
Australian Journal of Chemistry
Volume 65 Number 11 2012
RESEARCH FRONT: Do We Know All About Ionic Liquids?
CH124815th Australasian Symposium on Ionic Liquids

The 5th Australasian Symposium on Ionic Liquids was held in May 2012 in Clayton, Australia, and brought together internationally recognised researchers in a discipline that crosses borders incorporating theory, chemistry, and engineering. This research front, dedicated to ionic liquids, comprises a selection of papers representing the current look of this emerging field.
CH12324Ionic Liquids for Lignin Processing: Dissolution, Isolation, and Conversion
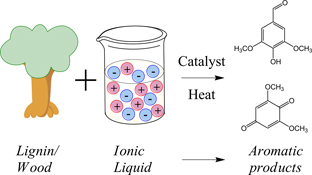
The use of ionic liquids as multifunctional solvents for lignin processing is reviewed. This includes the selective isolation of lignin from lignocellulosic materials such as wood and other biomass, and depolymerization of lignin to yield aromatic feedstock molecules for the chemical industry.
CH12344Computer-Aided Molecular Design of Ionic Liquids: An Overview
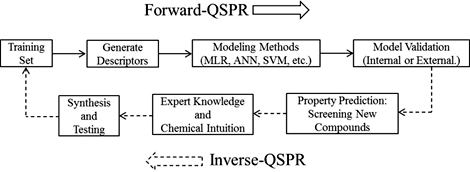
Computer-aided molecular design (CAMD) is a technique that helps select potential target molecules that will have desired properties before synthesis and testing in the laboratory, and provides an excellent complement to the chemical intuition possessed by experimentalists. In this overview, we present the general methodology for CAMD, and describe recent progress in this area related to ionic liquids.
CH12314Benchmark Values: Thermochemistry of the Ionic Liquid [C4Py][Cl]
A combination of experimental methods (DSC + TGA) with theoretical methods (quantum chemical calculations and MD simulations) provided benchmark thermochemical values for ionic liquid [C4Py][Cl].
CH12342Structure of Regenerated Celluloses Treated with Ionic Liquids and Comparison of their Enzymatic Digestibility by Purified Cellulase Components
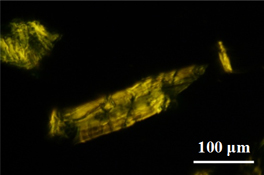
Dissolution of microcrystalline cellulose with ionic liquid and regeneration of cellulose by adding poor solvent cause the change of crystal allomorph and crystallinity of cellulose. These structural differences affect the adsorption ability of cellulase on the surface of regenerated cellulose and the enzymatic digestibility by monocomponent cellulase.
CH12307Use of Bacterial Cellulose from Nata de Coco as Base Polymer for Liquid Membranes Containing Ionic Liquids

We prepared bacterial cellulose membranes from nata de coco, an indigenous dessert of the Philippines, as a base polymer in the liquid membrane process. In the presence of Aliquat 336 (tri(C8–C10)methyl ammonium chloride) as an ionic liquid, we obtained stable bacterial cellulose membranes. Quinoline and pyridine successfully permeated through the membranes against heptane.
CH12316Role of Cation in Enhancing the Conversion of the Alzheimer’s Peptide into Amyloid Fibrils Using Protic Ionic Liquids
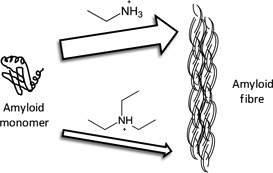
We report on the role of protic ionic liquids in enhancing the conversion of Aβ 16–22 monomers into amyloid fibrils. This is a process which is implicated in Alzheimer’s disease.
CH12343Electrodeposition of Lithium in Polystyrene Sphere Opal Structures on Copper from an Ionic Liquid

