
Australian Journal of Chemistry
Volume 68 Number 2 2015
RESEARCH FRONT: Dedication to Des Brown

This foreword commemorates the life and science of Dr Desmond J. Brown, formerly of the John Curtin School of Medical Research, ANU. It summarises his scientific career, his character as a person, and his rapport with his students, colleagues, collaborators, and friends. His published works are briefly mentioned with their pertinent references. A short scientific summary of the invited contributions and of the relationships of the senior authors with Desmond Brown are provided.
CH14482The Multiple Properties of Gliotoxin and Other Epipolythiodioxopiperazine Metabolites
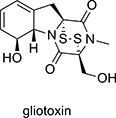
The bridged disulfide ring of the fungal metabolite gliotoxin presents both synthetic challenges and confers the molecule with a variety of interesting biological activities. This review summarises recent synthetic strategies used to insert the disulfide and polysulfide bridge across the diketopiperazine ring and describes the limited structure–activity data available for this class of molecule.
CH14349Reactions of Methyl Ketones and (Hetero)arylcarboxamides with N,N-Dimethylacetamide Dimethyl Acetal. A Simple Metal-Free Synthesis of 2,4,6-Trisubstituted Pyridines
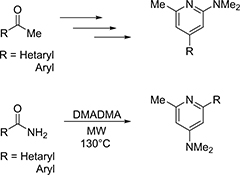
Two metal-free syntheses of 2,4,6-trisubstituted pyridines prepared either from aryl or heteroaryl methyl ketones and N,N-dimethylacetamide dimethyl acetal (DMADMA) via enaminones, or from the corresponding carboxamides by treatment with an excess of DMADMA, are described.
CH14401Acridine Synthesis by the Reaction of 2-(Perfluoroalkyl)aniline with ortho-Alkyl-Substituted Aromatic Grignard Reagent: Mechanistic Studies
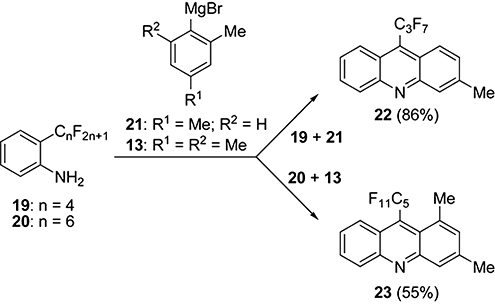
The reaction of 2-(perfluoroethyl)aniline and its higher perfluoroalkyl analogues with an arylmagnesium bromide substituted with a methyl or ethyl group at the ortho position furnishes an acridine containing a shorter perfluoroalkyl group at the 9-position and devoid of the methyl or ethyl group of the Grignard reagent. Yields are in the range of 46–93 %.
CH14425Synthesis of 6-Azapurines by Transformation of Toxoflavins and Reumycins (7-Azapteridines) and their Cytotoxicities
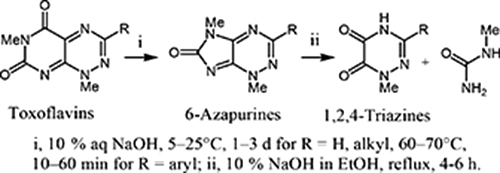
Novel 6-azapurines have been synthesised via a benzilic acid type rearrangement by treatment of toxoflavins and reumycins with 10 % aqueous or ethanolic NaOH. Further heating 6-azapurines in 10 % ethanolic NaOH afforded the corresponding 1-methyl-5,6-dioxo-1,4,5,6-tetrahydro-1,2,4-triazines with 1-methylurea. The antitumour activities of 6-azapurines against CCRF-HSB-2 and KB cell lines were also investigated.
CH14421Pteridines Part CXXI: Protection of (6R)-5,6,7,8-Tetrahydro-L-biopterin
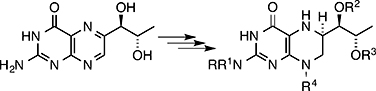
Procedures to protect the phtheridine (6R)-5,6,7,8-tetrahydo-l-biopterin have allowed the synthesis of derivatives with substituents in various positions.
