Evidence of genetic subdivision among populations of blacklip abalone (Haliotis rubra Leach) in Tasmania
Nepelle Temby A , Karen Miller A B C D and Craig Mundy BA School of Zoology, University of Tasmania, Private Bag 5, Hobart, Tas. 7001, Australia.
B Marine Research Laboratory, Tasmanian Aquaculture and Fisheries Institute, University of Tasmania, Private Bag 49, Hobart, Tas. 7001, Australia.
C Present address: Institute of Antarctic and Southern Ocean Studies, University of Tasmania, Private Bag 77, Hobart, Tas. 7001, Australia.
D Corresponding author. Email: karen.miller@utas.edu.au
Marine and Freshwater Research 58(8) 733-742 https://doi.org/10.1071/MF07015
Submitted: 30 January 2007 Accepted: 13 June 2007 Published: 27 August 2007
Abstract
The scale over which populations exchange individuals (migration) is central to ecology, and important for understanding recruitment and connectivity in commercial species. Field studies indicate that blacklip abalone (Haliotis rubra) have localised larval dispersal. However, genetic studies show differentiation only at large scales, suggesting dispersal over more than 100 km. Most genetic studies, however, have failed to test for subdivision at scales equivalent to field experiments. We used microsatellite DNA to investigate genetic structure at small scales (100 m to 10 km) in blacklip abalone in south-east Tasmania. We found significant subdivision (FST = 0.021; P < 0.05) among sites, and hierarchical FST analysis indicated 64% of genetic variation was at the smallest scale, supporting field studies that concluded larval dispersal is less than 100 m. We also tested if genetic differentiation varied predictably with wave exposure, but found no evidence that differences between adjacent sites in exposed locations varied from differences between adjacent sites in sheltered populations (mean FST = 0.016 and 0.017 respectively). Our results show the usefulness of microsatellites for abalone, but also identify sampling scales as critical in understanding gene flow and dispersal of abalone larvae in an ecologically relevant framework. Importantly, our results indicate that H. rubra populations are self-recruiting, which will be important for the management of this commercial species.
Additional keywords: abalone, fisheries management, gene flow, habitat, larval dispersal, microsatellite DNA.
Acknowledgements
This research was undertaken as part of a BSc honours degree by NT under the supervision of KM and CM. The research was funded through the Tasmanian Aquaculture and Fisheries Institute (TAFI). We thank Mike Porteus, Stewart Dickson and Chris Jarvis for their help with sample collection and Adam Smolenski for assistance in the laboratory. We also thank Nick Elliott for helpful advice regarding suitable microsatellite loci to use for the present study.
Ayre, D. J. (1985). Localized adaptation of clones of the sea anemone Actinia tenebrosa. Evolution 39, 1250–1260.
| Crossref | GoogleScholarGoogle Scholar |
Bohonak, A. J. (1999). Dispersal, gene flow, and population structure. The Quarterly Review of Biology 74, 21–43.
| Crossref | GoogleScholarGoogle Scholar | PubMed |
Brown, L. D. (1991). Genetic variation and population structure in the blacklip abalone, Haliotis rubra. Australian Journal of Marine and Freshwater Research 42, 77–90.
| Crossref | GoogleScholarGoogle Scholar |
Burnett, W. J. , Benzie, J. A. H. , Beardmore, J. A. , and Ryland, J. S. (1995). Patterns of genetic subdivision in populations of a clonal cnidarian, Zoanthus coppingeri, from the Great Barrier Reef. Marine Biology 122, 665–673.
| Crossref | GoogleScholarGoogle Scholar |
Chambers, M. D. , VanBlaricom, G. R. , Hauser, L. , Utter, F. , and Friedman, C. S. (2006). Genetic structure of black abalone (Haliotis cacherodii) populations in the California islands and central Californian coast: Impacts of larval dispersal and decimation from withering syndrome. Journal of Experimental Marine Biology and Ecology 331, 173–185.
| Crossref | GoogleScholarGoogle Scholar |
Evans, B. , White, R. W. G. , and Elliott, N. G. (2000). Characterisation of microsatellite loci in the Australian blacklip abalone (Haliotis rubra, Leach). Molecular Ecology 9, 1183–1184.
