A unique temperate rocky coastal hydrothermal vent system (Whakaari–White Island, Bay of Plenty, New Zealand): constraints for ocean acidification studies
R. Zitoun A H M , S. D. Connell B , C. E. Cornwall C D , K. I. Currie H , K. Fabricius E , L. J. Hoffmann F , M. D. Lamare G , J. Murdoch A H , S. Noonan E , S. G. Sander A H I , M. A. Sewell J , N. T. Shears K , C. M. G. van den Berg L and A. M. Smith G
C D , K. I. Currie H , K. Fabricius E , L. J. Hoffmann F , M. D. Lamare G , J. Murdoch A H , S. Noonan E , S. G. Sander A H I , M. A. Sewell J , N. T. Shears K , C. M. G. van den Berg L and A. M. Smith G
A Department of Chemistry, University of Otago, PO Box 56, Dunedin, 9054, New Zealand.
B Southern Seas Ecology Laboratories, School of Biological Sciences, University of Adelaide, Adelaide, SA 5005, Australia.
C School of Biological Sciences, Victoria University of Wellington, PO Box 600, Wellington, 6140, New Zealand.
D School of Earth Sciences and Oceans Institute, ARC Centre of Excellence for Coral Reef Studies, University of Western Australia, Crawley, WA 6009, Australia.
E Australian Institute for Marine Science, PMB 3, Townsville MC, Qld 4810, Australia.
F Department of Botany, University of Otago, PO Box 54, Dunedin, 9054, New Zealand.
G Department of Marine Science, University of Otago, PO Box 56, Dunedin, 9054, New Zealand.
H National Institute for Water and Atmospheric (NIWA)/University of Otago Research Centre for Oceanography, PO Box 56, Dunedin, 9054, New Zealand.
I International Atomic Energy Agency (IAEA) Environment Laboratories, Department of Nuclear Sciences and Applications, B.P. 800, 98000 Monaco, Principality of Monaco.
J School of Biological Sciences, University of Auckland, Private Bag 92019, Auckland, 1010, New Zealand.
K Leigh Marine Laboratory, Institute of Marine Science, University of Auckland, 160 Goat Island Road, Auckland, 0985, New Zealand
L School of Environmental Sciences, University of Liverpool, 74 Bedford Street, Liverpool, L69 7ZT, UK.
M Corresponding author. Email: rebecca.zitoun@outlook.com
Marine and Freshwater Research 71(3) 321-344 https://doi.org/10.1071/MF19167
Submitted: 13 May 2019 Accepted: 2 September 2019 Published: 18 November 2019
Journal Compilation © CSIRO 2020 Open Access CC BY-NC-ND
Abstract
In situ effects of ocean acidification are increasingly studied at submarine CO2 vents. Here we present a preliminary investigation into the water chemistry and biology of cool temperate CO2 vents near Whakaari–White Island, New Zealand. Water samples were collected inside three vent shafts, within vents at a distance of 2 m from the shaft and at control sites. Vent samples contained both seawater pH on the total scale (pHT) and carbonate saturation states that were severely reduced, creating conditions as predicted for beyond the year 2100. Vent samples showed lower salinities, higher temperatures and greater nutrient concentrations. Sulfide levels were elevated and mercury levels were at concentrations considered toxic at all vent and control sites, but stable organic and inorganic ligands were present, as deduced from Cu speciation data, potentially mediating harmful effects on local organisms. The biological investigations focused on phytoplankton, zooplankton and macroalgae. Interestingly, we found lower abundances but higher diversity of phytoplankton and zooplankton at sites in the direct vicinity of Whakaari. Follow-up studies will need a combination of methods and approaches to attribute observations to specific drivers. The Whakaari vents represent a unique ecosystem with considerable biogeochemical complexity, which, like many other vent systems globally, require care in their use as a model of ‘future oceans’.
Additional keywords: carbon dioxide, trace metal speciation, volcanic submarine vent.
Introduction
Three methods have generally been used to estimate the effects of changing seawater chemistry: mathematical modelling anchored to observational data, controlled experimentation in the laboratory or field-based mesocosm studies, and the study of natural systems representing analogues of past, present or future climate change scenarios (e.g. Boyd et al. 2018). Each of these approaches has its strengths and weaknesses, but together they have proved useful in developing preliminary projections, elucidating connections and understanding synergies (Le Treut et al. 2007) regarding ocean warming and ocean acidification (OA; Wisshak et al. 2013; Riebesell and Gattuso 2015; Gattuso et al. 2015).
Most of our current understanding of how marine biota will respond to climate change originates from controlled, laboratory-based, short-term experiments conducted in tanks and mesocosms as well as modelling approaches based on these experimental data (Wernberg et al. 2012). Controlled, laboratory-based manipulations (e.g. mesocosms) are useful in establishing a mechanistic understanding of how physical climate change is likely to drive ecological changes, but they tend to exaggerate their measured effects and obscure key processes (Goldenberg et al. 2018) because they are based on simplified artificial model systems ruled by first-order abiotic and biotic effects and thus suffer from a lack of realism (Wernberg et al. 2012; González-Delgado and Hernández 2018). To increase realism and collective inference, as well as to strengthen the generality and accuracy of our understanding of climate change as a driver of chemical and biological change in marine ecosystems, researchers have started using existing natural gradients in climate variables to examine how increased pCO2 (i.e. a change in ocean chemistry) will translate into ecological changes (e.g. Hall-Spencer et al. 2008; Wernberg et al. 2012; Teixidó et al. 2018). One example of an environment with natural abiotic gradients is the shallow submarine CO2 vent, and these vents are found around the globe (Tarasov et al. 2005; González-Delgado and Hernández 2018). Over the past 10 years, shallow CO2 vents have emerged as analogues to investigate the effects of OA on natural ecosystems (e.g. Hall-Spencer et al. 2008; Fabricius et al. 2011; Lombardi et al. 2011; Calosi et al. 2013; Lamare et al. 2016; Dahms et al. 2018). They have enabled research into the effects of long-term in situ exposure of multiple biota to elevated pCO2 under natural environmental variability, food, light and hydrodynamic conditions, as well as natural levels of interactions between species and their environment (e.g. Hall-Spencer et al. 2008; Fabricius et al. 2011, 2014; Brinkman and Smith 2015; Nagelkerken et al. 2016). Thus, submarine CO2 vents have gained increasing popularity in the past decade as natural laboratories to obtain a better understanding of CO2 effects on community, population, ecosystem structure and functionality changes, as well as species-specific adaptation mechanisms in a natural environment (e.g. Lombardi et al. 2011; Calosi et al. 2013; Lamare et al. 2016; Dahms et al. 2018; González-Delgado and Hernández 2018).
The main limitations in using shallow marine hydrothermal systems as natural analogues of future conditions are:
-
the large temporal variabilities in pH with periods of time where pH values reach ambient levels or undersaturated conditions (e.g. Boatta et al. 2013; Fabricius et al. 2014; Dahms et al. 2018; González-Delgado and Hernández 2018; Pichler et al. 2019)
-
the limited area affected by vent emissions and resulting area with low pH and potentially higher temperatures (Boatta et al. 2013; Pichler et al. 2019)
-
the continuous supply of larvae and propagules from outside the vent system (e.g. Levin et al. 2016; Boyd et al. 2018; Hawkins et al. 2018), which hampers the ability to use these sites to determine whether dispersive populations could adapt to environmental stressors over multiple generations
-
the eventuality that some organisms that live at CO2 vents could have higher tolerance thresholds for environmental stressors owing to the possibility of evolving and genetically adapting over long timescales to extreme physicochemical conditions (e.g. Hall-Spencer et al. 2008)
-
the chemical composition of hydrothermal fluids at many vent sites (i.e. the potential presence at toxic concentrations of elements and gases), which may have confounding effects on vent biota (e.g. Cunha et al. 2008; Chen et al. 2018; Dahms et al. 2018; González-Delgado and Hernández 2018; Hawkins et al. 2018; Pichler et al. 2019).
Most CO2 vent studies to date have focused on the ecological consequences of lower pH, lower carbonate saturation state or higher CO2 (e.g. Hall-Spencer et al. 2008; Fabricius et al. 2011, 2014; Brinkman and Smith 2015; Nagelkerken et al. 2016), whereas elevated levels of heavy metals in vent fluids compared with non-hydrothermal environments and their potential effects on the local environment (Chen et al. 2018; Hawkins et al. 2018) have either largely been overlooked, dismissed as insignificant or were studied using methods unable to detect bioactive trace metals with necessary sensitivity. Emanating fluids of many volcanic submarine CO2 vents are typically characterised by high concentrations of toxic metals such as Pb, Cd, Zn, Hg, Cu, Co, Mo and Sr (Cunha et al. 2008; Chen et al. 2018; Hawkins et al. 2018; Burger and Lichtscheidl 2019). These metal-rich fluids contaminate the surrounding water column, and potentially adversely affect the physiology of vent biota (Cunha et al. 2008; Chen et al. 2018; Hawkins et al. 2018). Thus, it is surprising that so far most shallow vent studies of biological nature limited their element measurements to gases (e.g. CO2, H2S, CH4, N2, O2; Pichler et al. 2019) and sediments (e.g. Vizzini et al. 2013), despite elements in solution being potentially key factors for the assessment of the suitability of vent systems for the study of climate change effects on marine biota (Vizzini et al. 2013). Consequently, an integrated picture of the carbonate chemistry, biology and, in particular, the geochemistry (i.e. element composition, metal speciation and bioavailability) of shallow submarine vents is required to simulate and investigate OA at CO2 vents.
The most well-studied shallow submarine CO2 vents in the context of climate change are in the warm temperate Mediterranean Sea (e.g. Hall-Spencer et al. 2008; Kroeker et al. 2011; Lidbury et al. 2012; Vizzini et al. 2013; Baggini et al. 2014; Ziveri et al. 2014; Cornwall et al. 2017a; Hawkins et al. 2018), in deep water (e.g. Rossi and Tunnicliffe 2017) or in tropical to subtropical coral reef environments in Papua New Guinea (e.g. Fabricius et al. 2011; Morrow et al. 2015; Takahashi et al. 2016; Pichler et al. 2019), Japan (Inoue et al. 2013; Agostini et al. 2015), Canary Islands (Hernández et al. 2016) and Mariana Islands (Enochs et al. 2015). OA studies of shallow vents in cooler waters of the Southern Hemisphere are still scarce. Therefore, the present study focused on volcanic submarine CO2 vents off Whakaari–White Island in New Zealand’s Bay of Plenty, in a temperate location for OA research (Brinkman and Smith 2015; Nagelkerken et al. 2016). Seawater immediately adjacent to the vents is lower in pH and higher in temperature than the ambient seawater, even given the natural diurnal variation typical of rocky kelp-dominated ecosystems (Cornwall et al. 2013). Nevertheless, the working area is home to a variety of calcified and non-calcified marine biota and is accessible by SCUBA, highlighting its potential for OA research. The results of previous surveys (Grace 1975; Brinkman and Smith 2015; Nagelkerken et al. 2016; Connell et al. 2018) suggested that these cool temperate CO2 vents can offer a natural laboratory for studying the response of temperate communities to future pH and temperature projections for the year 2100 (Gattuso et al. 2015). However, in some studies the effect of clearly enriched heavy metals and elements, such as Hg and sulfide, in the water column around the vents was either omitted in their overall conclusions or was thought to have negligible effects on prevalent marine biota (Brinkman and Smith 2015). Consequently, this study aimed to combine carbonate chemistry, biology and, in particular, geochemical data to evaluate the suitability of the Whakaari vents as potential natural study sites of OA in the Southern Hemisphere. To conclusively confirm the potential of these shallow vent areas for OA research, any possible effects of elevated trace element concentrations must be precluded (Vizzini et al. 2013).
Grange et al. (1992) and Tarasov (2006) described Whakaari–White Island as ‘possibly unique’. Certainly its accessibility, its continuous production of ash and steam (but not lava) and the proximity of a temperate kelp forest community to CO2 vents are special and unusual features, which could make Whakaari’s vents a valuable addition to current OA research so long as any geochemical parameters are within acceptable limits. In order to establish basic biogeochemical information and examine Whakaari’s potential for further study as a natural analogue for future climate change effects, a group of scientists converged on the island in December 2015. In a single week, physical, geochemical and biological characteristics were measured at three vent sites, three control sites near the volcano and one coastal control to provide a cross-disciplinary snapshot of the seawater chemistry and ecology around Whakaari in early summer. Here we report on the preliminary results of the ‘White Island Blitz’ expedition and evaluate the future utility and potential of Whakaari as a ‘living laboratory’ for climate change science.
Materials and methods
Ethics
This study does not report on animal work and thus did not require any animal ethics approval. Biological (i.e. algae) and water samples were collected under a Special Collection Permit from the Ministry of Primary Industries (number 644-2).
Study location
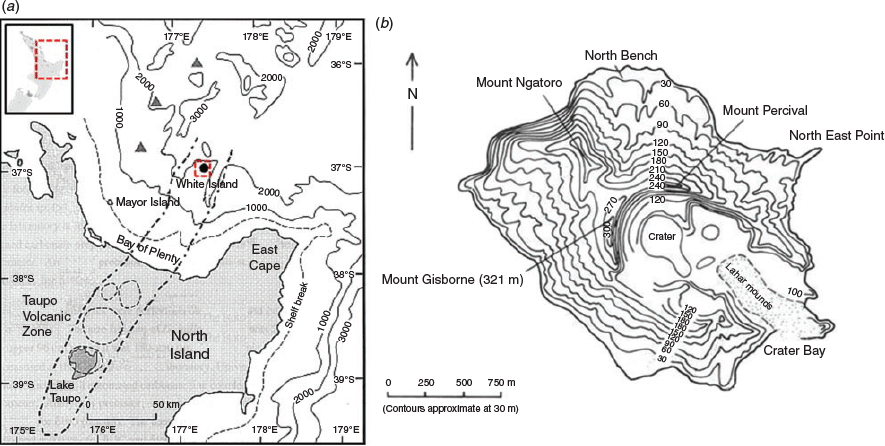
Whakaari–White Island is an active andesite stratovolcano located ~50 km offshore in the Bay of Plenty, North Island, New Zealand (37°31.19′S, 117°10.85′E; Fig. 1). The island is part of the Taupo Volcanic Zone in the back-arc basin of the Kermadec–Tonga subduction zone. The island is approximately circular, covering 238 ha, ~2 km in diameter (Fig. 1). The highest point of the crater wall, Mount Gisborne, is 321 m above sea level (Clarkson and Clarkson 1994) and ~1600 m above the sea floor (Fig. 1). White Island is New Zealand’s most active cone volcano, and has probably been active for at least 16 000 years (Grange et al. 1992). There have been periods of fairly quiet steaming, during which scrubby forests were able to develop, but then periods of heightened activity (e.g. 1976–90; Clarkson and Clarkson 1994), with nearly continuous outpouring of ash, steam and gas, killing trees and shrubs and resulting in a fairly barren landscape (Clarkson and Clarkson 1994). Nevertheless, the island presently supports a breeding colony of gannets and areas of vegetation. Eruptions, which are infrequent, can be quite violent, with andesite blocks and ash being hurled into the sea.
|
The island has a well-developed terrestrial hydrothermal system, consisting of saline fluids around the active vents (Giggenbach 1987). These magmatic fluids interact with meteoric fluids, magmatic gases (70 : 29 : 1 C : S : Cl; Giggenbach 1975), host rocks and metalliferous deposits (rich in V, Cr, Fe, Co, Ni, Rb, Mg, Al, Mn, Zn and Cu; Cole et al. 2000) to produce hot springs and fumaroles (fumarole gases are normally 100–300°C but rise to 1000°C during strong activity; Sarano et al. 1989). The composition of gas fumaroles around Whakaari varies with temperature (Giggenbach 1975), but in all cases the dominant component, besides water vapour, is CO2, followed by SO2, H2S and HCl. Dissolved volcanic gases in hydrothermal fluids give rise to strongly acidic (~1.7 pH) and thus metal-rich surface waters (Donachie et al. 2002) that drain into the surrounding sea, particularly at Crater Bay (Fig. 1), to the south-east of the island (Grace 1975).
Diffuse fluid sources and gas vents occur also in the shallow waters (10–30 m) north and east of the island (e.g. ‘Champagne Bay’; Sarano et al. 1989) with warm water (18–55°C; Tarasov 2006) and gas bubbles (containing CO2, N2, CH4, C2H6, H2, H2S; Propp et al. 1992; Tarasov et al. 1999; Hocking et al. 2010; Brinkman and Smith 2015) emerging from among boulders or out of the sand. Measurements of the emanating fluids showed pH values as low as 7.5 (Brinkman and Smith 2015) and high concentrations of elements and metals, such as S, K, Hg, Rb, Mg and Ca (Brinkman and Smith 2015).