CH14427In Vitro Synergy Between Some Cationic Amphipathic Cyclooctapeptides and Antibiotics
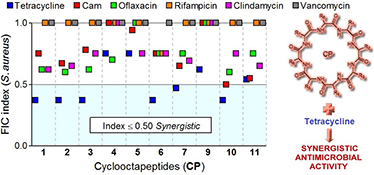
The antimicrobial activities of some cationic amphipathic cyclooctapeptides in combination with select common antibiotics are evaluated. These cyclopeptides exhibited fast killing bactericidal activities. They showed enhanced antimicrobial activities in combination with tetracycline against S. aureus; a greater than 4-fold decrease in the minimum inhibition concentration was observed for some of these cyclopeptides.
CH14464Synthesis and Tautomerism of Curcumin Derivatives and Related Compounds
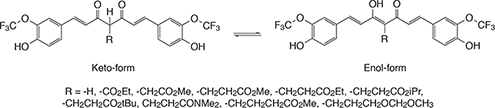
Curcumin and its derivatives have recently received attention for diagnostic and therapeutic uses for Alzheimer’s disease. And these compounds were found to bind to the Aβ aggregates in their enol form. We prepared curcumin derivatives with trifluoromethoxy groups, replacing the methoxy groups, and determined their keto/enol ratio in DMSO [D6].
CH14430A Combined Computational–Experimental Study of the Kinetics of Intramolecular Diels–Alder Reactions in a Series of 1,3,8-Nonatrienes
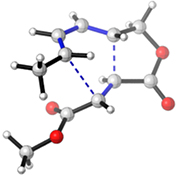
Experimental activation enthalpies for a series of intramolecular Diels–Alder reactions are used to benchmark computed values and recommendations are made. Kinetic isotope effects measurements confirm the computational predictions that the process is concerted but asynchronous.
CH14520A Chemoenzymatic Route to the (+)-Form of the Amaryllidaceae Alkaloid Narseronine
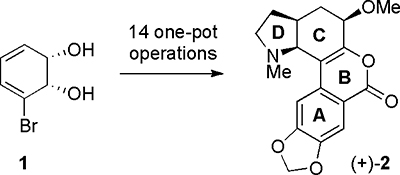
The enantiomerically pure metabolite 1, obtained through the whole-cell biotransformation of bromobenzene, has been converted into the (+)-form of the alkaloid narseronine [(+)-2]. This work complements that reported earlier in which the (–)-form of the alkaloid was produced, over a similar number of steps, from the same starting material.
CH14476A Simple Conversion of Creatinine to Creatol via Creatinine Chloroamine
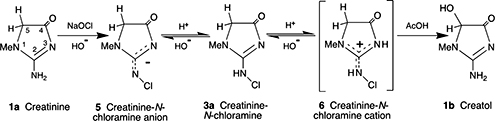
Creatinine (2-amino-1,5-dihydro-l-methylimidazol-4-one) (1a) was converted in a simple way to creatol (2-amino-1,5-dihydro-5-hydroxy-l-methylimidazol-4-one) (1b) via creatinine chloramine (2-N-chloroamino-1,5-dihydro-l-methylimidazol-4-one) (3a), without using protecting groups. The creatinine chloramine exists in the 2-N-chloroamino form (3a) in aqueous solution, but in the solid state X-ray crystal analysis shows that it has the 2-N-chloroimino structure (2-N-chloroimino-1,5-dihydro-1-methylimidazol-4-one) (4a).
CH14416Bromination of Pyrimidines: A Simple Inexpensive Method
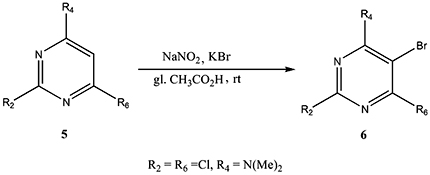
The introduction of halogens into the pyrimidine ring usually involves either specialised reagents or very aggressive conditions. This communication paper describes the introduction of bromine into position 5 of the pyrimidine ring using common inorganic salts at room temperature. An evaluation of the substituents required for successful reaction is provided.
CH14477Synthesis and Spectroscopic Properties of a Novel Bifunctional Pyrene Derivative and its Incorporation into OligoDNA
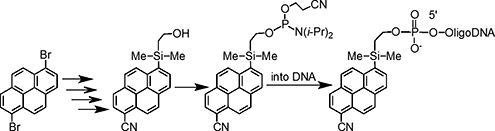
A novel bi-functional pyrene derivative bearing both a modifiable silyl function (electron-donating group) and cyano function (electron-withdrawing group) was synthesized as a new labelling agent for biological substances. The fluorescence quantum yield of the modified pyrene was significantly increased. The modified pyrene was also successfully incorporated into oligoDNA.
