
Australian Journal of Chemistry
Volume 65 Number 4 2012
CH11475 Low-cost Hollow Silica Supports for Environmental Pollution: High Removal Capacity and Low Desorption Rate of Neutral Red
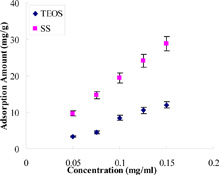
We exhibit the first example of constructing high ordered macroporous silica adsorbent by using a low-cost silica source. This silica-based macroporous material can be useful as a powerful separation tool to deal with environmental pollution because it is easy to separate simply by centrifugation without any modification. Interestingly, the absorption and separation for neutral red show that it can increase absorbent amount of dye and decrease the rate of dye desorbed in comparison to conventional hollow silica obtained via the Stöber method.
CH11463 An Approach to 2,3-Diaminosuccinic Acid Derivatives—Synthesis of 2-Thioxo-1,3-Imidazolidines by a Mannich Reaction
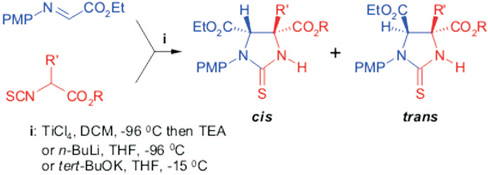
The first application of 2-isothiocyanatocarboxylic esters in Mannich reactions is described. The investigated method leads to 2-thioxo-1,3-imidazolidine derivatives—masked 2,3-diaminosuccinic acids.
CH11424 Synthesis and Structures of Halo-Substituted Aroylhydrazones with Antimicrobial Activity
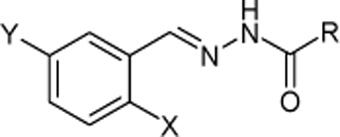
Seven new aroylhydrazones are prepared, characterized, and evaluated for their antimicrobial activities. N′-(2-Chloro-5-nitrobenzylidene)-2-fluorobenzohydrazide showed the greatest activity. The presence of electron-withdrawing groups in the aroylhydrazones can improve their activities.
CH11481Controlled Synthesis, Dye-fixing Performance and Dye-fixing Mechanisms on Cotton Fabric of Novel Reactive Cationic Copolymers of N,N-Diallyl-3-hydroxy-azetidinium Chloride and Dimethyldiallylammonium Chloride (P(DHAC-DMDAAC)s)
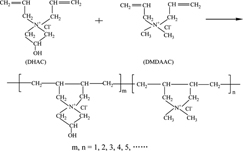
A series of novel reactive cationic copolymers (P(DHAC-DMDAAC)s) of N,N-diallyl-3-hydroxy-azetidinium chloride (DHAC) and dimethyldiallylammonium chloride (DMDAAC) with controlled structures and controlled molecular weights were first synthesized, then the dye-fixing performance and dye-fixing mechanisms on cotton fabric of the obtained P(DHAC-DMDAAC)s were further studied.
CH12035 N-Heterocyclic Carbene Adducts of Cyclopalladated Ferrocenylpyridazine: Synthesis, Structural Characterization, and Application in ?-Arylation of Ketones with Aryl Chlorides
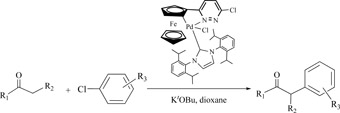
A new ferrocene-based ligand 3-chloro-6-pyridazinylferrocene 1 and its N-heterocyclic carbene adducts 2–3 were synthesized and characterized. Complex 3 exhibited high catalytic activity for α-arylation of ketones with aryl chlorides.
CH12040 Structures of 4-Iminopyrido[1,2-a]pyrimidines, Pyrido[1,2-a]pyrimidin-4-ones, Pyridopyrimidinium Olates, and Thiazolo[3,2-a]pyrimidine Analogues
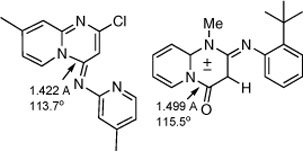
Extraordinarily long ‘amidine’ and ‘amide’ C–N bonds (up to 1.5 Å) and acute NCX angles in the title compounds are interpreted in terms of the Structure-Correlation Principle of Buergi and Dunitz. The structural features are in accord with the observed thermal ring opening to ketenimines and ketenes, respectively.
CH12041 Polymorphism in 3-Pyridylsydnone: Preparative and Structural Aspects
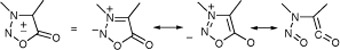
The formation and interconversion of two polymorphs of 3-(3′-pyridyl)sydnone are elucidated. A metastable P2/c form is obtained by recrystallization, and a thermodynamically more stable, P21/c polymorph, by sublimation or heating in the solid state.