Intertidal fauna are low in abundance and diversity (Grange et al. 1992) and exhibit shell pitting and staining, perhaps due to acidified rain or ashfall (Grace 1975). Subtidally, fish are plentiful and macroalgae (i.e. kelp Ecklonia radiata forests) are abundant, but species composition is different from and diversity is lower than nearby Mayor Island or other coastal islands around north-eastern New Zealand (Grange et al. 1992). Submarine invertebrates are neither diverse nor abundant around the vent sites, whereas bacterial biomass is noticeably greater around the CO2 vents (Grange et al. 1992). Subtropical marine species are sometimes found here because of the warm East Auckland Current (Grace 1975), which brings clear subtropical waters from the north-east of the North Island into the Bay of Plenty. Geostrophic currents flow across the Bay of Plenty from west to east, at an average speed of 0.14 m s–1 (Ridgway and Greig 1986). Mean sea surface temperature in the area is 21°C in summer and 15°C in winter, and surface salinity is typically 35.0–35.7 (Ridgway and Greig 1986).
Sampling sites
Collection of seawater and biological samples occurred during New Zealand’s summer in 2015 over three consecutive days in early December. Sampling was conducted during severe weather with heavy surge and strong winds resulting in challenging sampling conditions for divers and the boat crew. Sampling resolution was consequently lower than planned.
Three natural CO2 vents (V1, V2 and V3) and four control sites (C1, C2, C3 and C4) were sampled near White Island and in the Bay of Plenty (Fig. 2; Table 1). The seven sites were chosen based on their variety of vent activity and their biological diversity (i.e. calcified and non-calcified macroalgae, as well as numerous species of invertebrates and fish). The vent locations were chosen based on previous studies and included one large vent (V1; previously examined by Brinkman and Smith 2015), a smaller vent (V2) and a moderate vent (V3). Vent sites V1 and V2 were located 8–10 m below the surface at the north-eastern tip of the island, whereas V3 was on the western side at a depth of 14.5 m. Control sites were chosen, based on accessibility and weather conditions during sampling, with at a distance of at least 25–30 m from visible vent activity. Prevailing water flow in New Zealand’s Bay of Plenty is west to east as part of the East Auckland Current (Ridgway and Greig 1986), and thus control sites C3 and C4 experience flow regimes most similar to the sampled vent sites on the east side of the island. The control site C4 was located midway between V1 and V2 and was referred to as a ‘vent control’, whereas C3 was established in the outer part of the bay, north of the vent field, and was expected to be more representative of pH around the island with less influence from the vent field. The control site C2 located on the western site of the Island was established outside the influence of the Whakaari vent field and was thus expected to be more representative of ambient seawater conditions. The open ocean control sample (C1) was taken approximately mid-way between White Island and mainland New Zealand over the continental shelf, thus giving a regional context.

|
Sample collection and sampling procedure
Sampling – water chemistry. Temperature, conductivity (salinity) and depth were measured in situ at the study sites using an RBR CTD XR 620 (RBR Ltd, Stadhampton, UK) alongside water sampling. In addition, seawater pH (on the total scale, hereafter pHT) and temperature were monitored half hourly from 13 to 27 November using SeaFET pH sensors (Sea Bird Scientific, Bellevue, WA, USA) placed on the reef within V1 and V2 and at two nearby control locations (C3 and C4; Fig. 2). The SeaFET sensors were deployed to investigate the temporal variability of pHT and temperature around Whakaari. Unfortunately, there was an unknown problem with the SeaFET sensor at C3, and the data were deemed unreliable. However, additional data are available from C3 and C4 from before and after the long-term deployment, which allows some comparison in pHT between the two controls.
Vent samples for total dissolved trace metal concentrations, Cu speciation, total mercury (HgT), sulfide (the term ‘sulfide’ used in this study refers to total reduced sulfur; i.e. the sum of H2S, HS– and S2–) and nutrient (ammonium, nitrogen oxides (NOx) and phosphate) concentrations were taken by SCUBA divers directly at the vent outflow (which, for the larger vents, was beneath the reef surface), and at a distance of 2 m from the emanating vent fluid at ~1 m from the bottom using acid-cleaned 5-L collapsible containers. Surface and subsurface samples at the vent and control sites were taken from the research vessel for the same parameters using a peristaltic pump connected to acid-cleaned polyethylene tubing, which was rinsed with 1 L of sample seawater before sample collection, except for sample C3 SW, which was taken manually by submersing a sample bottle from aboard ship because of the huge swell. After recovery, sample aliquots reserved for trace metal, Cu speciation, HgT, sulfide and nutrient analysis were subsequently filtered in-line through a 0.2-μm polycarbonate cartridge filter (Acropak, Pall Corporation, Port Washington, NY, USA; one filter per site) and individually stored in precleaned and rinsed low-density polyethylene (LDPE) bottles. Total dissolved trace metal samples were stored in the dark at room temperature in two plastic bags until they were acidified to pHNBS 2 using quartz distilled HCl (analytical grade; Scharlau, Scharlab, Barcelona, Spain; 37%) in the laboratory of the University of Otago (Dunedin, New Zealand). Sample bottles for Cu speciation analysis were double-bagged and immediately frozen (–20°C) and kept frozen until analysis. Bottles reserved for nutrient and HgT analysis were placed into two resealable plastic bags, refrigerated (4°C) and stored in the dark until analysed in the laboratory. HgT samples were acidified in the laboratory of the University of Otago with 0.5% (v/v) HCl and subsequently sent to the University of California (Santa Cruz, CA, USA) for analysis. Sample analysis for sulfide was conducted on board. Equipment and sample bottles used for total dissolved trace metals, HgT and Cu speciation analysis had been cleaned according to conventional trace metal clean protocols (Sander et al. 2009; Leal et al. 2016).
Dissolved inorganic carbon (CT) and total alkalinity (AT) samples were collected following the method prescribed by Dickson et al. (2007). Vent samples were collected in 1-L Schott glass bottles without headspace by SCUBA divers, and surface samples were collected by hand off the side of the boat. Two samples were collected at each site and each sample was analysed for both CT and AT. Immediately after sample recovery, 5 mL of each sample was discarded and 0.5 mL of saturated HgCl2 solution was added to poison and preserve the samples (i.e. to avoid biological alteration) for later analysis at the National Institute for Water and Atmospheric (NIWA)/University of Otago Research Centre for Oceanography.
Sampling – biological samples. Samples for phytoplankton species composition were collected at five different stations (Table 1). Surface samples at Champagne Bay (C3 SW) were taken off the side of the boat, whereas the other deeper samples were pumped on board using a peristaltic pump set-up. Samples for larger phytoplankton cells (>20 µm) were stored in 200-mL brown borosilicate glass bottles and preserved with 0.5 mL of Lugol’s solution. Samples for analysis of small phytoplankton species (<20 µm) by flow cytometry were snap frozen in liquid nitrogen until analysis in the home laboratory at the University of Otago.
Zooplankton samples were collected using a vertical haul of a 0.5-m diameter, 150-µm plankton net from a depth of 10 m at C1, C3 and V1. Three replicate zooplankton hauls were taken at each location (total n = 9), which, assuming 100% efficiency, filtered 1.95 m3 of seawater. Samples were subsequently fixed with 2–4% formalin. Unfortunately, poor weather prevented a representative zooplankton sampling haul at V1 at a depth of 10 m. In addition, extreme mixing within Champagne Bay meant that the V1 sample was contaminated with benthic crustaceans and coralline algae.
Seven individuals of the two most abundant species of coralline alga (i.e. Corallina officinalis, n = 5; and Amphiroa anceps, n = 2) around the study sites were collected by SCUBA divers from V1 (n = 1), V2 (n = 2) and C2 (n = 4) using hammer and chisel. Specimens were stored on silica gel for analyses of δ11B at the University of Western Australia (Crawley, WA, Australia) to subsequently determine the pH in the calcifying fluid (pHcf) of the collected coralline alga (Cornwall et al. 2017b).
Analysis of water chemistry
Analysis of physicochemical parameters
Nutrient samples were analysed in duplicate at the University of Otago using a Lachat QuikChem 8500 Series 2 flame ionisation auto analyser (Hach, Auckland, New Zealand). CT and AT were both measured at the NIWA/University of Otago Research Centre for Oceanography. AT was measured by potentiometric titration in a closed cell using a custom-built system (Metrohm Dosimat 765, Metrohm AG, Herisau, Switzerland; Fluke high-precision voltmeter, Fluke Calibration, Everett, WA, USA; in-house-built closed cell and custom-written software) following the method of Dickson et al. (2007). CT was determined on a custom-built system (following the Guide to Best Practices SOP2; Dickson et al. 2007) by coulometric analysis (UIC Model 5011, UIC Inc., Joliet, IL, USA) of evolved gas (single-operator multi-parameter metabolic analyser (SOMMA) style CO2 extraction system). The accuracy of both methods, as determined by analysis of certified reference materials (provided by Andrew Dickson, Scripps Institution of Oceanography, San Diego, CA, USA), is estimated to be ±2 μmol kg–1 for AT and ±1 μmol kg–1 for CT. CT and AT data were used to calculate pHT (total scale), pCO2, CO32– concentration and calcite and aragonite saturation states (Ωcalcite and Ωaragonite respectively) of each sample at the in situ temperature, salinity and pressure (CTD XR 620 values) using the refitted Mehrbach CO2 equilibrium constants (Mehrbach et al. 1973; Dickson et al. 2007). Stored CT and AT samples were analysed within 3 months of collection.
Analysis of total dissolved trace metals, HgT, sulfide and Cu speciation
Total dissolved trace metal measurements for Mn, Fe, Co, Ni, Cu, Zn, Cd and Pb were made at the Centre for Trace Metal Analysis at the University of Otago using an automated off-line SeaFAST (Elemental Scientific Instruments, Omaha, NE, USA) preconcentration system (Biller and Bruland 2012; Middag et al. 2015) combined with a subsequent trace metal analysis by a Nu Attom high-resolution sector field inductively coupled plasma mass spectrometer (HR-SF-ICP-MS; Nu Instruments Ltd, Wrexham, UK). A detailed description of the method and sample preparation procedure is provided elsewhere (Biller and Bruland 2012; Bown et al. 2017; Rapp et al. 2017). Quantitative resin (Nobias-chelate PA1) recovery was within an acceptable range for the trace metals of interest and the accuracy of the method, verified by multiple measurements of certified reference material (SLEW-3; National Research Council (NRC), Ottawa, ON, Canada), was within ±7% (s.d.) of the certified values for Mn, Fe, Co, Ni, Cu, Zn, Cd and Pb (for more detail, see Table S1, available as Supplementary material to this paper).
Trace metal concentrations for Li, Rb, Sr, Mo, Cs, Ba and U were determined by ICP-MS (NexION; Perkin Elmer, Foster City, CA, USA) at the Jacobs University (Bremen, Germany). For quality control, the seawater reference material NASS-7 (NRC) and an in-house standard were measured along with the samples. The analytical error was within ±8% (s.d.) of the reference values for the trace metals of interest (for more detail, see Table S2).
HgT was measured at the University of California by cold vapour atomic fluorescence spectrometry using the methods of Bloom and Crecelius (1983) and Balcom et al. (2004). Samples were measured in duplicate for quality assurance.
Sulfide (H2S, HS– and S2–) was measured on board using a vibrating, mercury-coated, gold microwire electrode in conjunction with rapid electrochemical cathodic stripping square wave voltammetry (IviumStat electrochemical analyser, Ivium Technologies B.V., Eindhoven, Netherlands) following a procedure adapted from Bi et al. (2013) and Al-Farawati and van den Berg (1997). The electrode was precoated with mercury and calibrated in the field using thiourea, which was found to give a similar sensitivity to that of free sulfide. Samples were measured in duplicate. The concentration of hydrogen sulfide (H2S) in the samples was estimated using the calculation described by Boyd (2014).
Cu speciation parameters (i.e. binding strength of the organic Cu-complexing ligands (log(K)), concentration of organic ligands ([L]) and concentration of bioavailable Cu ([Cu′]; sum of the concentration of free hydrated Cu ions ([Cu2+]) and the concentration of Cu weakly bound to inorganic ligands ([CuXIN])) were determined using adsorptive cathodic stripping voltammetry (AdCSV) with salicylaldoxime as the complexing agent (Lucia et al. 1994; Kleint et al. 2015; Cotte et al. 2018) at the University of Otago on a Metrohm 663 VA stand (Metrohm Autolab B.V., Utrecht, Netherlands). A description of the instrumental set-up is given in Zitoun et al. (2018). Samples underwent pretreatment to remove acid volatile sulfides from the sample solutions, which enabled the electrochemical measurement of Cu′ in sulfide-rich vent samples (Sander et al. 2007; Kleint et al. 2015; Cotte et al. 2018). For comparison purposes, sample aliquots were measured before the acid pretreatment as well as after the acid pretreatment with subsequent filtration (0.2 μm) of the samples to remove potential metal precipitates formed during the pretreatment (Sander et al. 2007). For detailed information on removal of acid volatile sulfides, sample preparation and operating conditions of the instrument, see Sander et al. (2007) and Kleint et al. (2015). Samples were then analysed with the AdCSV method and every measurement was repeated three times. After completion of the titration, Cu speciation parameters of each sample were obtained using the one- or two-ligand complete complexation-fitting model incorporated in the ProMCC software (https://sites.google.com/site/mccprosece/download, accessed 14 August 2019; Omanović et al. 2010). Fitting of the titration data for a two-ligand-system failed with the ProMCC software, and thus samples were fitted to a one-ligand model.
Analysis of biological samples
Analysis of phytoplankton
Larger phytoplankton cells (>20 µm) of each station were counted and identified after the Utermöhl method in Utermöhl chambers (HYDRO-BIOS Apparatebau GmbH, Kiel-Altenholz, Germany) following procedures recommended in Karlson et al. (2011). An Accu-Scope inverted microscope (Olympus, Auckland, New Zealand) was used for counting. Phytoplankton was identified to genus level and down to species level when possible. Samples of small phytoplankton species (<20 µm) were analysed without the addition of preservative immediately after returning to the home laboratory at the University of Otago (within 10 days of sampling). Samples were analysed on an Accuri C6 (BD Biosciences, San Jose, CA, USA). Cryptophytes were identified and counted on the plots of phycoerythrin (PE) v. chlorophyll (CHL) fluorescence. After exclusion of this group, picoeukaryotes, nanoeukaryotes and coccolithophores were identified based on side scatter (SSC) v. CHL fluorescence. The different phytoplankton groups were identified as described in Tarran and Bruun (2015).
Analysis of zooplankton
Zooplankton samples of the three sampled sites were compared at the University of Auckland using a binocular microscope and a Bogoroy tray in a semiquantitative manner, identifying abundant (>10 individuals per sample) and rare categories of both holoplankton and meroplankton.
Analysis of coralline algae
Recent analysis of δ11B has implied that these values can be used as proxies for determining pH in the fluid (pHcf) where coralline algal calcite is formed (Cornwall et al. 2017b, 2018; Comeau et al. 2018). This finding enables us to understand how the vent systems can alter the calcification physiology of these species. Coralline algae collected around Whakaari were processed in the clean laboratory of the Advanced Geochemical Facility for Indian Ocean Research (AGFIOR), University of Western Australia). Sample analysis and preparation followed that of Cornwall et al. (2017b). Boron was quantitatively separated on ion exchange columns according to McCulloch et al. (2014) and boron isotopes were subsequently measured using the NU Plasma II multicollector ICP-MS (Nu Instruments, Charlestown, MA, USA). Boron isotopes were transformed to pHcf using the calculations of Trotter et al. (2011) assuming all primary boron uptake was as the borate species at a temperature of 17.5°C and salinity of 35.0.
