
Australian Journal of Chemistry
Volume 67 Number 3 2014
Heron 6
CH140416th Heron Island Conference on Reactive Intermediates and Unusual Molecules

The 6th Heron Island Conference on Reactive Intermediates and Unusual Molecules was held at the Heron Island Resort on the Great Barrier Reef from 7 to 13 July 2013. This special issue of the Journal presents a collection of papers authored by delegates at the meeting. The cover art for this issue was created by Svetlana Tšupova in the group of A. Stephen K. Hashmi (University of Heidelberg).
CH13423Intermediates in the Formation and Thermolysis of Peroxides from Oxidations with Singlet Oxygen
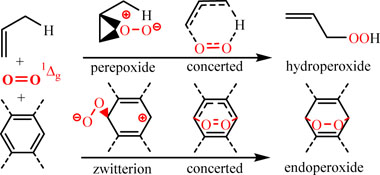
Concerted or stepwise? This is the question. Experimental and theoretical work give evidence that the ene reaction of singlet oxygen with alkenes proceeds via intermediates structurally similar to hydroperoxides. On the other hand, photooxygenations of smaller acenes are concerted, and zwitterions are observed for higher acenes affording endoperoxides. Finally, the thermolysis of endoperoxides proceeds mainly via radical intermediates.
CH13533Azides – Diazonium Ions – Triazenes: Versatile Nitrogen-rich Functional Groups
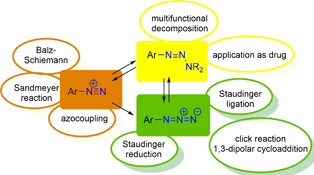
Within this highlight article, we focus on the diverse chemistry of the azide, the diazonium, and the triazene moiety. In particular, we present our latest applications of this highly interesting class of nitrogen-rich functional groups.
CH13591From 'Lab & Light on a Chip' to Parallel Microflow Photochemistry
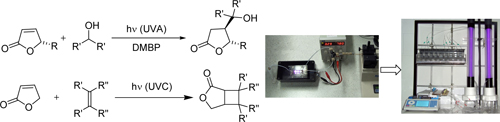
Continuous-low microreactors offer major advantages for photochemical applications. This mini-review summarizes the technological development of microflow devices in the Applied and Green Photochemistry Group at James Cook University, and its associates, from fixed microchips for microscale synthesis to flexible multicapillary systems for parallel photochemistry.
CH13307Natural Product Synthesis via the Rhodium Carbenoid-Mediated Cyclization of α-Diazo Carbonyl Compounds
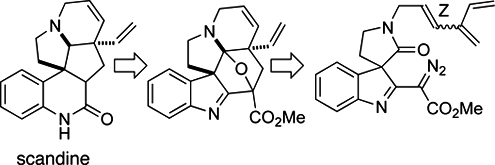
The creation of carbonyl ylides from the reaction of diazo compounds with ketones in the presence of RhII catalysts has significantly broadened their applicability for natural product synthesis. The ease of generating the dipole and the predictability of its regiochemistry have contributed to the popularity of this synthetic method, which is described in this mini-review.
CH13436Thermal and Photocatalytic Production of Hydrogen Peroxide and its Use in Hydrogen Peroxide Fuel Cells
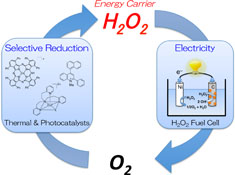
Our recent developments on the thermal and photocatalytic production of hydrogen peroxide and its use in hydrogen peroxide fuel cells have been reviewed. These developments are beneficial to make hydrogen peroxide an energy carrier for the next generation.
CH13576Recent Developments in the Synthetic Uses of Silyl-protected Enoldiazoacetates for Heterocyclic Syntheses
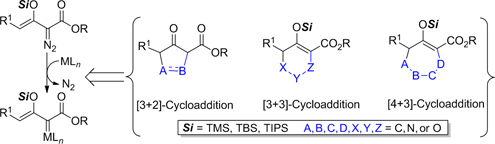
Annulations of metal enolcarbenes by cycloaddition reactions with unsaturated systems, 1,3-dipoles, and dienes provide convenient access to five-, six-, and seven-membered-ring heterocyclic compounds, including furans, pyrroles, lactones, tropanes, oxazines, tetrahydropyridazines, pyranones, oxepins, and bicyclic analogues.