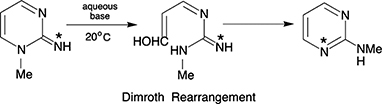
This article focuses on the involvement of Desmond Joseph Brown in the establishment of the Department of Medical Chemistry, The John Curtin School of Medical Research, ANU, and later his leadership of the Medical Chemistry Group. It also mentions his major contributions to the literature and research in heterocyclic chemistry, including the Dimroth Rearrangement, and in the training of students.
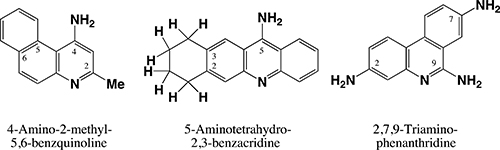
This focus paper describes the reminiscences of Heinz Duewell with Des Brown in the laboratory, and the bush environs, in their formative years at the University of Sydney. The enduring friendship with Des and his wife Jan over the years was strengthened by their bond of common interest in natural history.
CH14126Oxyhalogen–Sulfur Chemistry: Kinetics and Mechanism of Oxidation of Tiopronin by Acidified Bromate and Aqueous Bromine
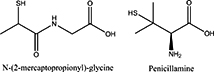
Biologically active compounds containing a thiol group are oxidatively bioactivated in physiological environment. This study involves the putative mechanism of oxidative bioactivation of tiopronin. Tiopronin, N-2-(mercaptopropionyl)glycine (MPG), is a glycine derivative with a thiol group that has properties similar to those of penicillamine, one of the flagship antibiotics.
CH14113Synthesis and Photoreaction of 2-Amino-3-cyano-4-aryl-4H-pyrans
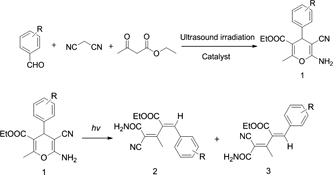
A series of 2-amino-3-cyano-4-aryl-4H-pyrans were synthesised by reacting malononitrile, ethyl acetoacetate and aromatic aldehydes under ultrasound irradiation. The photoreaction of 2-amino-3-cyano-4-aryl-4H-pyrans were tested in conventional solvents to generate the 1,3-butadienes under irradiation with UV light. The 1,3-butadiene structures were determined by 1H and 13C NMR spectra, high-resolution mass spectrometry, and single crystal X-ray diffraction analysis.
CH13734Enzymatic Synthesis of p-Cresol Oligomers and Evaluation of their Free Radical Scavenging Activity
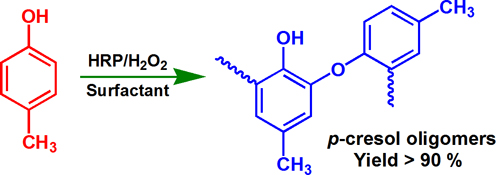
An environmentally benign aqueous micelle system was successfully used as reaction medium to perform enzymatic polymerization of p-cresol. The structure of the resulting p-cresol oligomers is similar to that of polyphenolic compounds in natural plants. p-Cresol oligomers show good free radical scavenging activity and good prospects as novel antioxidant.
CH14123Synergistic Antibacterial Activity of Nanohybrid Materials ZnO–Ag and ZnO–Au: Synthesis, Characterization, and Comparative Analysis of Undoped and Doped ZnO Nanoparticles
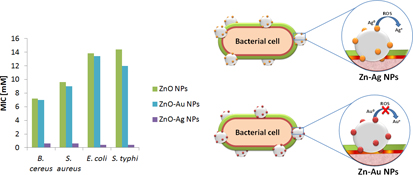
Because ZnO and Ag nanoparticles (NPs) effectively inhibit foodborne pathogens, both metals are expected to produce a more effective inhibitor as a result of hybrid properties. It has been reported that Ag-doped ZnO NPs and ZnO NPs do not show much difference in antibacterial activities, but show an increase in bacterial inhibition. To explain why doping of Ag NPs, unlike Au NPs, in ZnO NPs influences the antibacterial activities, ZnO, ZnO–Ag, and ZnO–Au NPs were synthesized.