Results
Water chemistry
Physicochemical parameters
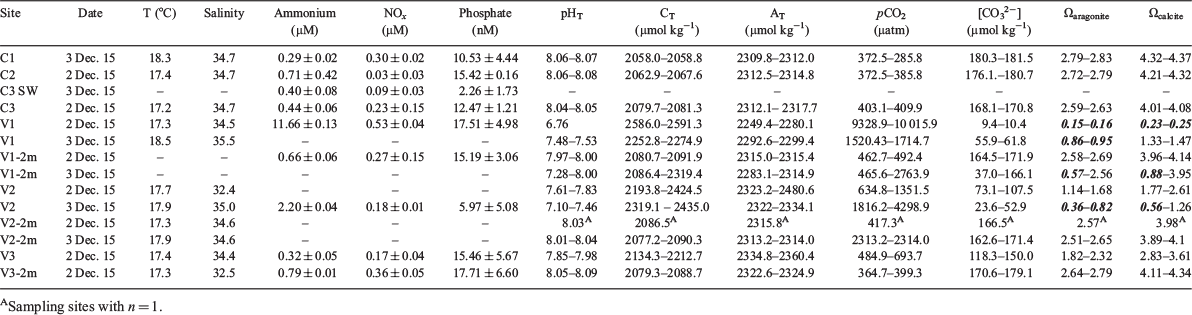
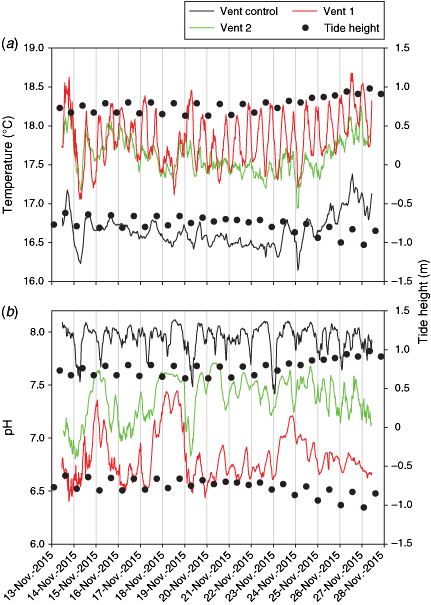
Mean (±s.d.) temperature and salinity values obtained with the CTD XR 620 were 17.68 ± 0.43°C and 34.39 ± 0.91 ppt (Fig. 3; Table 2) during the observation period with rough weather conditions. The highest mean temperatures were recorded at the vent shafts, followed by control sites and then vent 2-m sites. Mean salinities followed the order of control sites > vent 2-m sites > vent shafts (Fig. 3; Table 2). Temperature and salinity values showed some temporal variability of up to ±1.2°C and ±2.59 ppt at the vent sites. Calculated mean (±s.d.) sample pHT for the control stations was consistent with normal present-day pH of seawater (8.06 ± 0.01), whereas vent fluids showed lower mean pHT values of 7.43 ± 0.43 within the vents and 7.94 ± 0.25 at a distance of 2 m from the vent outflow (Fig. 3; Table 2). As expected, because of dilution and mixing processes of the vent fluid with seawater, calculated pHT values commonly increased to ambient seawater pH levels with 2-m distance from the vent outflow. Calculated pHT values showed some temporal variability of up to ±0.71 at the vent sites for samples taken on the same day (n = 2) and up to ±0.77 for samples collected on the two different sampling occasions (Fig. 3; Table 2). The temporal variability of pHT and temperature is more obvious in the SeaFET data. It is worth noting that the CTD XR 620 data were collected during large swells, which possibly resulted in mixing around the sites where measurements were taken, whereas the SeaFET operated from 13 to 27 November under reasonably calm conditions.

|
|
The water temperature obtained with the SeaFET within V1 and V2 was consistently higher than at the vent control site (i.e. C4; Fig. 4a). On average, the water temperature at V1 and V2 was 1.19 and 0.95°C higher than at C4. Consistent with the CTD XR 620 data, temperature was most variable at V1, and this appears to be related to tidal height, with warmest temperatures within the vent consistently associated with low tide (Fig. 4a). Temperatures at V2 did not appear related to tidal height and were positively correlated with temperatures at C4 (r2 = 0.79). Temperature at C4, and to a lesser extent at V2, exhibited diel variation, peaking between 1300 and 1700 hours and lowest from 0200 to 0700 hours (for more detail, see Fig. S1).
|
Seawater pHT obtained with the SeaFET within the vents was highly variable and was, on average 0.54 (V2) and 1.19 (V1) pH units lower than seawater at C4 (Fig. 4b), which is consistent with the CTD XR 620 data. In addition, diel variation was evident at the two vents. Similarly, at C4 mean pH was 7.95 and showed periodic drops in pH to ~7.5. These drops in pH at C4 typically coincided with low tide, particularly at night-time. Overall pH at C4 was weakly correlated with pH at V1 and V2 (r2 = ~0.33 and 0.38 respectively), but many of the large drops in pH at C4 coincided with drops in pH at the vents, suggesting some effect of the vents on pH at C4. The pH at V1 and V2 was more strongly correlated (r2 = 0.54; Fig. 4b). Diel variation in pH was most evident at C4, with lowest pH between midnight and 0700 hours, a steady increase throughout the day and a decline after ~1700 hours (for more detail, see Fig. S1). Additional pH data collected at C3 and C4 suggested pH at C3 was consistently higher (typically >8), with less variation in pH than seen at C4 (Fig. S2). Drops in pH were evident at C3, but they were not as large as those observed at C4.
Nutrient concentrations in the study area were in the range of 0.29–11.66 μM for ammonium, 0.03–0.53 μM for NOx and 2.26–17.71 nM for phosphate. Ammonium and NOx concentrations decreased with distance from the vent outflow, following the order of vent fluids > vent 2-m sites > control sites, whereas phosphate concentrations followed the order of vent 2-m sites > vent fluids > control sites (Table 2).
Values of CT, AT and pCO2 declined from vent fluids to vent 2-m sites to control sites, whereas CO32– concentrations and Ωcalcite and Ωaragonite levels increased along this gradient (control sites > vent 2-m sites > vent fluids). Carbonate chemistry samples collected with distance from the vent fluids (i.e. 2 m from V1, V2 and V3) did not differ significantly from data obtained at the control sites (C1, C2 and C3). Undersaturation with respect to aragonite was observed for V1 during both sampling periods and for V1-2m and V2 on 3 December. Values of Ωcalcite < 1 were calculated for V1 on 2 December and for V1-2m and V2 on 3 December. Values of CT, AT, pCO2, CO32– concentration, Ωcalcite and Ωaragonite suggest large temporal (i.e. samples taken on 2 and 3 December) and environmental (i.e. samples taken on the same day; n = 2) variability in the carbonate chemistry of the vent sites.
Total dissolved trace metals, HgT and sulfide
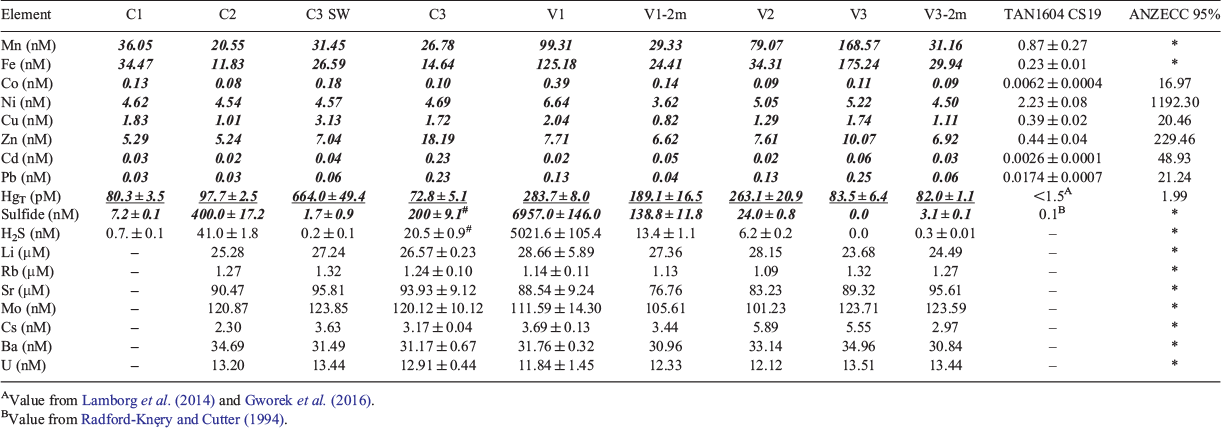
Trace element and metal concentrations of Mn, Fe, Co, Ni, Cu, Zn, Cd, Pb, HgT and sulfide were between ~2 to 70 000-fold higher at all nine study sites than values commonly measured in open ocean seawater samples around New Zealand (TAN1604, Station CS19, depth 25 m, Hauraki Gulf; R. Middag, C. Stirling, M. Reid, and K. Seyitmuhammedov, unpubl. data; Table 3). Mn, Fe and Cs concentrations declined from vent fluids to vent 2-m sites to control sites, whereas Zn, Cu, HgT, Sr and Rb concentrations were in the order control sites > vent fluids > vent 2-m sites (Table 3). Concentrations of Ba, Li, Co, Pb and Ni were in the order vent fluids > control sites > vent 2-m sites, whereas sulfide concentrations were in the order vent 2-m sites > vent fluids > control sites (Table 3). Cd, Mo, and U concentrations were higher at the control sites, followed by vent 2-m sites and then vent fluids (Table 3). However, Li, Rb, Sr, Mo, Ba and U concentrations did not vary considerably between vent and control sites (difference between the maximum and minimum concentration <5%; Table 3). Concentrations of Ni, Zn, Cd, Pb, Co and Cu at all nine study sites at Whakaari were well below the Australian and New Zealand Environment and Conservation Council (2000) guidelines for a 95% level of protection for biota in saltwater systems, whereas the concentration of HgT far exceeded the guideline threshold by up to two orders of magnitude (Table 3).
|
Copper speciation
Titration data demonstrated the presence of one ligand class with uniform Cu-binding capacities (log(K)) of 11.9 ± 0.2 (mean ± s.d.) for sulfide-containing samples and 12.6 ± 0.6 for sulfide-free samples (Table 4). Ligand concentrations ([L]) were higher in the sulfide-containing vent samples (i.e. V1, V2 and V1-2m; Table 3), before the pretreatment compared with [L] in pretreated samples (Table 4). By contrast, the change in [L] between the untreated and pretreated control sample (C3) that had fairly moderate sulfide concentrations was negligible. This trend highlights the important role of sulfide as an inorganic ligand around the CO2 vents at Whakaari.

|
In pretreated samples, mean (±s.d.) [L] ranged from as low as 3.9 ± 0.9 nM for the V1-2m station to 29.4 ± 8.4 nM for the C3 station. The highest [L] was found at C3, followed by V1, V2 and then V1-2m. At V1, [L] decreased with distance from the vent outflow (Table 4), which appears reasonable considering prevailing fluid–seawater mixing and dilution processes. [L] after the pretreatment was always in excess of the total dissolved Cu concentration ([CuT]; Tables 3, 4), with the consequence that [Cu2+] was low in the sampling area. Bioavailable Cu concentrations ([Cu′]) ranged from 0.6 to 2.6 pM, with highest [Cu′] found at C3, followed by V2, V1-2m and then V1.
Biological samples
Phytoplankton community
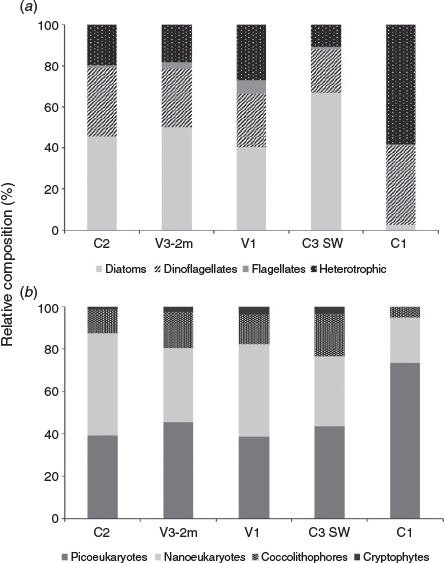
The larger phytoplankton community in the working area was reasonably similar and showed no major differences in either cell concentrations or the number of phytoplankton species between the vent and control sites in the direct vicinity of Whakaari (Fig. 5; Table 5). However, the number of larger phytoplankton species at C1, halfway between White Island and the main coast, was approximately half that of all other sites close to Whakaari (12 v. 21–30; Table 5).
|

|
Further, there were distinct differences in the relative phytoplankton composition of the larger phytoplankton community around Whakaari, with diatoms accounting for 42.9–73.9% of the phytoplankton community at sites in the direct vicinity of Whakaari (i.e. C2, C3 SW, V1 and V3-2m), but only 2.65% at C1 (Fig. 5). At the same time, the relative contribution of dinoflagellates and flagellates to the larger phytoplankton community was fairly stable (at 24.0–39.6 and 0.5–7.5% respectively) in the whole working area (Fig. 5).
At C1, total cell numbers for large phytoplankton were up to twofold higher compared with stations around the island (Table 6). At the same time, at C1 we found less than half the large phytoplankton species compared with the Whakaari stations (Table 5). A large difference was again observed for heterotrophic plankton species, which accounted for 60.0% of total cell numbers at C1 but for only 12.0–22.2% at the four sites (C2, V3-2m, V1 and C3 SW; Fig. 5) in the direct vicinity of Whakaari.

|
The picoeukaryote species (i.e. small phytoplankton groups), as identified by flow cytometry, showed a similar trend, with total cell numbers being approximately twofold higher and only three groups of picoeukaryotes identified at C1 compared with four groups of picoeukaryotes identified at the four sites in the direct vicinity of Whakaari (Fig. 5; Table 6). In addition, the picoeukaryote community showed differences in relative group composition. Although four distinct groups could be identified by flow cytometry at the four sites close to Whakaari, with picoeukaryotes accounting for 38.6–45.5%, nanoeukaryotes accounting for 33.4–48.3%, coccolithophores accounting for 11.2–20.0% and cryptophytes accounting for 1.4–3.8%, only three groups were identified at C1 (Fig. 5). At C1, picoeukaryotes clearly dominated the small phytoplankton community (accounting for 73.6%), whereas nanoeukaryotes and coccolithophores accounted for 21.2 and 5.2% respectively.
Zooplankton community
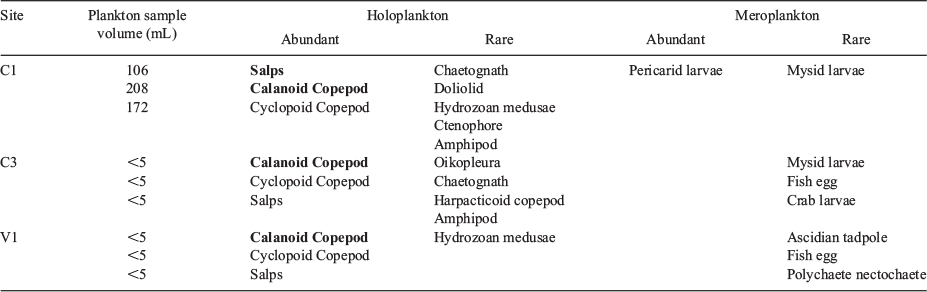
The zooplankton samples of C1 were more than 20-fold the volume of samples collected at the two stations close to Whakaari (C3 and V1) because of the dominance of salps. Calanoid copepods and pericarid larvae were the dominant zooplankton component at C1 (Table 7). Zooplankton samples collected in the direct vicinity of White Island (C3 and V1) contained the same abundant taxa as C1 within a very small plankton volume (<5 mL), with a slightly higher diversity of meroplankton (Table 7). Overall, there was no compositional difference in the abundant zooplankton taxa between vent and control sites.
|
Benthic algae, especially corallines
Articulate and crustose coralline algae were present in reasonably high abundances at Whakaari, both near active venting locations (i.e. V1 and V2) and control locations (i.e. C2; C. Cornwall, pers. obs.). The articulate coralline alga C. officinalis maintained a mean (±s.d.) pHcf of 8.66 ± 0.04 at the venting site and 8.75 ± 0.07 at the control site. A. anceps had a mean (±s.d.) pHcf of 8.67 ± 0.06 at the control site, but was unfortunately not sampled at the vent location (Table S3). Although pHcf is affected by external seawater pH, it was still elevated at the venting location.
Discussion
Even though shallow submarine CO2 vents are not exact analogues of future ocean conditions, they are commonly used as the best natural settings to investigate community composition shifts and ecosystem-scale effects of OA, over long timescales (Hall-Spencer et al. 2008; Fabricius et al. 2011; Pichler et al. 2019). The suitability of the unique rocky coastal temperate submarine CO2 vents around Whakaari–White Island in the Bay of Plenty (New Zealand) as a ‘living laboratory’ for OA science is discussed below. However, it has to be noted that the research findings of this study are limited by the short sampling period and the low sampling resolution, which prohibited statistical analysis. These limitations add a level of uncertainty to data interpretation.
