladium-catalyzed direct cross-coupling of quinones with N,N′-dialkyluracils via 2-fold C–H activation has been developed to rapidly construct uracil substituted quinone structural motifs.
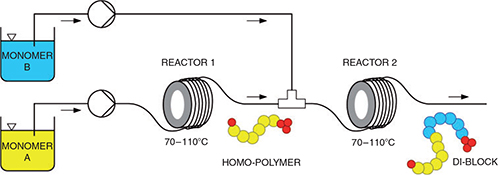
The synthesis of block copolymers using continuous flow in combination with the Reversible Addition–Fragmentation chain Transfer (RAFT) method has proven to be more efficient and less time-consuming than traditional batch processes. This paper reviews one of the first RAFT polymerizations and two of the latest articles reporting the synthesis of block copolymers using RAFT technology in continuous flow.