CH13440An Enantiomerically Pure Pyridine NC-Palladacycle Derived from [2.2]Paracyclophane
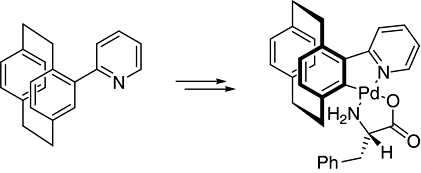
An enantiomerically pure non-metallocenic planar chiral palladacycle can be synthesised in four steps from readily available [2.2]paracyclophane. The structure of the new palladacycles are discussed and their potential in both coupling reactions and the functionalisation of [2.2]paracyclophane is demonstrated.
CH13444Benzonitrile Oxide Cycloadditions with Exocyclic Methylene Benzothiazepine Dioxides
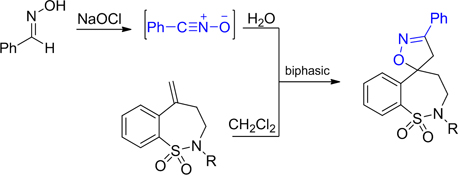
N-substituted methylene-benzothiazepines underwent 1,3-dipolar cycloaddition with benzonitrile oxide to give 5-spiro isoxazoline adducts with complete regioselectivity and high facial selectivity.
CH13451Layered γ-Zirconium Phosphate Intercalated with Ru(bpy)3-Viologen Dyads as Unusual Materials for Dye-Sensitised Solar Cells: Improving Efficiency by Double Sensitisation
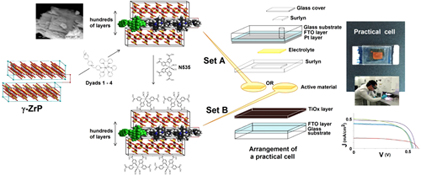
The intercalation materials prepared from the laminar salt γ-zirconium phosphate and various dyads formed from Ru(bpy)3 and viologens were tested as the photoactive components in dye-sensitised solar cells. The efficiencies significantly increased, up to 0.2 %, when the materials were treated with the panchromatic N535 dye.
CH13454Redox Properties of Iron Complexes with Pentadentate Bispidine Ligands
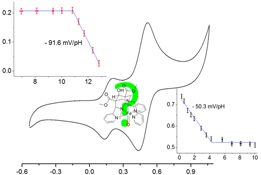
Cyclic voltammetry of iron-bispidine complexes in water and acetonitrile reveals that all observed redox potentials are attributable to the FeIII/II couple. Varying H2O/MeCN ratios, anions, and pH leads to differences attributable to variations of the co-ligand (NCMe, Cl–, OH2, OH–). Solutions of the oxygenated ferrous precursors also gave the FeIII/II couple of the spontaneously reduced ferryl complex.
CH13460The Zwitterionic Structure of the Parent Amidinium Tetrazolide and a Rare Tetrazole Ring-Opening Reaction
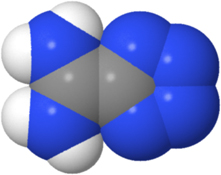
Crystals of pure 5-amidinium tetrazolide enabled the unambiguous assignment of the structure as a zwitterion rather than the 5-amidinotetrazole postulated previously. 3-Amino-4-azido-1,2,5-thiadiazole (2) is formed from the reaction of amidinium tetrazolide with thionyl chloride.
CH13464Selective Cleavage of Carbamate Protecting Groups from Aziridines with Otera's Catalyst
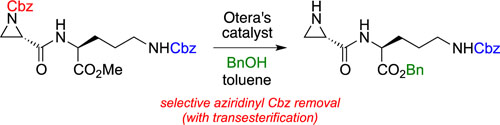
Carbamate protecting groups on azidirinyl nitrogens are selectively cleaved in the presence of other carbamates upon treatment with Otera’s catalyst. Chemoselective cleavage of one Cbz-protecting group in the presence of another Cbz group is demonstrated.
CH13467Electronic Spectroscopy of a C7H4+ Isomer in a Neon Matrix: Methyltriacetylene Cation

The electronic absorption and fluorescence spectra of methyltriacetylene cation were generated and investigated.