Overall water chemistry
Physicochemical parameters
At Whakaari–White Island, vent sites were strongly influenced by the tides and the volcanic CO2 seeps resulting in localised acidification of seawater (i.e. decreased mean pHT), large pHT fluctuations and a mean decrease in Ωcalcite and Ωaragonite relative to control sites (Fig. 3, 4; Table 2). In the proximity of the vents (i.e. V1, V2 and V3), pCO2 levels were high, ranging between 484 and 10 015 µatm, and calculated seawater pHT values were constantly lower than 7.98, which are projected average global sea surface pH values for the year 2100 and beyond (e.g. Caldeira and Wickett 2003; Gattuso and Lavigne 2009). As a result, Ωcalcite and Ωaragonite at the vent sites (V1 and V2) substantially decreased to values <1, producing progressively lower saturation states for prevalent calcifying organisms. In addition, the water temperature at V1 and V2 was consistently higher than at the control site C4 (Fig. 4a), which makes the White Island CO2 vents also useful analogues for the effect of ocean warming on marine biota (González-Delgado and Hernández 2018). As expected, the effects of volcanic vent emissions on pCO2, pHT and carbonate chemistry drop off quickly with distance from the vent outflow (Brinkman and Smith 2015; Pichler et al. 2019), creating close to ambient seawater conditions for biota at a distance of 2 m from the active vent sites on the dates sampled for this study during rough weather.
Even though the SeaFET data of Whakaari demonstrate a well-mixed water column during the ‘White Island Blitz’ due to the large swell (Fig. S2), pHT values and other carbonate chemistry parameters still showed large temporal variability at the vent sites during the 3-day sampling trip (Table 2). SeaFET data taken during an earlier study in the working area in November 2015 (see Fig. 4, S2) show more intense sudden changes, extreme variations and irregular fluctuations in pHT at Champagne Bay, which is in accordance with pH data recorded at other CO2 vent settings off Ischia (Italy; Kerrison et al. 2011) and Ambitle Island (Papua New Guinea; Pichler et al. 2019). This variability is typical for dynamic CO2 vent systems that can undergo short-term changes in physicochemical conditions due to changes in geological conditions (e.g. German and Von Damm 2003), volcanic activity (Chiodini et al. 2006), discharge rates, chemical composition of hydrothermal fluids and gases (Pichler et al. 2019) and hydrodynamic conditions (i.e. tidal cycle, storms, wind, stratification, currents) of the surrounding water column. Hence, as suggested by Pichler et al. (2019), it is important to describe hydrothermal study sites using either continuous or multiple discrete measurements over time of abiotic parameters (i.e. temperature, salinity, pH, CT, AT, pHT, pCO2, [CO32–], Ωcalcite and Ωaragonite) to better quantify means and variability, as well as to account for short-term fluctuations that can affect the biological responses of prevalent biota. This is particularly important for calcifying organisms when undersaturated conditions for aragonite and calcite continue for a long time, affecting their calcification rates and acid–base regulation processes in the absence of protective mechanisms (Fabry et al. 2008). However, the biological implications of natural physical and chemical fluctuations for flora and fauna adjacent to vent sites are so far scarce (e.g. Kerrison et al. 2011), and their effects are difficult to reproduce in laboratory experiments (Rivest et al. 2017). Nevertheless, with seawater pH, temperature and pCO2 variability expected to intensify in the future global ocean, considerations of the natural environmental variability of abiotic parameters are essential to understand species susceptibilities to future ocean conditions and to predict future organism responses at physiological, ecological and evolutionary levels.
In the marine environment, nitrogen and phosphorous are important nutrients affecting primary production, aquatic plant growth and the community composition of phytoplankton, macroalgae and vascular plants (e.g. Ngatia et al. 2019). The Redfield ratio (N : P) in the working area of Whakaari ranged between 31 and 695, and was thus above the optimal N : P ratio for marine biota of 16 (Redfield et al. 1963). However, an N : P ratio >16 suggest a P limitation of the study area (Redfield et al. 1963). The Australian and New Zealand Environment and Conservation Council (1992) recommends that marine (coastal) nitrogen and phosphorus concentrations should not exceed PO4-P values of 10 µg L–1, and NO3-N values of 60 µg L–1, and should not fall short of NH4-N values of 5 µg L–1 to prevent adverse effects on the ecosystem (i.e. eutrophication leading to hypoxia and anoxia, reduced water quality, changes in food web structure, habitat degradation, loss of biodiversity and noxious and harmful algal blooms; Ngatia et al. 2019). All nutrient samples measured around Whakaari–White Island were within these Australian and New Zealand Environment and Conservation Council (1992) guidelines, suggesting that the nutrient composition of the study area is suited to the requirements of prevalent biota.
Total dissolved trace metals, HgT and sulfide
Elements are naturally present in seawater and are either enriched or impoverished in hydrothermal fluids relative to their background content in ambient seawater (e.g. German and Von Damm 2003; Tarasov 2006; Kleint et al. 2015; Pichler et al. 2019). The main processes affecting the concentration, speciation and bioavailability of elements in the water column around CO2 vents are precipitation as sulfides, sediment resuspension, complexation with ligands and biological uptake (e.g. Bruland et al. 2014; Sander and Koschinsky 2011). Therefore, the enrichment of trace metals in the hydrothermal fluids of the working area relative to their concentrations in open ocean seawater samples around New Zealand (TAN1604, Station CS19, depth 25 m, Hauraki Gulf; R. Middag et al., unpubl. data; Table 3) may be explained by ligand complexation, which stabilises dissolved metals in solution, or by the resuspension of metal-rich sediments within the dynamic Taupo Volcanic Zone (Sarano et al. 1989). Zn, Cu, Cd, Sr, Rb, Mo, U and HgT concentrations were highest at the control sites, suggesting a terrestrial input of these metals, whereas Ba, Cs, Li, Mn, Fe, Ni, Pb and Co concentrations were highest at the vent shafts, which hints towards a hydrothermal source of these metals. However, Li, Rb, Sr, Mo, Ba and U concentrations were quite uniformly mixed in the working area due to the large swell experienced during sampling, which highlights the difficulties in finding suitable control sites in the Bay of Plenty, at least during bad weather conditions. Because sulfide is a common low-temperature product of hydrothermal activity (Boatta et al. 2013), we expected enriched sulfide concentrations in the working area, except for V3, particularly because Whakaari has large terrestrial sulfur deposits (Nairn et al. 1996). Future studies are needed to assess bioaccumulation and the importance of biological uptake of these elements and metals in the working area.
Some trace metals, such as Mn, Fe, Co and Zn, are essential elements for the metabolism of organisms, whereas other metals, such as Cd, Pb and Hg, have no biological function and are entirely toxic to marine organisms, even at low concentrations (Rainbow 2002; Jakimska et al. 2011; Tercier-Waeber et al. 2012), or, in the case of Cu, are both essential and toxic depending on the concentration (Bruland et al. 1991; Kleint et al. 2015). Concentrations of Zn, Cu, Co, Cd, Pb and Ni around Whakaari were well below the Australian and New Zealand Environment and Conservation Council (2000) guidelines for a 95% level of protection for biota in saltwater systems, whereas the concentration of HgT far exceeded the guideline threshold at all sites, including the open ocean control site C1 ~40 km away from White Island, by up to two orders of magnitude. High Hg concentrations around White Island were also reported by Brinkman and Smith (2015). As a result of the high HgT concentrations in the water column around White Island, there is a risk of its accumulation in aquatic organisms, which is not only a threat for the organisms themselves, but also a concern for human health following the consumption of locally caught seafood from this area (e.g. Mousavi et al. 2011; Gworek et al. 2016). However, understanding Hg toxicity requires an understanding of its speciation, because different forms of Hg are variously toxic to aquatic biota and humans (Mousavi et al. 2011). In light of addressing the toxicological concerns of Hg around White Island, future studies need to investigate the occurrence and speciation of Hg at the vent sites, and quantify the degree of methylation and the adsorption and accumulation levels of Hg in local biota. Owing to bioaccumulation, the more toxic organic Hg species are usually elevated in higher trophic levels of fish, which may be a concern for human health at this popular recreational fishing spot. Similarly, macroalgae accumulate contaminants in proportion to their environmental concentrations (e.g. Karthick et al. 2012), and thus future studies could include macroalgae to monitor and characterise the status of environmental risk and water quality in the shallow waters around Whakaari.
Trigger values for Li, Rb, Sr, Mo, Cs, Ba, U, Mn, Fe and sulfide are not given in the Australian and New Zealand Environment and Conservation Council (2000) guidelines. However, H2S is a known toxicant in the marine environment because it imposes severe respiratory stress to aquatic biota even at low concentrations, and thus affects the health, survival, productivity and distribution of various organisms (Jahn and Theede 1997; Boyd 2014). HS– anions may also contribute to toxicity at high sulfide concentrations (Jahn and Theede 1997; Boyd 2014), whereas S2– ions are not an issue because they only occur in the water column at pH values >12 (Boyd 2014). The 96-h LC50 values of H2S to marine species range from 1.5 to 14.7 µM, but should not exceed 146.6 nM in aquaculture (Boyd 2014). According to these values, the water column around V1 is considered harmful for local biota. Nevertheless, vent organisms could be ecologically adapted to high sulfide concentrations in their environment (Dahms et al. 2018), but more research is needed to validate this assumption.
The elevated HgT and sulfide concentrations, among other elements, found at the control sites, especially the open ocean control site C1, suggest that other sites in the Bay of Plenty are affected by extensive hydrothermal activity, which accords with earlier findings of Sarano et al. (1989) and Kleint et al. (2015). These two studies concluded that hydrothermal activity and element signatures are not confined to the vicinity of the White Island CO2 vents, but result from the positioning of various other submarine CO2 vents and gas seeps in the Bay of Plenty that extend along the whole Taupo Volcanic Zone (mainly between Whale Island and White Islands). Therefore, there are no ‘real’ experimental control sites in the Bay of Plenty with regard to the element and metal composition of the water column. However, there may be a possible bias in the hydrothermal element signature collected during the ‘White Island Blitz’ due to the influence of large swells, which likely magnified the spatial distribution of the elements in the water column around the working area. That is, if the regional context of water chemistry is to be understood, there is a need to understand the temporal variability of the element and metal composition in the Bay of Plenty during both calm and well-mixed periods.
For the shallow CO2 vents around Whakaari sampled in this study, high amounts of Mn, Fe, Co, Ni, Cu, Zn, Cd, Pb, HgT and sulfide were observed, which could confound the effects of pCO2 and low pH on the ecosystem in isolation. However, we emphasise that this study does not establish such confounding effects. We suggest that scientists should be cautious when relating ecosystem-scale effects and biological responses of prevalent biota along pH gradients in the working area to the unifactorial and direct effect of pH, because interactions with concurrent, multiple simultaneous stressors, including bioactive metals, high sulfide concentrations and fluctuation of carbonate chemistry, may occur (Boyd 2010; Vizzini et al. 2013; Chen et al. 2018; Dahms et al. 2018). Knowledge of whether these stressors will have synergistic, antagonistic or additive effects is limited because of the scarcity of published research, but multiple stressors have the potential to affect the environment of Whakaari at multiple scales (e.g. Ivanina and Sokolova 2015; Boyd et al. 2018). To progress an understanding of the individual and interactive effects of these multiple stressors, both laboratory and field studies are needed so that the effects of these stressors on biotic responses can be understood when using vents as a natural laboratory for studying biotic responses to projections of future climate.
Copper speciation
Chemical speciation is the driving factor for understanding metal bioavailability and toxicity to marine organisms (Van Briesen et al. 2010). According to the literature (e.g. Morel 1983; Kozelka and Bruland 1998; Paquin et al. 2000), metal toxicity is primarily related to the labile inorganic metal fraction, consisting of free hydrated trace metal ions and trace metals weakly complexed to inorganic ligands, rather than the total dissolved metal concentration in a system. However, some forms of organically complexed metals are also considered bioavailable (Lorenzo et al. 2005; Semeniuk et al. 2015), but it is generally accepted that the labile inorganic metal fraction represents the most readily bioavailable, and thus toxic, form of metals to marine organisms. Organic ligands generally increase the stability and effective solubility of trace metals in the water column, and thereby decrease the biologically available metal fraction in the surrounding water and the metal toxicity to marine biota (e.g. Laglera and Van den Berg 2009; Bundy et al. 2015; Whitby et al. 2018).
The voltametric data of the present study demonstrated the presence of stable organic and inorganic (i.e. thiols and sulfide) Cu complexes in the hydrothermal vent fluids around Whakaari. Values of [L] in analysed samples were consistently in excess of the measured [CuT], suggesting that most of the CuT in the water column was complexed and stabilised by ligands (~99.9%), thereby reducing the concentration of the bioavailable free Cu2+ ion to optimal levels between Cu2+ deficiency (≤10 fM; Peers et al. 2005; Maldonado et al. 2006; Annett et al. 2008; Amin et al. 2013; Jacquot et al. 2014) and Cu2+ toxicity (>1 pM; Brand et al. 1986) for prevalent marine microorganisms (Kiaune and Singhasemanon 2011; Tercier-Waeber et al. 2012). Further, [L] exceeded [CuT] and the [CuT] was significantly higher at C3 and the [L] followed that trend, which strongly hints towards an active biological ligand production by local biota to mitigate the toxic effects of elevated Cu2+ concentrations around C3. This theory is further supported by the mean log(K) value of 12.6 ± 0.6 for sulfide-free samples around Whakaari, because log(K) values in the range of 10–13 are predominantly attributed to marine-derived humic substances or thiols (Whitby et al. 2018) produced by bacteria (e.g. Shimotori et al. 2009; Romera-Castillo et al. 2011), decaying phytoplankton (e.g. diatoms; Lorenzo et al. 2007), mussel embryos (Mytilus galloprovincialis; Zitoun et al. 2018) and other microorganisms (e.g. Synechococcus, Fucus vesiculosus and Emiliania huxleyi; Brand et al. 1986; Moffett et al. 1990; Moffett and Brand 1996; Gledhill et al. 1999; Leal et al. 1999; Klevenz et al. 2012; Echeveste et al. 2018) during Cu2+ stress, which adds an additional cost for their fitness (e.g. growth, development and reproduction; Ivanina and Sokolova 2015) but provides an advantage when exposed to potentially harmful Cu loads. All in all, ligands play an important role in controlling the solubility, bioavailability and toxicity of CuT around the shallow CO2 vents of White Island.
The active production of ligands may be a general survival strategy of prevalent biota around Whakaari, and some of these ligands may have binding affinities for multiple metals (Kogut and Voelker 2001) and thus ameliorate the potential toxicities of several free metal ions by the formation of metal–ligand complexes. As a result, the importance of ligands in the working area may not be confined to Cu, and may be conferred to other bioactive metals that are enriched in the water column, including Mn, Fe, Ni, Zn and Hg (e.g. Lamborg et al. 2003; Vraspir and Butler 2009; Oldham et al. 2015). Consequently, ligand complexation of bioactive metals around Whakaari may allow biota to tolerate otherwise toxic metal concentrations in their environment, and hence may play a critical role in shaping the environmental niches and survival strategies of vent organisms. Nevertheless, more speciation research is necessary to evaluate the importance of organic and inorganic ligands in lowering the lethal and sublethal effects of elevated metal concentrations, especially Hg, to the local communities around White Island.
Ecological responses
Phytoplankton and zooplankton
The phytoplankton community structure at all stations around Whakaari (including the control stations) was very similar, which is not surprising given the strong water mixing in the area. However, it was surprising to see higher absolute cell numbers and lower diversity of the phytoplankton community at C1, the control station further away from the island. The C1 station showed the lowest ammonia level and a moderate NOx concentration, which could indicate that there has been elevated phytoplankton growth in this region for a longer time, resulting in the higher cell numbers and lower nutrient concentrations observed. The difference in phytoplankton biomass and diversity between the control station and all stations around the island could be caused by differences in concentrations of potentially toxic trace metals such as Hg, Pb and Cd (Thomas and Seibert 1977; Thomas et al. 1980; Tortell and Price 1996). The much higher relative concentration of diatoms around the island compared with C1 could be explained by the high ratio of Mn : Cd, Mn : Cu and Mn : Zn in the water column because all four metals are taken up by the same membrane uptake system in diatoms and high Mn concentrations are known to block the biological uptake of toxic metals (Sunda et al. 1981; Sunda and Huntsman 1996, 1998a, 1998b, 1998c). Therefore, diatoms are able to tolerate higher concentrations of toxic metals released by volcanic material that also release high Mn concentrations compared with other phytoplankton groups (Hoffmann et al. 2012). In addition, diatoms and coccolithophores are known to have a high metal tolerance because of the production of metal-binding ligands (e.g. Lorenzo et al. 2007; Echeveste et al. 2018). This statement is supported by our data, showing a greater [L] at C3 relative to V1 (Table 4) accompanied by a higher abundance of these two species at C3 SW (Fig. 5).