CH11462 Synthesis of Cyclic Carbonate From Carbon Dioxide and Epoxide Using Amino Acid Ionic Liquid Under 1 atm Pressure
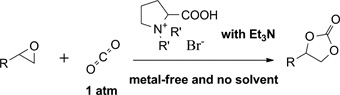
The cycloaddition of carbon dioxide into epoxide was realized by using an amino acid ionic liquid. With triethylamine as co-catalyst, the catalytic activity of a l-proline based ionic liquid was greatly enhanced, and up to 97 % isolated yield of cyclic carbonate was achieved. This method affords an alternative access to cyclic carbonate under 1 atm carbon dioxide pressure without metal or solvent.
CH11410 Aggregation-Induced Emission and Large Two-Photon Absorption Cross-Sections of Diketopyrrolopyrrole (DPP) Derivatives
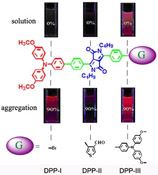
A new series of triphenylamine-based diketo-pyrrolo-pyrrole (DPP) compounds (DPP-I, DPP-II, DPP-III) have been synthesized and shown to exhibit aggregation-induced emission properties and large two-photon absorption cross-section.
CH11397Towards the Development of a Direct Electrochemical Biodetector of Avidin Based on the Poly(chloro amino-β-styryl terthiophene)-Coated Glassy Carbon Electrode
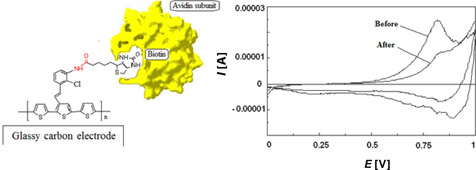
In this study, a simple and direct biodetector of avidin was proposed, using biotin immobilized onto a conducting polymer-coated electrode based on poly(chloro amino-β-styryl terthiophene). The biotin-avidin interaction was monitored by cyclic voltammetry for the detection of avidin at 4 × 10–6 mol L–1.
CH12018 The Bioreductive Alkylation of DNA by Kalafungin: A Theoretical Investigation
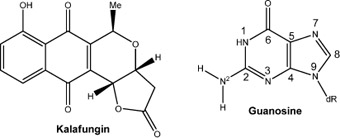
The thermochemical cascades for the bioreductive alkylation of DNA by kalafungin were calculated. According to them both one- and two-electron reduction is possible in vivo. Furthermore, a clear pathway was found for both mono- and bis-alkylations of guanine. The results presented here support the idea that this compound readily forms covalent bonds with DNA resulting in pro-mutagenic lesions.
CH12025 A New and Efficient Procedure for Friedländer Synthesis of Quinolines in Low Melting Tartaric Acid-Urea Mixtures
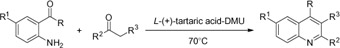
Low melting mixture of L-(+)-tartaric acid and N,N-dimethylurea has been used for the synthesis of quinoline derivatives via the Friedländer reaction of 2-aminoaryl ketones and α-methylene ketones. The melt acts as both the reaction medium and catalyst, furnishing quinolines in high to excellent yields.
CH12046The Nucleophilic Addition of β-Lactam Carbenes to Alkylphenylketenes for a Ready Approach to Spiro[β-lactam-2,1′-indene] Derivatives
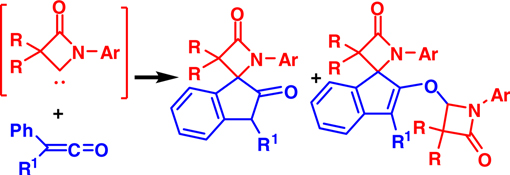
The nucleophilic reaction of ambident β-lactam carbenes with alkylphenylketenes is studied. Two types of spiro[β-lactam-2,1′-indene] derivatives are produced with total yields of 55–89 %.
CH11446Oxidative Damage of Pyrimidine Nucleosides by the Environmental Free Radical Oxidant NO3• in the Absence and Presence of NO2• and Other Radical and Non-Radical Oxidants
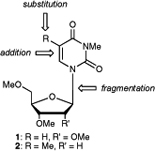
Product studies showed that the reaction of the night-time atmospheric free-radical oxidant NO3• with uridine 1 and thymidine 2 is very complex and is influenced by the presence of other free radical species, specifically NO2•. Depending on the conditions, the reaction can either lead to cleavage of the glycosidic bond, addition to the C=C double bond, or substitution of R at the nucleobase.