CH13468A New Synthesis of 4,5,6,7-Tetrahydropyrazolo[1,5-c]pyrimidines by a Retro-Mannich Cascade Rearrangement
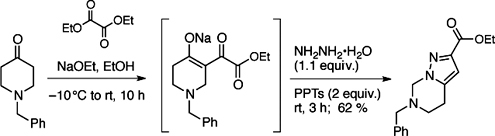
A retro-Mannich cascade reaction of in situ prepared pyrazolopyridines results in novel pyrazolopyrimidines.
CH13479Vibrational Properties of the Isotopomers of the Water Dimer Derived from Experiment and Computations
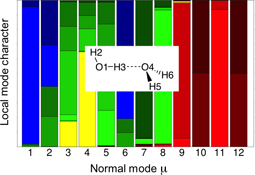
Starting from the measured vibrational frequencies of the water dimer, we derive exact frequencies for its 11 deuterium isotopomers using a perturbational approach based on coupled cluster calculations. A decomposition of the 12 normal modes of the water dimer into local modes provides a detailed description of the changes from H-to D-bonding between the water monomers.
CH13480Description of Bond Pseudorotation, Bond Pseudolibration, and Ring Pseudoinversion Processes Caused by the Pseudo-Jahn–Teller Effect: Fluoro Derivatives of the Cyclopropane Radical Cation
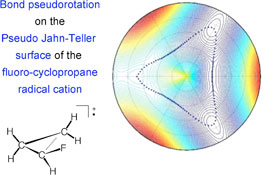
Curvilinear coordinates are used to describe the pseudo-Jahn–Teller surface of F-substituted cyclopropane radical cations using an equation-of-motion coupled cluster approach. The monofluoro derivative undergoes bond pseudolibration at low temperature, only hindered by a barrier of 2.2 kcal mol–1. Bond pseudorotation and ring pseudoinversion have barriers of 12.1 and 16.5 kcal mol–1, respectively.
CH13530Structure, Stability, and Cycloaddition Reactions of Nitrile Selenides
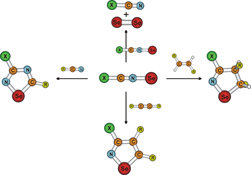
Nitrile selenides are expected to be useful 1,3-dipolarophiles for cycloaddition reactions; however, their chemistry is unexplored to date. To examine their application, equilibrium structure, unimolecular stability, and bimolecular reactions of nitrile selenides, and their cycloaddition reactions with ethynes, ethenes, and nitriles were investigated using quantum-chemical methods.
CH13531Role of Polarization in Halogen Bonds
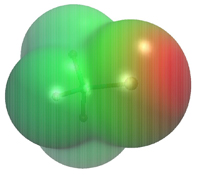
The changes in the molecular electrostatic potential at the isodensity surface of CF3Cl induced by complexation with a classical point-charge model for a halogen-bonded water molecule. The colour code ranges from –5 (blue) to +5 (red) kcal mol–1.
CH13536Sydnone Photochemistry: Direct Observation of Earl's Bicyclic Lactone Valence Isomers (Oxadiazabicyclo[2.1.0]pentanones), Formation of Carbodiimides, Reaction Mechanism, and Photochromism
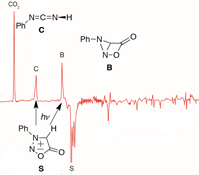
The bicyclic lactone valence isomers of sydnones, the oxadiazabicyclo[2.1.0]pentanones (B), are generated by photolysis of sydnones (S) in Ar matrices and characterized by matrix-isolation infrared spectroscopy. These bicyclic valence isomers undergo further photolysis to carbodiimides (C) and CO2. The mechanisms of formation of the lactones, carbodiimides, nitrile imines, and photochromism are discussed.
CH13546Simple Synthesis of New Mixed Isocyanide-NHC-Platinum(II) Complexes and Their Catalytic Activity
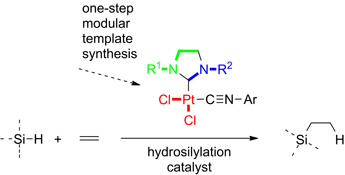
A modular template synthesis allows the easy synthesis of platinum(ii)-NHC-isocyanide complexes. These complexes show catalytic activity in the hydrosilylation of alkenes and alkynes.