The zooplankton community showed parallel patterns to the phytoplankton, with a lower abundance at White Island compared with C1 in the Bay of Plenty. Chavtur (1992) also identified abundant holoplanktonic and meroplanktonic species at White Island with overlapping composition to the ‘White Island Blitz’ sampling (e.g. Oikopleura sp., calanoid and cyclopoid copepods), but also noting lower densities compared with the Hauraki Gulf. In the present study, sampling at the vent and control sites focused on the water column, and thus future sampling could also include waters in the immediate vicinity of the vent outlets, where high numbers of harpacticoid copepods are found (Tarasov et al. 1986).
Macroalgae
The natural history of Whakaari–White Island is broadly typical of subtidal temperate rocky coasts of Australia (Connell and Irving 2008) and northern New Zealand (Shears and Babcock 2007). It comprises mosaics of kelp (E. radiata), infrequent and small patches of turf-forming macroalgae (<10 cm in height) and urchin barrens (i.e. hard substratum sea urchin barrens devoid of vegetation). Turf-forming algae at the vents appear to be more productive and have a greater biomass at vents (Connell et al. 2017), which is comparable to algae raised under similar conditions in controlled laboratory and mesocosm experiments (Connell and Russell 2010; Connell et al. 2013). Such concurrence between experimental field and laboratory observations across a wide variety of conditions provides the kind of investigative approaches needed to illustrate CO2 as a driver of ecological responses in nature (Connell et al. 2013). The most conspicuous herbivores were damselfishes (Parma alboscapularis) and sparse densities of urchins common to Australia (Centrostephanus rodgersii) and moderate densities of urchins common to New Zealand (Evechinus chloroticus; Connell et al. 2018; Ferreira et al. 2018). Where a species has a positive association with turfs, its density can be substantially greater near vents (Connell et al. 2017). It is worth noting that the differences between these biological studies and the present study may be due to the fact that the studies were conducted in different locations, with the biological studies done at greater distances from vents (~25 m) than the sampling undertaken in the present study (i.e. within the vent fluids and 2 m from the vents).
Coralline algae
Evidence supports the hypothesis that those coralline algae that are capable of regulating pH at the site of calcification in their internal calcifying fluid (pHcf) under OA demonstrate more robust responses to lower pH conditions (Cornwall et al. 2017b). The articulate coralline alga C. officinalis maintained mean (±s.d.) pHcf of 8.66 ± 0.04 at the venting site and 8.75 ± 0.07 at the control site. Although pHcf is affected by external seawater pH, it is still elevated at the venting location relative to the surrounding seawater. The capacity of C. officinalis to maintain elevated pH in the calcifying fluid under OA could explain why this species of coralline algae is still in such high abundance near the high-CO2 locations at Whakaari. This mechanism of resistance could enable some species of coralline algae to persist in the future under OA, despite past conclusions that they are, in general, among the most susceptible taxa to OA (Fabricius et al. 2011).
Articulate and crustose coralline algae were present in seemingly high abundances at Whakaari, both near active venting sites and at control sites. This is in contrast with other vents sites studied off Vulcano (Italy; Cornwall et al. 2017a), Ischia Island (Italy; Hall-Spencer et al. 2008) and Milne Bay (Papua New Guinea; Fabricius et al. 2015), where coralline algae are nearly absent. Other coralline algae from New Zealand have shown similar robust responses to OA (Cornwall et al. 2013, 2014; James et al. 2014) compared with large declines in calcification observed in other coralline algal species (Anthony et al. 2008; Fabricius et al. 2015). The high abundance of coralline algae around the vents at Whakaari could include the fact that mean pH is higher or variability in pH lower at the study site relative to other shallow CO2 vent sites in the Mediterranean (Hall-Spencer et al. 2008; Cornwall et al. 2017a) and Papua New Guinea (Fabricius et al. 2015), which would elicit different responses. Some coralline algae are particularly susceptible to both increased variability in pH and abrupt changes in pH (Cornwall et al. 2013; Kamenos et al. 2013; Roleda et al. 2015). However, pHcf usually responds to changes in seawater mean pH rather than pH variability (Cornwall et al. 2018). This is most likely also occurring at White Island. Further research is required to understand the comparatively high coralline algal abundance here compared with other CO2 seep locations.
Conclusions
Findings of the ‘White Island Blitz’ suggest that the CO2 system of Whakaari–White Island represents one of many globally distributed vent systems that require care in their use as modern models of the ‘future ocean’. Although they create a pH and carbonate chemistry environment suitable for OA research (e.g. for predictions of 2100 and beyond), they also have several caveats, some of which have the potential to co-vary with scales of biological sampling, although this was not observed in this study. First, seawater carbonate chemistry was altered in only quite small areas around the vents, preventing a study of potential responses of pelagic and wide-ranging mobile organisms. Second, as around most other natural vents, the carbonate chemistry showed substantial variability, with unknown physiological and ecological consequences. Third, HgT concentrations at both vent and control sites reached concentrations that are considered biologically toxic (Australian and New Zealand Environment and Conservation Council 2000). Finally, even though concentrations of most elements and metals around the study site were within acceptable ranges, some (Mn, Fe, Co, Ni, Cu, Zn, Cd, Pb and sulfide) were elevated at both the vent and control sites, potentially affecting the metabolic rates of sensitive taxa.
Our results provide particular warning for the ability of metals and elements to confound studies of CO2 research at this hydrothermal vent system. The inconsistent variation of elements and dissolved metals in the working area across such small spatial scales suggests that these non-CO2 effects need to be measured and understood, along with CO2 concentrations, to improve our ability to reliably predict and manage for further ocean changes. It is possible that the ecology of Whakaari is different from assemblages elsewhere because of the general and variable chemical environment that envelops the broader coast of the Bay of Plenty. Therefore, the reporting of biological responses needs greater attention to potential prevalent multiple environmental stressors.
Our initial interdisciplinary results highlight that considerably more work is needed to better understand the biological responses to and ecosystem-scale effects of the environmental factors around White Island and vent systems worldwide. To tackle the lack of information and to better quantify the suitability of the temperate CO2 vents at White Island, or any other marine CO2 vent system, as a climate change analogue, future studies are encouraged to include the following steps and practices:
-
map the spatial and temporal changes in pH and other parameters of seawater carbonate chemistry during calm and well-mixed periods
-
consider the location of suitable control sites outside the influence of the hydrothermal signatures of the numerous CO2 vents at the study site to provide a regional context for biotic and abiotic interpretations
-
make detailed a priori investigations of the temporal and spatial trace element distribution and speciation of bioactive trace metals (e.g. Cu, Hg, Cd and Pb) at both vent and control sites
-
characterise the effects of OA on metal speciation, bioavailability and toxicity (Future decreases in OH– and CO32– concentrations will directly affect the solubility, distribution and speciation of metals in natural waters (Millero et al. 2009). Trace metals that form inorganic complexes with OH– and CO32– will undergo significant changes in speciation as the pH of seawater decreases, resulting in elevated levels of the bioavailable free ionic form of these metals (e.g. Millero et al. 2009; Ivanina and Sokolova 2015; Stockdale et al. 2016). Although some OA-induced changes in metal speciation will be beneficial (e.g. in the open ocean and in deep waters where productivity is limited by the low availability of micronutrients), others will exacerbate the harmful effects of bioactive metals to marine organisms (Ivanina and Sokolova 2015). Lower pH will also affect the adsorption of metals to organic ligands (Millero et al. 2009; Stockdale et al. 2016). However, the effect of pH on organic metal complexation in the marine environment is so far not well characterised and future studies are needed to examine the effect of OA on organic metal complexation and associated effects on marine organisms (e.g. Millero et al. 2009; Ivanina and Sokolova 2015; Stockdale et al. 2016).)
-
understand the interaction between CO2 and heavy metal concentrations, especially Hg, on biological responses (Based on published research, OA can change the toxicity, accumulation and intracellular binding processes of metals in several marine organisms, thereby affecting key physiological functions, including acid–base regulation, protein synthesis and energy homeostasis (e.g. Ivanina and Sokolova 2015; Zeng et al. 2015). Nevertheless, because of the scarcity of published research on OA–metal interactions, it is difficult to assess the degree to which both stressors are likely to affect the survival, performance and fitness-related functions (e.g. reproduction and growth) of marine organisms in the future ocean (Ivanina and Sokolova 2015).)
-
improve understanding of biological responses to sulfide, particularly at the scales of biological observation and experimentation
-
distinguish and quantify the individual and interactive effects of multiple stressors in the field and in the laboratory (e.g. laboratory-based experimental studies could be used to tease apart the effects of the increased CO2 and Hg on vent biota)
-
improve understanding regarding the biological and ecosystem-scale effects of the environmental variability around the vent sites
-
perform detailed laboratory investigations of the effects of CO2 on target taxa so that variation in response among taxa at vents can be better interpreted; calcareous organisms, such as mussels and coralline algae, may be investigated for their capacity to integrate CO2 exposure over time in their skeletons
-
focus on benthic sessile organisms because these communities integrate environmental conditions around the vent sites and thus provide a good proxy for the investigation of ecosystem processes (e.g. Pichler et al. 2019)
-
compare broad-scale and long-term OA and climate change studies in the field with laboratory-based experiments to validate findings and associated conclusions (González-Delgado and Hernández 2018)
Although research of CO2 vents was intended to improve the realism of OA research, which relies heavily on laboratory and mesocosm experiments, we acknowledge that CO2 vents are not perfect analogues for forecasts of OA, and Whakaari–White Island is no exception. Indeed, there are few study sites or methods that can be relied upon to progress an entire discipline. Multiple approaches are needed, and each method needs constant reflection and refinement as to whether they establish the cause (abiotic change) and effect (biological response) for the chemical parameters of concern. Do ecological studies of volcanic vents measure the consequences of CO2 enrichment? The ‘White Island Blitz’ shows that care is needed in the use of this particular multivariate site in New Zealand as a natural laboratory for studying temperate communities in future pH projections. The ‘White Island Blitz’ points to the need for validation by intensive laboratory and field investigations. Although laboratory investigations (e.g. mesocosms) tend to oversimplify and exaggerate their measured effects of OA and obscure important natural processes (Goldenberg et al. 2018), they provide insight into the types of responses and their mechanisms and causes. Although field investigations of naturally complex ecological systems incorporate a multitude of drivers, and thus reflect a more environmentally realistic scenario than laboratory-based experiments, they make any one driver or even multiple stressors challenging to isolate (Sommer 2012; Ivanina and Sokolova 2015; Boyd et al. 2018). If research into OA is not to stagnate, we need to understand biological or ecological responses to OA, as well as the sensitivity and legitimacy of our tests of OA.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Declaration of funding
The ‘White Island Blitz’ was funded primarily by the University of Otago Ocean Acidification Research Theme, and the authors thank the Otago Research Committee for their support. Rebecca Zitoun received a doctoral scholarship from the University of Otago. The deployment of the SeaFETs in November 2015 was funded by the University of Auckland in a grant to Nick Shears. The International Atomic Energy Agency is grateful to the Government of the Principality of Monaco for the support provided to its Environment Laboratories.
Acknowledgements
This paper is dedicated to Keith Hunter, who sadly passed away on 24 October 2018. New Zealand, and the world, has lost, much too early, a great marine and analytical chemist with a true view for the big picture. Keith was able to not only comprehend the link between all facets of the marine system, but was also able to model it and translate it into user-friendly software applications. Keith Hunter has been extremely influential in the initiation of many researchers’ careers, including scientific pathways of some of the authors of this paper. His leadership and mentorship is greatly missed by all who knew him. In each of the papers published in this special issue, a part of him will live on because he helped lay the foundations of trace element and OA research in New Zealand, hosted early meetings at the University of Otago and was instrumental in getting the University of Otago Ocean Acidification Research Theme going. It was that theme that allowed the ‘White Island Blitz’ to happen. The authors thank Laura Miscoe for help with microscopic phytoplankton identification and Malcolm T. McCulloch for providing materials for the δ11B analysis of the coralline algae. The authors also thank Carl Lamborg from the University of California (Santa Cruz, CA, USA) for quantification of total mercury, and Andrea Koschinsky and Charlotte Kleint from Jacobs University (Bremen, Germany) for trace metal quantification of Li, Rb, Sr, Mo, Cs, Ba and U. The authors are also grateful to Claudine Stirling, Malcolm Reid and Kyyas Seyitmuhammedov from the University of Otago (Dunedin, New Zealand), and Rob Middag from the Royal Netherlands Institute for Sea Research (Texel, Netherlands) for providing access to unpublished trace metal data from the TAN1604-CS19 sample.
References
Agostini, S., Wada, S., Kon, K., Omori, A., Kohtsuka, H., Fujimura, H., Tsuchiya, Y., Sato, T., Shinagawa, H., Yamada, Y., and Inaba, K. (2015). Geochemistry of two shallow CO2 seeps in Shikine Island (Japan) and their potential for ocean acidification research. Regional Studies in Marine Science 2, 45–53.| Geochemistry of two shallow CO2 seeps in Shikine Island (Japan) and their potential for ocean acidification research.Crossref | GoogleScholarGoogle Scholar |
Al-Farawati, R., and van den Berg, C. M. G. (1997). The determination of sulfide in seawater by flow-analysis with voltammetric detection. Marine Chemistry 57, 277–286.
| The determination of sulfide in seawater by flow-analysis with voltammetric detection.Crossref | GoogleScholarGoogle Scholar |
Amin, S. A., Moffett, J. W., Martens-Habbena, W., Jacquot, J. E., Han, Y., Devol, A., Ingalls, A. E., Stahl, D. A., and Armbrust, E. (2013). Copper requirements of the ammonia-oxidizing archaeon Nitrosopumilus maritimus SCM1 and implications for nitrification in the marine environment. Limnology and Oceanography 58, 2037–2045.
| Copper requirements of the ammonia-oxidizing archaeon Nitrosopumilus maritimus SCM1 and implications for nitrification in the marine environment.Crossref | GoogleScholarGoogle Scholar |
Annett, A. L., Lapi, S., Ruth, T. J., and Maldonado, M. T. (2008). The effects of Cu and Fe availability on the growth and Cu : C ratios of marine diatoms. Limnology and Oceanography 53, 2451–2461.
| The effects of Cu and Fe availability on the growth and Cu : C ratios of marine diatoms.Crossref | GoogleScholarGoogle Scholar |
Anthony, K. R. N., Kline, D. I., Diaz-Pulido, G., Dove, S., and Hoegh-Guldberg, O. (2008). Ocean acidification causes bleaching and productivity loss in coral reef builders. Proceedings of the National Academy of Sciences of the United States of America 105, 17442–17446.
| Ocean acidification causes bleaching and productivity loss in coral reef builders.Crossref | GoogleScholarGoogle Scholar |
Australian and New Zealand Environment and Conservation Council (1992). ‘Australian Water Quality Guidelines for Fresh and Marine Waters. National Water Quality Management Strategy.’ (ANZECC and Agriculture and Resource Management Council of Australia and New Zealand: Canberra, ACT, Australia.)
Australian and New Zealand Environment and Conservation Council (2000). ‘Australian and New Zealand Guidelines for Fresh and Marine Water Quality.’ (ANZECC and Agriculture and Resource Management Council of Australia and New Zealand: Canberra, ACT, Australia.)