| Crossref | GoogleScholarGoogle Scholar | PubMed |
Kimura, M. , and Weiss, G. H. (1964). The stepping stone model of populations structure and the decrease of genetic correlation with distance. Genetics 49, 561–576.
| PubMed |
McShane, P. E. (1995a). Recruitment variation in abalone: its importance to fisheries management. Marine and Freshwater Research 46, 555–570.
| Crossref | GoogleScholarGoogle Scholar |
Palumbi, S. R. (2003). Population genetics, demographic connectivity, and the design of marine reserves. Ecological Applications 13, 146–158.
| Crossref | GoogleScholarGoogle Scholar |
Prince, J. D. , Sellers, T. L. , Ford, W. B. , and Talbot, S. R. (1987). Experimental evidence for limited dispersal of Haliotid larvae (genus Haliotis: Mollusca: Gastropoda). Journal of Experimental Marine Biology and Ecology 106, 243–263.
| Crossref | GoogleScholarGoogle Scholar |
Shepherd, S. A. , and Brown, L. D. (1993). What is an abalone stock – Implications for the role of Refugia in conservation. Canadian Journal of Fisheries and Aquatic Sciences 50, 2001–2009.
Shepherd, S. A. , Rodda, K. R. , and Vargas, K. M. (2001). A chronicle of collapse in two abalone stocks with proposals for precautionary management. Journal of Shellfish Research 20, 843–856.
van Oosterhout, C. , Hutchinson, W. F. , Wills, D. P. M. , and Shipley, P. (2004). MICRO-CHECKER: software for identifying and correcting genotyping errors in microsatellite data. Molecular Ecology Notes 4, 535–538.
| Crossref | GoogleScholarGoogle Scholar |
Zhongbao, L. , Appleyard, S. A. , and Elliott, N. G. (2006). Population structure of Haliotis rubra from South Australia inferred from nuclear and mtDNA analyses. Acta Oceanologica Sinica 25, 99–112.

Zuniga, G. , Guzman Del Proo, S. , Cisneros, R. , and Rodriguez, G. (2000). Population genetic analysis of the abalone Haliotis fulgens (Mollusca: Gastropoda) in Baja California, Mexico. Journal of Shellfish Research 19, 853–859.

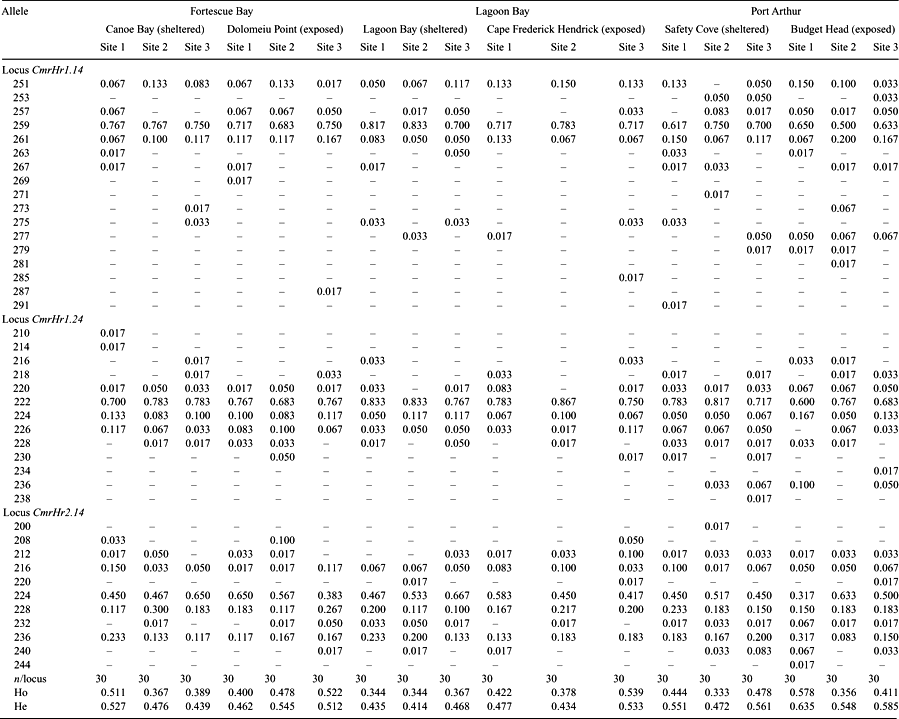
|