CH13548Synthesis of Fluorinated Aromatic Compounds by One-Pot Benzyne Generation and Nucleophilic Fluorination
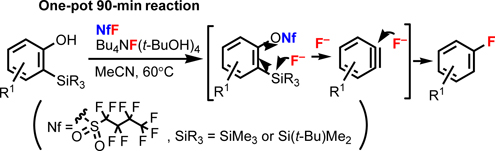
A great number of pharmaceutical products containing fluorine atoms on their aromatic rings are in the market, and the fluorine atom plays an important role in their biological activities and stabilities; however, traditional synthetic methods for aryl fluorides require harsh reaction conditions. In this paper, the one-pot three-step nucleophilic fluorination of benzynes under mild conditions is reported.
CH13552Gold Catalysis: Biarylphosphine Ligands as Key for the Synthesis of Dihydroisocoumarins
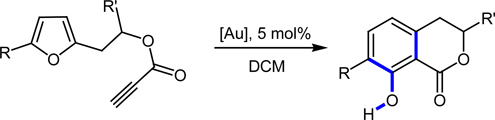
Buchwald-type biaryl phosphane ligands are needed to synthesize dihydroisocoumarins by the furan–yne reaction.
CH13562Gold Catalysis: AuCl-induced Polymerization of Styrene and n-Butylvinylether
Gold(i) catalysts deliver a monomodal distribution in the polymerization of the tested olefins, even if a second portion of olefin is added after the first portion is consumed. This is accompanied by an increase of the molecular weight, indicating a type of living polymerization probably based on gold(i)–alkene complexes as the resting state.
CH13557Formation and HERON Reactivity of Cyclic N,N-Dialkoxyamides
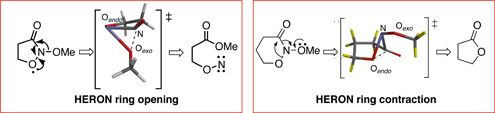
Cyclic N,N-dialkoxyamides, a new class of lactams, have been synthesised and undergo HERON reactions under stereoelectronic control. Theoretical properties of the lactams as well as their HERON reactivity have been computed.
CH13198B-Methyl Amine Borane Derivatives: Synthesis, Characterization, and Hydrogen Release
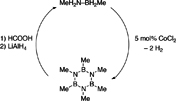
Ammonia borane (AB, BH3-NH3) and N-alkyl derivatives of AB are widely studied as potential hydrogen storage materials. In comparison, B-alkyl AB derivatives have received far less attention. Here we describe the synthesis of MeH2N–BH2Me (3) and H3N–BH2Me (4) (with 6.8 wt-% and 8.9 wt-% hydrogen capacity, respectively) and demonstrate hydrogen release as well as spent-fuel regeneration.
CH13449Reactions of Adamantanethione with Ketenes: Formation of a 1,3,5-Dioxathiane
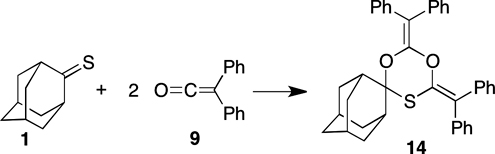
The reaction between adamantanethione 1 and diphenylketene 9 yields 2,4-bis(diphenylmethylidene)-1,3,5-dioxathiane 14 via ketene–thione zwitterions. This is the first example of the formation of a 1,3,5-dioxathiane in a ketene reaction.
CH13459Boric Acid Catalyzed Methyl Esterification of Sugar Acids
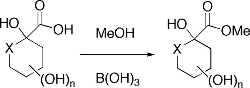
Boric acid catalyzes clean methyl esterification of some, but not all, sugar acids. However, closely related sugar acids failed to esterify under the same conditions. Factors governing this dichotomy are discussed.
CH13544CuAAC and RuAAC with Alkyne-functionalised Dihydroazulene Photoswitches and Determination of Hammett σ-Constants for Triazoles
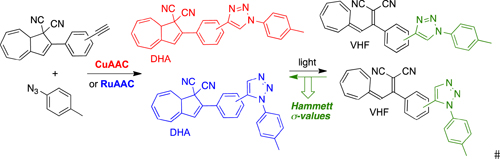
Triazole-functionalised derivatives of the dihydroazulene photoswitch were obtained by copper and ruthenium catalysed azide–alkyne cycloadditions (CuAAC and RuAAC). By comparing the rates of VHF to DHA conversions with those of previously investigated donor- and acceptor-functionalised VHFs, the electron-withdrawing character of the triazole ring was quantified by Hammett constants.