Baggini, C., Salomidi, M., Voutsinas, E., Bray, L., Krasakopoulou, E., and Hall-Spencer, J. M. (2014). Seasonality affects macroalgal community response to increases in pCO2. PLoS One 9, e106520.
| Seasonality affects macroalgal community response to increases in pCO2.Crossref | GoogleScholarGoogle Scholar | 25184242PubMed |
Balcom, P. H., Fitzgerald, W. F., Vandal, G. M., Lamborg, C. H., Rolfhus, K. R., Langer, C. S., and Hammerschmidt, C. R. (2004). Mercury sources and cycling in the Connecticut River and Long Island Sound. Marine Chemistry 90, 53–74.
| Mercury sources and cycling in the Connecticut River and Long Island Sound.Crossref | GoogleScholarGoogle Scholar |
Bi, Z. S., Salaun, P., and van den Berg, C. M. G. (2013). Study of bare and mercury-coated vibrated carbon, gold and silver microwire electrodes for the determination of lead and cadmium in seawater by anodic stripping voltammetry. Electroanalysis 25, 357–366.
| Study of bare and mercury-coated vibrated carbon, gold and silver microwire electrodes for the determination of lead and cadmium in seawater by anodic stripping voltammetry.Crossref | GoogleScholarGoogle Scholar |
Biller, D. V., and Bruland, K. W. (2012). Analysis of Mn, Fe, Co, Ni, Cu, Zn, Cd, and Pb in seawater using the Nobias-chelate PA1 resin and magnetic sector inductively coupled plasma mass spectrometry (ICP-MS). Marine Chemistry 130–131, 12–20.
| Analysis of Mn, Fe, Co, Ni, Cu, Zn, Cd, and Pb in seawater using the Nobias-chelate PA1 resin and magnetic sector inductively coupled plasma mass spectrometry (ICP-MS).Crossref | GoogleScholarGoogle Scholar |
Bloom, N. S., and Crecelius, E. A. (1983). Determination of mercury in seawater at sub-nanogram per liter levels. Marine Chemistry 14, 49–59.
| Determination of mercury in seawater at sub-nanogram per liter levels.Crossref | GoogleScholarGoogle Scholar |
Boatta, F., D’Alessandro, W., Gagliano, A. L., Liotta, M., Milazzo, M., Rodolfo-Metalpa, R., Hall-Spencer, J. M., and Parello, F. (2013). Geochemical survey of Levante Bay, Vulcano Island (Italy), a natural laboratory for the study of ocean acidification. Marine Pollution Bulletin 73, 485–494.
| Geochemical survey of Levante Bay, Vulcano Island (Italy), a natural laboratory for the study of ocean acidification.Crossref | GoogleScholarGoogle Scholar | 23465567PubMed |
Bown, J., Laan, P., Ossebaar, S., Bakker, K., Rozema, P., and de Baar, H. J. (2017). Bioactive trace metal time series during Austral summer in Ryder Bay, Western Antarctic Peninsula. Deep-sea Research – II. Topical Studies in Oceanography 139, 103–119.
| Bioactive trace metal time series during Austral summer in Ryder Bay, Western Antarctic Peninsula.Crossref | GoogleScholarGoogle Scholar |
Boyd, R. S. (2010). Heavy metal pollutants and chemical ecology: exploring new frontiers. Journal of Chemical Ecology 36, 46–58.
| Heavy metal pollutants and chemical ecology: exploring new frontiers.Crossref | GoogleScholarGoogle Scholar | 20108028PubMed |
Boyd, C. E. (2014). Hydrogen sulfide, toxic but manageable. In Global Aquaculture Advocate, pp. 34–36. Available at https://aquafishcrsp.oregonstate.edu/sites/aquafishcrsp.oregonstate.edu/files/boyd2014hydrogensulfide_gaa.pdf [Verified 10 October 2019].
Boyd, P. W., Collins, S., Dupont, S., Fabricius, K., Gattuso, J. P., Havenhand, J., Hutchins, D. A., Riebesell, U., Rintoul, M. S., Vichi, M., and Biswas, H. (2018). Experimental strategies to assess the biological ramifications of multiple drivers of global ocean change – a review. Global Change Biology 24, 2239–2261.
| Experimental strategies to assess the biological ramifications of multiple drivers of global ocean change – a review.Crossref | GoogleScholarGoogle Scholar | 29476630PubMed |
Brand, L. E., Sunda, W. G., and Guillard, R. R. (1986). Reduction of marine phytoplankton reproduction rates by copper and cadmium. Journal of Experimental Marine Biology and Ecology 96, 225–250.
| Reduction of marine phytoplankton reproduction rates by copper and cadmium.Crossref | GoogleScholarGoogle Scholar |
Brinkman, T. J., and Smith, A. M. (2015). Effect of climate change on crustose coralline algae at a temperate vent site, White Island, New Zealand. Marine and Freshwater Research 66, 360–370.
| Effect of climate change on crustose coralline algae at a temperate vent site, White Island, New Zealand.Crossref | GoogleScholarGoogle Scholar |
Bruland, K. W., Donat, J. R., and Hutchins, D. A. (1991). Interactive influences of bioactive trace metals on biological production in oceanic waters. Limnology and Oceanography 36, 1555–1577.
| Interactive influences of bioactive trace metals on biological production in oceanic waters.Crossref | GoogleScholarGoogle Scholar |
Bruland, K. W., Middag, R., and Lohan, M. C. (2014). Controls of trace metals in seawater. In ‘Treatise on Geochemistry’, 2nd edn. (Eds M. J. Mottl and H. Elderfield.) pp. 19–51. (Saunders, Elsevier: Philadelphia, PA, USA.)
Bundy, R. M., Abdulla, H. A., Hatcher, P. G., Biller, D. V., Buck, K. N., and Barbeau, K. A. (2015). Iron-binding ligands and humic substances in the San Francisco Bay estuary and estuarine-influenced shelf regions of coastal California. Marine Chemistry 173, 183–194.
| Iron-binding ligands and humic substances in the San Francisco Bay estuary and estuarine-influenced shelf regions of coastal California.Crossref | GoogleScholarGoogle Scholar |
Burger, A., and Lichtscheidl, I. (2019). Strontium in the environment: review about reactions of plants towards stable and radioactive strontium isotopes. The Science of the Total Environment 653, 1458–1512.
| Strontium in the environment: review about reactions of plants towards stable and radioactive strontium isotopes.Crossref | GoogleScholarGoogle Scholar | 30759584PubMed |
Caldeira, K., and Wickett, M. E. (2003). Oceanography: anthropogenic carbon and ocean pH. Nature 425, 365.
| Oceanography: anthropogenic carbon and ocean pH.Crossref | GoogleScholarGoogle Scholar | 14508477PubMed |
Calosi, P., Rastrick, S. P. S., Lombardi, C., de Guzman, H. J., Davidson, L., Jahnke, M., Giangrande, A., Hardege, J. D., Schulze, A., Spicer, J. I., and Gambi, M.-C. (2013). Adaptation and acclimatization to ocean acidification in marine ectotherms: an in situ transplant experiment with polychaetes at a shallow CO2 vent system. Philosophical Transactions of the Royal Society of London – B. Biological Sciences 368, 20120444.
| Adaptation and acclimatization to ocean acidification in marine ectotherms: an in situ transplant experiment with polychaetes at a shallow CO2 vent system.Crossref | GoogleScholarGoogle Scholar | 23980247PubMed |
Chavtur, V. G. (1992). Specifics of structure and distribution of zooplankton in condition of underwater volcanism at the shelf and at the upper divisions of continental slope of the West Pacific. Deposition to VINITI 22.09.92, Manuscript N615–B, 92, Vladivostok, Russia. [In Russian].
Chen, X. G., Lyu, S. S., Garbe-Schönberg, D., Lebrato, M., Li, X., Zhang, H. Y., Zhang, P. P., Chen, C. T. A., and Ye, Y. (2018). Heavy metals from Kueishantao shallow-sea hydrothermal vents, offshore northeast Taiwan. Journal of Marine Systems 180, 211–219.
| Heavy metals from Kueishantao shallow-sea hydrothermal vents, offshore northeast Taiwan.Crossref | GoogleScholarGoogle Scholar |
Chiodini, G., Caliro, S., Caramanna, G., Granieri, D., Minopoli, C., Moretti, R., Perotta, L., and Ventura, G. (2006). Geochemistry of the submarine gaseous emissions of Panarea (Aeolian Islands, Southern Italy): magmatic vs. hydrothermal origin and implications for volcanic surveillance. Pure and Applied Geophysics 163, 759–780.
| Geochemistry of the submarine gaseous emissions of Panarea (Aeolian Islands, Southern Italy): magmatic vs. hydrothermal origin and implications for volcanic surveillance.Crossref | GoogleScholarGoogle Scholar |
Clarkson, B. D., and Clarkson, B. R. (1994). Vegetation decline following recent eruptions on White Island (Whakaari), Bay of Plenty, New Zealand. New Zealand Journal of Botany 32, 21–36.
| Vegetation decline following recent eruptions on White Island (Whakaari), Bay of Plenty, New Zealand.Crossref | GoogleScholarGoogle Scholar |
Cole, J. W., Thordarson, T., and Burt, R. M. (2000). Magma origin and evolution of White Island (Whakaari) volcano, Bay of Plenty, New Zealand. Journal of Petrology 41, 867–895.
| Magma origin and evolution of White Island (Whakaari) volcano, Bay of Plenty, New Zealand.Crossref | GoogleScholarGoogle Scholar |
Comeau, S., Cornwall, C. E., DeCarlo, T. M., Krieger, E., and McCulloch, M. T. (2018). Similar controls on calcification under ocean acidification across unrelated coral reef taxa. Global Change Biology 24, 4857–4868.
| Similar controls on calcification under ocean acidification across unrelated coral reef taxa.Crossref | GoogleScholarGoogle Scholar | 29957854PubMed |
Connell, S. D., and Irving, A. D. (2008). Integrating ecology with biogeography using landscape characteristics: a case study of subtidal habitat across continental Australia. Journal of Biogeography 35, 1608–1621.
| Integrating ecology with biogeography using landscape characteristics: a case study of subtidal habitat across continental Australia.Crossref | GoogleScholarGoogle Scholar |
Connell, S. D., and Russell, B. D. (2010). The direct effects of increasing CO2 and temperature on non-calcifying organisms: increasing the potential for phase shifts in kelp forests. Proceedings of the Royal Society of London – B. Biological Sciences 277, 1409–1415.
| The direct effects of increasing CO2 and temperature on non-calcifying organisms: increasing the potential for phase shifts in kelp forests.Crossref | GoogleScholarGoogle Scholar |
Connell, S. D., Kroeker, K. J., Fabricius, K. E., Kline, D. I., and Russell, B. D. (2013). The other ocean acidification problem: CO2 as a resource among competitors for ecosystem dominance. Philosophical Transactions of the Royal Society of London – B. Biological Sciences 368, 20120442.
| The other ocean acidification problem: CO2 as a resource among competitors for ecosystem dominance.Crossref | GoogleScholarGoogle Scholar | 23980244PubMed |
Connell, S. D., Doubleday, Z. A., Hamlyn, S. B., Foster, N. R., Harley, C. D. G., Kelaher, B. P., Nagelkerken, I., Sara, G., and Russell, B. D. (2017). How ocean acidification can benefit calcifiers. Current Biology 27, R95–R96.
| How ocean acidification can benefit calcifiers.Crossref | GoogleScholarGoogle Scholar | 28171763PubMed |
Connell, S. D., Doubleday, Z. A., Foster, N. R., Hamlyn, S. B., Harley, C. D., Helmuth, B., Kelaher, B. P., Nagelkerken, I., Rodgers, K. L., Sarà, G., and Russell, B. D. (2018). The duality of ocean acidification as a resource and a stressor. Ecology 99, 1005–1010.
| The duality of ocean acidification as a resource and a stressor.Crossref | GoogleScholarGoogle Scholar | 29714829PubMed |
Cornwall, C. E., Hepburn, C. D., McGraw, C. M., Currie, K. I., Pilditch, C. A., Hunter, K. A., Boyd, P. W., and Hurd, C. L. (2013). Diurnal fluctuations in seawater pH influence the response of a calcifying macroalga to ocean acidification. Proceedings of the Royal Society of London – B. Biological Sciences 280, 20132201.
| Diurnal fluctuations in seawater pH influence the response of a calcifying macroalga to ocean acidification.Crossref | GoogleScholarGoogle Scholar |
Cornwall, C. E., Boyd, P. W., McGraw, C. M., Hepburn, C. D., Pilditch, C. A., Morris, J. N., Smith, A. M., and Hurd, C. L. (2014). Diffusion boundary layers ameliorate the negative effects of ocean acidification on the temperate coralline macroalga Arthrocardia corymbosa. PLoS One 9, e97235.
| Diffusion boundary layers ameliorate the negative effects of ocean acidification on the temperate coralline macroalga Arthrocardia corymbosa.Crossref | GoogleScholarGoogle Scholar | 24824089PubMed |
Cornwall, C. E., Revill, A. T., Hall-Spencer, J. M., Milazzo, M., Raven, J. A., and Hurd, C. L. (2017a). Inorganic carbon physiology underpins macroalgal responses to elevated CO2. Scientific Reports 7, 46297.
| Inorganic carbon physiology underpins macroalgal responses to elevated CO2.Crossref | GoogleScholarGoogle Scholar | 28417970PubMed |
Cornwall, C. E., Comeau, S., and McCulloch, M. T. (2017b). Coralline algae elevate pH at the site of calcification under ocean acidification. Global Change Biology 23, 4245–4256.
| Coralline algae elevate pH at the site of calcification under ocean acidification.Crossref | GoogleScholarGoogle Scholar | 28370806PubMed |
Cornwall, C. E., Comeau, S., DeCarlo, T. M., Moore, B., D’Alexis, Q., and McCulloch, M. T. (2018). Resistance of corals and coralline algae to ocean acidification: physiological control of calcification under natural pH variability. Proceedings of the Royal Society of London – B. Biological Sciences 285, 20181168.
| Resistance of corals and coralline algae to ocean acidification: physiological control of calcification under natural pH variability.Crossref | GoogleScholarGoogle Scholar |
Cotte, L., Omanović, D., Laës, A., Cathalot, C., Sarradin, P.-M., and Riso, R. (2018). On the nature of dissolved copper ligands in the early buoyant plume of hydrothermal vents. Environmental Chemistry 15, 58–73.
| On the nature of dissolved copper ligands in the early buoyant plume of hydrothermal vents.Crossref | GoogleScholarGoogle Scholar |
Cunha, L., Amaral, A., Medeiros, V., Martins, G. M., Wallenstein, F. F., Couto, R. P., Neto, A. I., and Rodrigues, A. (2008). Bioavailable metals and cellular effects in the digestive gland of marine limpets living close to shallow water hydrothermal vents. Chemosphere 71, 1356–1362.
| Bioavailable metals and cellular effects in the digestive gland of marine limpets living close to shallow water hydrothermal vents.Crossref | GoogleScholarGoogle Scholar | 18166212PubMed |
Dahms, H. U., Schizas, N. V., James, R. A., Wang, L., and Hwang, J. S. (2018). Marine hydrothermal vents as templates for global change scenarios. Hydrobiologia 818, 1–10.
| Marine hydrothermal vents as templates for global change scenarios.Crossref | GoogleScholarGoogle Scholar |
Dickson, A. G., Sabine, C. L., and Christian, J. R. (Eds) (2007). Guide to best practices for ocean CO2 measurements. PICES Special Publication 3, IOCCP Report number 8, North Pacific Marine Science Organization, Sidney, BC, Canada.