In this paper, it is shown that lithium deposition in a polystyrene sphere opal structure can lead to hollow spheres or to a regular macroporous structure, depending on the experimental conditions.
CH12322Role of H+ in Polypyrrole and Poly(3,4-ethylenedioxythiophene) Formation Using FeCl3·6H2O in the Room Temperature Ionic Liquid, C4mpyrTFSI
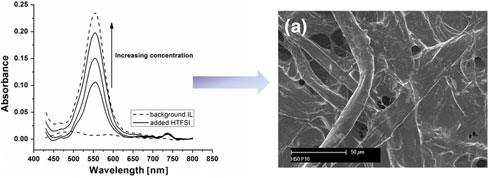
An investigation of the polymerisation reaction of pyrrole and 3,4-ethylenedioxythiophene using FeCl3·6H2O in the room temperature ionic liquid C4mpyrTFSI reveals that H+, present due to deprotonation of coordinated waters, may itself act as the chemical oxidant for the polymerisation reaction.
CH12305Influence of Additives on the Speciation, Morphology, and Nanocrystallinity of Aluminium Electrodeposition
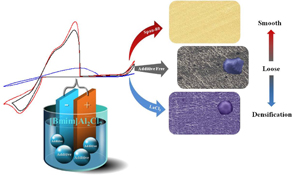
Four series of additives have significant influence on the speciation and morphology of aluminium electrodeposition. The smoothness or uniformity of deposits can be adjusted by using different additives. Moreover, some of the aluminium deposits are nanocrystalline in nature.
CH12330Aluminium Nanowire Electrodes for Lithium-Ion Batteries
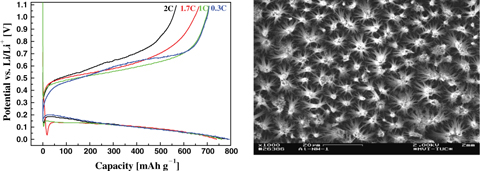
In this paper, it is shown that electrochemically made self-standing aluminium nanowires are promising as host materials for lithium in lithium-ion batteries. Lithium deposition/stripping in this material delivers an amperometric charge density close to 800 mAh g–1 with ionic liquids as electrolyte. The material does not show tremendous degradation after at least 50 CV cycles.
CH12334A Combined Scanning Electron Micrograph and Electrochemical Study of the Effect of Chemical Interaction on the Cyclability of Lithium Electrodes in an Ionic Liquid Electrolyte

The effect of storage time on the cyclability of lithium electrodes in 0.5 m LiBF4/[C3mpyr+][FSI–] was investigated. Chemical interaction results in surface passivation and impacts on the charging/discharging process. The electrode resistance decreased after cycling due to significant structural rearrangement of the surface layer.
CH12332The Influence of Water and Metal Ions on the Transport Properties of Trihexyl(tetradecyl)phosphonium Chloride
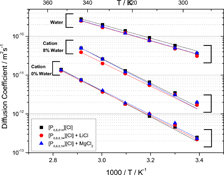
The physical chemistry of an ionic liquid electrolyte with the addition of water and different metal salts is explored with numerous techniques including NMR. The conductivity of the electrolytes increases an order of magnitude when saturated with water. The metal salts interestingly also increase conductivity at low water concentrations.
CH12282Selective Transport of Water-Soluble Proteins from Aqueous to Ionic Liquid Phase via a Temperature-Sensitive Phase Change of These Mixtures

Complete separation of some water-soluble proteins was facilitated in a moment by the salting-in of the hydrated ionic liquid phase.
CH12374Surface Composition of Mixtures of Ethylammonium Nitrate, Ethanolammonium Nitrate, and Water
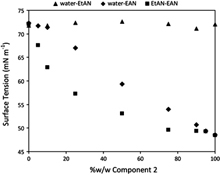
Surface tensiometry of binary mixtures of ethylammonium nitrate, ethanolammonium nitrate, and water reveals amphiphilic character for the ethylammonium cation, but not for ethanolammonium. The surface film incorporates nitrate counterions, and electrostatic and H-bonding interaction, rather than alkyl chain packing, determine the saturated adsorbed film structure.
CH12331Preparation of Magnetic Ionic Liquids Composed of Hybrid-Type Anions
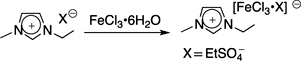
1-Ethyl-3-methylimidazolium ethylsulfate ([C2mim][EtSO4]) was mixed with iron(III) chloride•6H2O to give a magnetic ionic liquid composed of the hybrid-type anion, [C2mim][FeCl3•EtSO4], which showed a slightly different magnetic property than [C2mim][FeCl4]. On the other hand, reaction of [C2mim][EtSO4] with cobalt(II) chloride hydrate formed [C2mim]2[CoIICl4], the structure of which was characterised by X-ray crystallographic analysis.