CH14088Aerosol-Assisted Chemical Vapour Deposition for Iron Selenide Thin Films from Single Source Ferrocene-Incorporated Selenourea Precursor in the Presence of Surfactants

This article presents the synthesis and characterisation of a single source molecular precursor 1-(2-fluorolbenzoyl)-3-(3-ferrocenylphenyl)selenourea. This precursor has been used for the fabrication of FeSe thin films by aerosol-assisted chemical vapour deposition in the presence of different concentrations of two different surfactants (triton and span).
CH14153Synthesis, Structure, and Visible Phosphorescence Emission of Novel CuI Coordination Polymers Based on 4,4′-Bis(3-pyridyl)-2,2′-bis(hydroxylmethyl) Biphenyl
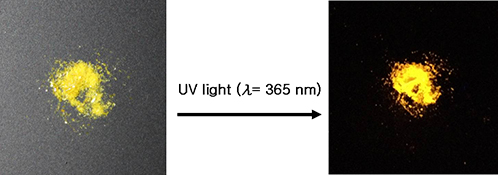
New CuI coordination polymers emit strong orange and yellow–orange phosphorescence.
CH14016MnII-Doped ZnS Quantum Dots Modified with Tiopronin for Mercury(II) Detection
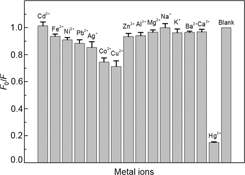
A simple, low-cost, and rapid fluorescent detection of aqueous mercuric ions based on MnII-doped ZnS quantum dots modified with tiopronin is described. The detection limit of the introduced assay (8.9 nM) is lower than the concentration of 30 nM recommended by the World Health Organization (WHO) for inorganic mercury in drinking water.
CH14092Hydrothermal Syntheses, Crystal Structures, and Luminescent Properties of Two MnII Coordination Polymers: From Two-Fold Interpenetrated pcu Topological Net to Polycatenated 2D+2D→3D Framework
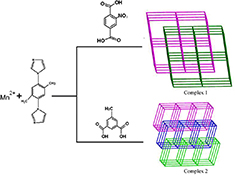
Two new coordination polymers were successfully constructed using 1,4-bis(1-imidazol-yl)-2,5-dimethyl benzene with two dicarboxylate ligands. Complex 1 exhibits a 2-fold interpenetrated pcu net. Complex 2 shows an unusual polycatenated 2D+2D→3D framework.
CH14156A Facile Synthesis of Tris(indolyl)propanes by Concerted 1,2- and 1,4-Friedel-Crafts Attack of Indoles to α,β-Unsaturated Aldimines
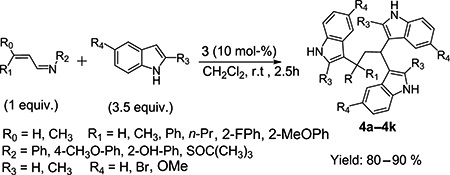
The Friedel-Crafts reaction of indoles with a wide range of α,β-unsaturated aldimines catalysed by binaphthyl phosphoric acid is demonstrated. The resulting tris(indolyl)propanes are presumably generated by a concerted 1,2- and 1,4-nucleophilic attack to the conjugated imines. According to the preliminary bioassay results, the compound 4e exhibits moderate activity against pancreatic cancer cells.
CH14254A Novel Approach to the Functionalisation of Pristine Carbon Fibre Using Azomethine 1,3-Dipolar Cycloaddition
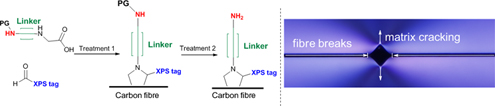
The use of reactive azomethine ylides, generated in situ, as a methodology to graft organic molecules onto the surface of unsized carbon fibre is presented. It was found that compounds bearing a pendant amine demonstrated enhanced fibre-to-matrix adhesion using the single-fibre fragmentation test in addition to samples showing transverse cracking within the epoxy matrix.
CH14343Analysis of the Enol–Keto Tautomers of Indole-3-pyruvic Acid

The keto and enol forms of indole-3-pyruvic acid (IPyA), an important intermediate in plant hormone biosynthesis, are separable by ultra high-performance liquid chromatography and have been characterized by NMR spectroscopy.