Donachie, S. P., Christenson, B. W., Kunkel, D. D., Malahoff, A., and Alam, M. (2002). Microbial community in acidic hydrothermal waters of volcanically active White Island, New Zealand. Extremophiles 6, 419–425.
| Microbial community in acidic hydrothermal waters of volcanically active White Island, New Zealand.Crossref | GoogleScholarGoogle Scholar | 12382119PubMed |
Echeveste, P., Croot, P., and von Dassow, P. (2018). Differences in the sensitivity to Cu and ligand production of coastal vs offshore strains of Emiliania huxleyi. The Science of the Total Environment 625, 1673–1680.
| Differences in the sensitivity to Cu and ligand production of coastal vs offshore strains of Emiliania huxleyi.Crossref | GoogleScholarGoogle Scholar | 29056389PubMed |
Enochs, I. C., Manzello, D. P., Donham, E. M., Kolodziej, G., Okano, R., Johnston, L., Young, C., Iguel, J., Edwards, C. B., Fox, M. D., and Valentino, L. (2015). Shift from coral to macroalgae dominance on a volcanically acidified reef. Nature Climate Change 5, 1083–1088.
| Shift from coral to macroalgae dominance on a volcanically acidified reef.Crossref | GoogleScholarGoogle Scholar |
Fabricius, K. E., Langdon, C., Uthicke, S., Humphrey, C., Noonan, S., De’ath, G., Okazaki, R., Muehllehner, N., Glas, M. S., and Lough, J. M. (2011). Losers and winners in coral reefs acclimatized to elevated carbon dioxide concentrations. Nature Climate Change 1, 165–169.
| Losers and winners in coral reefs acclimatized to elevated carbon dioxide concentrations.Crossref | GoogleScholarGoogle Scholar |
Fabricius, K. E., De’ath, G., Noonan, S., and Uthicke, S. (2014). Ecological effects of ocean acidification and habitat complexity on reef-associated macro-invertebrate communities. Proceedings of the Royal Society of London – B. Biological Sciences 281, 20132479.
| Ecological effects of ocean acidification and habitat complexity on reef-associated macro-invertebrate communities.Crossref | GoogleScholarGoogle Scholar |
Fabricius, K. E., Kluibenschedl, A., Harrington, L., Noonan, S., and De’Ath, G. (2015). In situ changes of tropical crustose coralline algae along carbon dioxide gradients. Scientific Reports 5, 9537.
| In situ changes of tropical crustose coralline algae along carbon dioxide gradients.Crossref | GoogleScholarGoogle Scholar | 25835382PubMed |
Fabry, V. J., Seibel, B. A., Feely, R. A., and Orr, J. C. (2008). Impacts of ocean acidification on marine fauna and ecosystem processes. ICES Journal of Marine Science 65, 414–432.
| Impacts of ocean acidification on marine fauna and ecosystem processes.Crossref | GoogleScholarGoogle Scholar |
Ferreira, C. M., Nagelkerken, I., Goldenberg, S. U., and Connell, S. D. (2018). CO2 emissions boost the benefits of crop production by farming damselfish. Nature Ecology & Evolution 2, 1223–1226.
| CO2 emissions boost the benefits of crop production by farming damselfish.Crossref | GoogleScholarGoogle Scholar |
Gattuso, J. P., and Lavigne, H. (2009). Approaches and software tools to investigate the impact of ocean acidification. Biogeosciences 6, 2121–2133.
| Approaches and software tools to investigate the impact of ocean acidification.Crossref | GoogleScholarGoogle Scholar |
Gattuso, J. P., Magnan, A., Billé, R., Cheung, W. W., Howes, E. L., Joos, F., Allemand, D., Bopp, L., Cooley, S. R., Eakin, C. M., and Hoegh-Guldberg, O. (2015). Contrasting futures for ocean and society from different anthropogenic CO2 emissions scenarios. Science 349, aac4722.
| Contrasting futures for ocean and society from different anthropogenic CO2 emissions scenarios.Crossref | GoogleScholarGoogle Scholar | 26138982PubMed |
German, C. R., and Von Damm, K. L. (2003). Hydrothermal processes. Treatise on Geochemistry 6, 181–222.
| Hydrothermal processes.Crossref | GoogleScholarGoogle Scholar |
Giggenbach, W. F. (1975). Variations in the carbon, sulfur and chlorine contents of volcanic gas discharges from White Island, New Zealand. Bulletin Volcanologique 39, 15–27.
| Variations in the carbon, sulfur and chlorine contents of volcanic gas discharges from White Island, New Zealand.Crossref | GoogleScholarGoogle Scholar |
Giggenbach, W. F. (1987). Redox processes governing the chemistry of fumarolic gas discharges from White Island, New Zealand. Applied Geochemistry 2, 143–161.
| Redox processes governing the chemistry of fumarolic gas discharges from White Island, New Zealand.Crossref | GoogleScholarGoogle Scholar |
Gledhill, M., Nimmo, M., Hill, S. J., and Brown, M. T. (1999). The release of copper-complexing ligands by the brown alga Fucus vesiculosus (Phaeophyceae) in response to increasing total copper levels. Journal of Phycology 35, 501–509.
| The release of copper-complexing ligands by the brown alga Fucus vesiculosus (Phaeophyceae) in response to increasing total copper levels.Crossref | GoogleScholarGoogle Scholar |
Goldenberg, S. U., Nagelkerken, I., Marangon, E., Bonnet, A., Ferreira, C. M., and Connell, S. D. (2018). Ecological complexity buffers the impacts of future climate on marine consumers. Nature Climate Change 8, 229–233.
| Ecological complexity buffers the impacts of future climate on marine consumers.Crossref | GoogleScholarGoogle Scholar |
González-Delgado, S., and Hernández, J. C. (2018). The importance of natural acidified systems in the study of ocean acidification: what have we learned? Advances in Marine Biology 80, 57–99.
| The importance of natural acidified systems in the study of ocean acidification: what have we learned?Crossref | GoogleScholarGoogle Scholar | 30368306PubMed |
Grace, R. V. (1975). White Island notes. Tane 21, 91–100.
Grange, K. R., Murdoch, R. C., Battershill, C. N., and Singleton, R. J. (1992). A survey of the marine habitats and communities of White Island, Bay of Plenty. Department of Conservation Investigation number P977, New Zealand Oceanographic Institute DSIR 1992/8, Wellington, New Zealand.
Gworek, B., Bemowska-Kałabun, O., Kijeńska, M., and Wrzosek-Jakubowska, J. (2016). Mercury in marine and oceanic waters – a review. Water, Air, and Soil Pollution 227, 371.
| Mercury in marine and oceanic waters – a review.Crossref | GoogleScholarGoogle Scholar | 27656005PubMed |
Hall-Spencer, J. M., Rodolfo-Metalpa, R., Martin, S., Ransome, E., Fine, M., Turner, S. M., Rowley, S. J., Tedesco, D., and Buia, M.-C. (2008). Volcanic carbon dioxide vents show ecosystem effects of ocean acidification. Nature 454, 96–99.
| Volcanic carbon dioxide vents show ecosystem effects of ocean acidification.Crossref | GoogleScholarGoogle Scholar | 18536730PubMed |
Hawkins, S. J., Evans, A. J., Dale, A. C., Firth, L. B., and Smith, I. P. (2018). The carbon dioxide vents of Ischtia, Italy. A natural system to assess impacts of ocean acidification on marine ecosystems: an overview of research and comparisons with other vent systems. Oceanography and Marine Biology – an Annual Review 56, 237–310.
Hernández, C. A., Sangil, C., and Hernández, J. C. (2016). A new CO2 vent for the study of ocean acidification in the Atlantic. Marine Pollution Bulletin 109, 419–426.
| A new CO2 vent for the study of ocean acidification in the Atlantic.Crossref | GoogleScholarGoogle Scholar | 27210563PubMed |
Hocking, M. W., Hannington, M. D., Percival, J. B., Stoffers, P., Schwarz-Schampera, U., and de Ronde, C. E. J. (2010). Clay alteration of volcaniclastic material in a submarine geothermal system, Bay of Plenty, New Zealand. Journal of Volcanology and Geothermal Research 191, 180–192.
| Clay alteration of volcaniclastic material in a submarine geothermal system, Bay of Plenty, New Zealand.Crossref | GoogleScholarGoogle Scholar |
Hoffmann, L. J., Breitbarth, E., Ardelan, M. V., Duggen, S., Olgun, N., Hassellöv, M., and Wängberg, S.-Å. (2012). Influence of trace metal release from volcanic ash on growth of Thalassiosira pseudonana and Emiliania huxleyi. Marine Chemistry 132–133, 28–33.
| Influence of trace metal release from volcanic ash on growth of Thalassiosira pseudonana and Emiliania huxleyi.Crossref | GoogleScholarGoogle Scholar |
Inoue, S., Kayanne, H., Yamamoto, S., and Kurihara, H. (2013). Spatial community shift from hard to soft corals in acidified water. Nature Climate Change 3, 683–687.
| Spatial community shift from hard to soft corals in acidified water.Crossref | GoogleScholarGoogle Scholar |
Ivanina, A. V., and Sokolova, I. M. (2015). Interactive effects of metal pollution and ocean acidification on physiology of marine organisms. Current Zoology 61, 653–668.
| Interactive effects of metal pollution and ocean acidification on physiology of marine organisms.Crossref | GoogleScholarGoogle Scholar |
Jacquot, J. E., Horak, R. E., Amin, S. A., Devol, A. H., Ingalls, A. E., Armbrust, E. V., Stahl, D. A., and Moffett, J. W. (2014). Assessment of the potential for copper limitation of ammonia oxidation by Archaea in a dynamic estuary. Marine Chemistry 162, 37–49.
| Assessment of the potential for copper limitation of ammonia oxidation by Archaea in a dynamic estuary.Crossref | GoogleScholarGoogle Scholar |
Jahn, A., and Theede, H. (1997). Different degrees of tolerance to hydrogen sulphide in populations of Macoma balthica (Bivalvia, Tellinidae). Marine Ecology Progress Series 154, 185–196.
| Different degrees of tolerance to hydrogen sulphide in populations of Macoma balthica (Bivalvia, Tellinidae).Crossref | GoogleScholarGoogle Scholar |
Jakimska, A., Konieczka, P., Skóra, K., and Namieśnik, J. (2011). Bioaccumulation of metals in tissues of marine animals, Part II: metal concentrations in animal tissues. Polish Journal of Environmental Studies 20, 1127–1146.
James, R. K., Hepburn, C. D., Cornwall, C. E., McGraw, C. M., and Hurd, C. L. (2014). Growth response of an early successional assemblage of coralline algae and benthic diatoms to ocean acidification. Marine Biology 161, 1687–1696.
| Growth response of an early successional assemblage of coralline algae and benthic diatoms to ocean acidification.Crossref | GoogleScholarGoogle Scholar |
Kamenos, N. A., Burdett, H. L., Aloisio, E., Findlay, H. S., Martin, S., Longbone, C., Dunn, J., Widdicombe, S., and Calosi, P. (2013). Coralline algal structure is more sensitive to rate, rather than the magnitude, of ocean acidification. Global Change Biology 19, 3621–3628.
| Coralline algal structure is more sensitive to rate, rather than the magnitude, of ocean acidification.Crossref | GoogleScholarGoogle Scholar | 23943376PubMed |
Karlson, B., Godhe, A., Cusack, C., and Bresnan, E. (2011). Introduction to methods for quantitative phytoplankton analysis. In ‘Microscopic and Molecular Methods for Quantitative Phytoplankton Analysis IOC Manuals and Guides’. (Eds B. Karlson, A. Godhe, C. Cusack, and E. Bresnan.) Number 55, pp. 5–12. (UNESCO: Paris, France.)
Karthick, P., Sankar, R. S., Kaviarasan, T., and Mohanraju, R. (2012). Ecological implications of trace metals in seaweeds: bioindication potential for metal contamination in Wandoor, South Andaman Island. The Egyptian Journal of Aquatic Research 38, 227–231.
| Ecological implications of trace metals in seaweeds: bioindication potential for metal contamination in Wandoor, South Andaman Island.Crossref | GoogleScholarGoogle Scholar |
Kerrison, P., Hall-Spencer, J. M., Suggett, D. J., Hepburn, L. J., and Steinke, M. (2011). Assessment of pH variability at a coastal CO2 vent for ocean acidification studies. Estuarine, Coastal and Shelf Science 94, 129–137.
| Assessment of pH variability at a coastal CO2 vent for ocean acidification studies.Crossref | GoogleScholarGoogle Scholar |
Kiaune, L., and Singhasemanon, N. (2011). Pesticidal copper (I) oxide: environmental fate and aquatic toxicity. Reviews of Environmental Contamination and Toxicology 213, 1–26.
| Pesticidal copper (I) oxide: environmental fate and aquatic toxicity.Crossref | GoogleScholarGoogle Scholar | 21541846PubMed |
Kleint, C., Kuzmanovski, S., Powell, Z., Bühring, S. I., Sander, S. G., and Koschinsky, A. (2015). Organic Cu-complexation at the shallow marine hydrothermal vent fields off the coast of Milos (Greece), Dominica (Lesser Antilles) and the Bay of Plenty (New Zealand). Marine Chemistry 173, 244–252.
| Organic Cu-complexation at the shallow marine hydrothermal vent fields off the coast of Milos (Greece), Dominica (Lesser Antilles) and the Bay of Plenty (New Zealand).Crossref | GoogleScholarGoogle Scholar |
Klevenz, V., Sander, S. G., Perner, M., and Koschinsky, A. (2012). Amelioration of free copper by hydrothermal vent microbes as a response to high copper concentrations. Chemistry and Ecology 28, 405–420.
| Amelioration of free copper by hydrothermal vent microbes as a response to high copper concentrations.Crossref | GoogleScholarGoogle Scholar |
Kogut, M. B., and Voelker, B. M. (2001). Strong copper-binding behaviour of terrestrial humic substances in seawater. Environmental Science & Technology 35, 1149–1156.
| Strong copper-binding behaviour of terrestrial humic substances in seawater.Crossref | GoogleScholarGoogle Scholar |
Kozelka, P. B., and Bruland, K. W. (1998). Chemical speciation of dissolved Cu, Zn, Cd, Pb in Narragansett Bay, Rhode Island. Marine Chemistry 60, 267–282.
| Chemical speciation of dissolved Cu, Zn, Cd, Pb in Narragansett Bay, Rhode Island.Crossref | GoogleScholarGoogle Scholar |
Kroeker, K. J., Micheli, F., Gambi, M. C., and Martz, T. R. (2011). Divergent ecosystem responses within a benthic marine community to ocean acidification. Proceedings of the National Academy of Sciences of the United States of America 108, 14515–14520.
| Divergent ecosystem responses within a benthic marine community to ocean acidification.Crossref | GoogleScholarGoogle Scholar | 21844331PubMed |
Laglera, L. M., and Van den Berg, C. M. (2009). Evidence for geochemical control of iron by humic substances in seawater. Limnology and Oceanography 54, 610–619.
| Evidence for geochemical control of iron by humic substances in seawater.Crossref | GoogleScholarGoogle Scholar |
Lamare, M. D., Liddy, M., and Uthicke, S. (2016). In situ developmental responses of tropical sea urchin larvae to ocean acidification conditions at naturally elevated pCO2 vent sites. Proceedings of the Royal Society of London – B. Biological Sciences 283, 20161506.
| In situ developmental responses of tropical sea urchin larvae to ocean acidification conditions at naturally elevated pCO2 vent sites.Crossref | GoogleScholarGoogle Scholar |
Lamborg, C. H., Tseng, C. M., Fitzgerald, W. F., Balcom, P. H., and Hammerschmidt, C. R. (2003). Determination of the mercury complexation characteristics of dissolved organic matter in natural waters with ‘reducible Hg’ titrations. Environmental Science & Technology 37, 3316–3322.
| Determination of the mercury complexation characteristics of dissolved organic matter in natural waters with ‘reducible Hg’ titrations.Crossref | GoogleScholarGoogle Scholar |
Lamborg, C., Bowman, K., Hammerschmidt, C., Gilmour, C., Munson, K., Selin, N., and Tseng, C. M. (2014). Mercury in the anthropocene ocean. Oceanography 27, 76–87.
| Mercury in the anthropocene ocean.Crossref | GoogleScholarGoogle Scholar |
Le Treut, H., Somerville, R., Cubasch, U., Ding, Y., Mauritzen, C., Mokssit, A., Peterson, T., and Prather, M. (2007). Historical overview of climate change. In ‘Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change’. (Eds S. Solomon, D. Qin, M. Manning, Z. Chen, M. Marquis, K. B. Averyt, M. Tignor, and H. L. Miller.) pp. 95–127. (Cambridge University Press: Cambridge, UK, and New York, NY, USA.)
Leal, M. F. C., Vasconcelos, M. T. S. D., and Van den Berg, C. M. (1999). Copper-induced release of complexing ligands similar to thiols by Emiliania huxleyi in seawater cultures. Limnology and Oceanography 44, 1750–1762.
| Copper-induced release of complexing ligands similar to thiols by Emiliania huxleyi in seawater cultures.Crossref | GoogleScholarGoogle Scholar |
Leal, P. P., Hurd, C. L., Sander, S. G., Armstrong, E., and Roleda, M. Y. (2016). Copper ecotoxicology of marine algae: a methodological appraisal. Chemistry and Ecology 32, 786–800.
| Copper ecotoxicology of marine algae: a methodological appraisal.Crossref | GoogleScholarGoogle Scholar |
Levin, L. A., Baco, A. R., Bowden, D. A., Colaco, A., Cordes, E. E., Cunha, M. R., Demopoulos, A. W., Gobin, J., Grupe, B. M., Le, J., and Metaxas, A. (2016). Hydrothermal vents and methane seeps: rethinking the sphere of influence. Frontiers in Marine Science 3, 72.
| Hydrothermal vents and methane seeps: rethinking the sphere of influence.Crossref | GoogleScholarGoogle Scholar |
Lidbury, I., Johnson, V., Hall-Spencer, J. M., Munn, C. B., and Cunliffe, M. (2012). Community-level response of coastal microbial biofilms to ocean acidification in a natural carbon dioxide vent ecosystem. Marine Pollution Bulletin 64, 1063–1066.
| Community-level response of coastal microbial biofilms to ocean acidification in a natural carbon dioxide vent ecosystem.Crossref | GoogleScholarGoogle Scholar | 22414852PubMed |
Lombardi, C., Rodolfo-Metalpa, R., Cocito, S., Gambi, M. C., and Taylor, P. D. (2011). Structural and geochemical alterations in the Mg calcite bryozoan Myriapora truncata under elevated seawater pCO2 simulating ocean acidification. Marine Ecology 32, 211–221.
| Structural and geochemical alterations in the Mg calcite bryozoan Myriapora truncata under elevated seawater pCO2 simulating ocean acidification.Crossref | GoogleScholarGoogle Scholar |
Lorenzo, J. I., Beiras, R., Mubiana, V. K., and Blust, R. (2005). Copper uptake by Mytilus edulis in the presence of humic acids. Environmental Toxicology and Chemistry 24, 973–980.
| Copper uptake by Mytilus edulis in the presence of humic acids.Crossref | GoogleScholarGoogle Scholar | 15839573PubMed |
Lorenzo, J. I., Nieto-Cid, M., Álvarez-Salgado, X. A., Pérez, P., and Beiras, R. (2007). Contrasting complexing capacity of dissolved organic matter produced during the onset, development and decay of a simulated bloom of the marine diatom Skeletonema costatum. Marine Chemistry 103, 61–75.
| Contrasting complexing capacity of dissolved organic matter produced during the onset, development and decay of a simulated bloom of the marine diatom Skeletonema costatum.Crossref | GoogleScholarGoogle Scholar |
Lucia, M., Campos, M. L. A. M., and van den Berg, C. M. G. (1994). Determination of copper complexation in sea water by cathodic stripping voltammetry and ligand competition with salicylaldoxime. Analytica Chimica Acta 284, 481–496.
| Determination of copper complexation in sea water by cathodic stripping voltammetry and ligand competition with salicylaldoxime.Crossref | GoogleScholarGoogle Scholar |
Maldonado, M. T., Allen, A. E., Chong, J. S., Lin, K., Leus, D., Karpenko, N., and Harris, S. L. (2006). Copper-dependent iron transport in coastal and oceanic diatoms. Limnology and Oceanography 51, 1729–1743.
| Copper-dependent iron transport in coastal and oceanic diatoms.Crossref | GoogleScholarGoogle Scholar |
McCulloch, M. T., Holcomb, M., Rankenburg, K., and Trotter, J. A. (2014). Rapid, high-precision measurements of boron isotopic compositions in marine carbonates. Rapid Communications in Mass Spectrometry 28, 2704–2712.
| Rapid, high-precision measurements of boron isotopic compositions in marine carbonates.Crossref | GoogleScholarGoogle Scholar | 25380492PubMed |
Mehrbach, C., Culberson, C. H., Hawley, J. E., and Pytkowicx, R. M. (1973). Measurement of the apparent dissociation constants of carbonic acid in seawater at atmospheric pressure. Limnology and Oceanography 18, 897–907.
| Measurement of the apparent dissociation constants of carbonic acid in seawater at atmospheric pressure.Crossref | GoogleScholarGoogle Scholar |
Middag, R., Séférian, R., Conway, T. M., John, S. G., Bruland, K. W., and de Baar, H. J. (2015). Intercomparison of dissolved trace elements at the Bermuda Atlantic Time Series station. Marine Chemistry 177, 476–489.
| Intercomparison of dissolved trace elements at the Bermuda Atlantic Time Series station.Crossref | GoogleScholarGoogle Scholar |
Millero, F. J., Woosley, R., Ditrolio, B., and Waters, J. (2009). Effect of ocean acidification on the speciation of metals in seawater. Oceanography 22, 72–85.
| Effect of ocean acidification on the speciation of metals in seawater.Crossref | GoogleScholarGoogle Scholar |
Moffett, J. W., and Brand, L. E. (1996). Production of strong, extracellular Cu chelators by marine cyanobacteria in response to Cu stress. Limnology and Oceanography 41, 388–395.
| Production of strong, extracellular Cu chelators by marine cyanobacteria in response to Cu stress.Crossref | GoogleScholarGoogle Scholar |
Moffett, J., Zika, R., and Brand, L. (1990). Distribution and potential sources and sinks of copper chelators in the Sargasso Sea. Deep-Sea Research – A. Oceanographic Research Papers 37, 27–36.
| Distribution and potential sources and sinks of copper chelators in the Sargasso Sea.Crossref | GoogleScholarGoogle Scholar |
Morel, F. M. (1983). ‘Principles of Aquatic Chemistry.’ (Wiley: New York, NY, USA.)
Morrow, K. M., Bourne, D. G., Humphrey, C., Botté, E. S., Laffy, P., Zaneveld, J. J., Uthicke, S., Fabricius, K. E., and Webster, N. S. (2015). Natural volcanic CO2 seeps reveal future trajectories for host–microbial associations in corals and sponges. The ISME Journal 9, 894–908.
| Natural volcanic CO2 seeps reveal future trajectories for host–microbial associations in corals and sponges.Crossref | GoogleScholarGoogle Scholar | 25325380PubMed |
Mousavi, A., Chávez, R. D., Ali, A. M. S., and Cabaniss, S. E. (2011). Mercury in natural waters: a mini-review. Environmental Forensics 12, 14–18.
| Mercury in natural waters: a mini-review.Crossref | GoogleScholarGoogle Scholar |
Nagelkerken, I., Russell, B. D., Gillanders, B. M., and Connell, S. D. (2016). Ocean acidification alters fish populations indirectly through habitat modification. Nature Climate Change 6, 89–93.
| Ocean acidification alters fish populations indirectly through habitat modification.Crossref | GoogleScholarGoogle Scholar |
Nairn, I. A., Houghton, B. F., and Cole, J. W. (1996). ‘Volcanic Hazards at White Island’, 2nd edn. Volcanic Hazards Information Series 3. (Ministry of Civil Defence: Palmerston North, New Zealand.)
Ngatia, L. III, J. G., Moriasi, D., and Taylor, R. (2019). Nitrogen and phosphorus eutrophication in marine ecosystems. In ‘Monitoring of Marine Pollution’. (Eds H. B. Fouzia.) pp. 1–17. (IntechOpen.)
Oldham, V. E., Owings, S. M., Jones, M. R., Tebo, B. M., and Luther, G. W. (2015). Chesapeake Bay. Marine Chemistry 171, 58–66.
| Chesapeake Bay.Crossref | GoogleScholarGoogle Scholar |
Omanović, D., Garnier, C., Louis, Y., Lenoble, V., Mounier, S., and Pižeta, I. (2010). Significance of data treatment and experimental setup on the determination of copper complexing parameters by anodic stripping voltammetry. Analytica Chimica Acta 664, 136–143.
| Significance of data treatment and experimental setup on the determination of copper complexing parameters by anodic stripping voltammetry.Crossref | GoogleScholarGoogle Scholar | 20363395PubMed |
Paquin, P. R., Santore, R. C., Wu, K. B., Kavvadas, C. D., and Di Toro, D. M. (2000). The biotic ligand model: a model of the acute toxicity of metals to aquatic life. Environmental Science & Policy 3, 175–182.
| The biotic ligand model: a model of the acute toxicity of metals to aquatic life.Crossref | GoogleScholarGoogle Scholar |
Peers, G., Quesnel, S. A., and Price, N. M. (2005). Copper requirements for iron acquisition and growth of coastal and oceanic diatoms. Limnology and Oceanography 50, 1149–1158.
| Copper requirements for iron acquisition and growth of coastal and oceanic diatoms.Crossref | GoogleScholarGoogle Scholar |
Pichler, T., Biscéré, T., Kinch, J., Zampighi, M., Houlbrèque, F., and Rodolfo-Metalpa, R. (2019). Suitability of the shallow water hydrothermal system at Ambitle Island (Papua New Guinea) to study the effect of high pCO2 on coral reefs. Marine Pollution Bulletin 138, 148–158.
| Suitability of the shallow water hydrothermal system at Ambitle Island (Papua New Guinea) to study the effect of high pCO2 on coral reefs.Crossref | GoogleScholarGoogle Scholar | 30660256PubMed |
Propp, L. N., Obzhirov, A. I., and Propp, M. V. (1992). The gas and hydrochemical anomalies in the bottom layer of water in area of volcanic activity (Bay of Plenty, New Zealand). Okeanologiya 32, 680–686.
Radford-Knȩry, J., and Cutter, G. A. (1994). Biogeochemistry of dissolved hydrogen sulfide species and carbonyl sulfide in the western North Atlantic Ocean. Geochimica et Cosmochimica Acta 58, 5421–5431.
| Biogeochemistry of dissolved hydrogen sulfide species and carbonyl sulfide in the western North Atlantic Ocean.Crossref | GoogleScholarGoogle Scholar |
Rainbow, P. S. (2002). Trace metal concentrations in aquatic invertebrates: why and so what? Environmental Pollution 120, 497–507.
| Trace metal concentrations in aquatic invertebrates: why and so what?Crossref | GoogleScholarGoogle Scholar | 12442773PubMed |
Rapp, I., Schlosser, C., Rusiecka, D., Gledhill, M., and Achterberg, E. P. (2017). Automated preconcentration of Fe, Zn, Cu, Ni, Cd, Pb, Co, and Mn in seawater with analysis using high-resolution sector field inductively-coupled plasma mass spectrometry. Analytica Chimica Acta 976, 1–13.
| Automated preconcentration of Fe, Zn, Cu, Ni, Cd, Pb, Co, and Mn in seawater with analysis using high-resolution sector field inductively-coupled plasma mass spectrometry.Crossref | GoogleScholarGoogle Scholar | 28576313PubMed |
Redfield, A. C., Ketchum, B. H., and Richards, F. A. (1963). The influence of organisms on the composition of sea-water. In ‘The Composition of Seawater: Comparative and Descriptive Oceanography. The Sea: Ideas and Observations on Progress in the Study of the seas, Vol. 2’. (Ed. M. N. Hill.) pp. 26–77. (Interscience Publishers: New York, NY, USA.)
Ridgway, N. M., and Greig, M. J. N. (1986). Water movements in Bay of Plenty, New Zealand. New Zealand Journal of Marine and Freshwater Research 20, 447–453.
| Water movements in Bay of Plenty, New Zealand.Crossref | GoogleScholarGoogle Scholar |
Riebesell, U., and Gattuso, J. P. (2015). Lessons learned from ocean acidification research. Nature Climate Change 5, 12–14.
| Lessons learned from ocean acidification research.Crossref | GoogleScholarGoogle Scholar |
Rivest, E. B., Comeau, S., and Cornwall, C. E. (2017). The role of natural variability in shaping the response of coral reef organisms to climate change. Current Climate Change Reports 3, 271–281.
| The role of natural variability in shaping the response of coral reef organisms to climate change.Crossref | GoogleScholarGoogle Scholar |
Roleda, M. Y., Cornwall, C. E., Feng, Y., McGraw, C. M., Smith, A. M., and Hurd, C. L. (2015). Effect of ocean acidification and pH fluctuations on the growth and development of coralline algal recruits, and an associated benthic algal assemblage. PLoS One 10, e0140394.
| Effect of ocean acidification and pH fluctuations on the growth and development of coralline algal recruits, and an associated benthic algal assemblage.Crossref | GoogleScholarGoogle Scholar | 26469945PubMed |
Romera-Castillo, C., Sarmento, H., Álvarez-Salgado, X. A., Gasol, J. M., and Marrasé, C. (2011). Net production/consumption of fluorescent coloured dissolved organic matter by natural bacterial assemblages growing on marine phytoplankton exudates. Applied and Environmental Microbiology 77, 7490–7498.
| Net production/consumption of fluorescent coloured dissolved organic matter by natural bacterial assemblages growing on marine phytoplankton exudates.Crossref | GoogleScholarGoogle Scholar | 21742918PubMed |
Rossi, G., and Tunnicliffe, V. (2017). Trade-offs in a high CO2 habitat on a subsea volcano: condition and reproductive features of a bathymodioline mussel. Marine Ecology Progress Series 574, 49–64.
| Trade-offs in a high CO2 habitat on a subsea volcano: condition and reproductive features of a bathymodioline mussel.Crossref | GoogleScholarGoogle Scholar |
Samson, C. R., Sikes, E. L., and Howard, W. R. (2005). Deglacial paleoceanographic history of the Bay of Plenty, New Zealand. Paleoceanography 20, PA4017.
| Deglacial paleoceanographic history of the Bay of Plenty, New Zealand.Crossref | GoogleScholarGoogle Scholar |
Sander, S. G., and Koschinsky, A. (2011). Metal flux from hydrothermal vents increased by organic complexation. Nature Geoscience 4, 145–150.
| Metal flux from hydrothermal vents increased by organic complexation.Crossref | GoogleScholarGoogle Scholar |
Sander, S. G., Koschinsky, A., Massoth, G., Stott, M., and Hunter, K. A. (2007). Organic complexation of copper in deep-sea hydrothermal vent systems. Environmental Chemistry 4, 81–89.
| Organic complexation of copper in deep-sea hydrothermal vent systems.Crossref | GoogleScholarGoogle Scholar |
Sander, S. G., Hunter, K., and Frew, R. (2009). 14 – Sampling and measurements of trace metals in seawater. In ‘Practical Guidelines for the Analysis of Seawater’. (Ed. O. Wurl.) pp. 305–327. (CRC Press: Boca Raton, FL, USA.)
Sarano, F., Murphy, R. C., Houghton, B. F., and Hedenquist, J. W. (1989). Preliminary observations of submarine geothermal activity in the vicinity of White Island volcano, Taupo volcanic zone, New Zealand. Journal of the Royal Society of New Zealand 19, 449–459.
| Preliminary observations of submarine geothermal activity in the vicinity of White Island volcano, Taupo volcanic zone, New Zealand.Crossref | GoogleScholarGoogle Scholar |
Semeniuk, D. M., Bundy, R. M., Payne, C. D., Barbeau, K. A., and Maldonado, M. T. (2015). Acquisition of organically complexed copper by marine phytoplankton and bacteria in the northeast subarctic Pacific Ocean. Marine Chemistry 173, 222–233.
| Acquisition of organically complexed copper by marine phytoplankton and bacteria in the northeast subarctic Pacific Ocean.Crossref | GoogleScholarGoogle Scholar |
Shears, N. T., and Babcock, R. C. (2007). Quantitative description of mainland New Zealand’s shallow subtidal reef communities. Science & Technical Publication, Department of Conservation, Wellington, New Zealand.
Shimotori, K., Omori, Y., and Hama, T. (2009). Bacterial production of marine humic-like fluorescent dissolved organic matter and its biogeochemical importance. Aquatic Microbial Ecology 58, 55–66.
| Bacterial production of marine humic-like fluorescent dissolved organic matter and its biogeochemical importance.Crossref | GoogleScholarGoogle Scholar |
Sommer, U. (2012). Experimental systems in aquatic ecology. In ‘eLS’. pp. 1–8. (Wiley: Chichester, UK.) https://doi.org/
Stockdale, A., Tipping, E., Lofts, S., and Mortimer, R. J. (2016). Effect of ocean acidification on organic and inorganic speciation of trace metals. Environmental Science & Technology 50, 1906–1913.
| Effect of ocean acidification on organic and inorganic speciation of trace metals.Crossref | GoogleScholarGoogle Scholar |
Sunda, W. G., and Huntsman, S. A. (1996). Antagonisms between cadmium and zinc toxicity and manganese limitation in a coastal diatom. Limnology and Oceanography 41, 373–387.
| Antagonisms between cadmium and zinc toxicity and manganese limitation in a coastal diatom.Crossref | GoogleScholarGoogle Scholar |
Sunda, W. G., and Huntsman, S. A. (1998a). Control of Cd concentrations in a coastal diatom by interactions among free ionic Cd, Zn, and Mn in seawater. Environmental Science & Technology 32, 2961–2968.
| Control of Cd concentrations in a coastal diatom by interactions among free ionic Cd, Zn, and Mn in seawater.Crossref | GoogleScholarGoogle Scholar |
Sunda, W. G., and Huntsman, S. A. (1998b). Interactions among Cu2+, Zn2+, and Mn2+ in controlling cellular Mn, Zn, and growth rate in the coastal algae Chlamydomonas. Limnology and Oceanography 43, 1055–1064.
| Interactions among Cu2+, Zn2+, and Mn2+ in controlling cellular Mn, Zn, and growth rate in the coastal algae Chlamydomonas.Crossref | GoogleScholarGoogle Scholar |
Sunda, W. G., and Huntsman, S. A. (1998c). Interactive effects of external manganese, the toxic metals copper and zinc, and light in controlling cellular manganese and growth in a coastal diatom. Limnology and Oceanography 43, 1467–1475.
| Interactive effects of external manganese, the toxic metals copper and zinc, and light in controlling cellular manganese and growth in a coastal diatom.Crossref | GoogleScholarGoogle Scholar |
Sunda, W. G., Barber, R. T., and Huntsman, S. A. (1981). Phytoplankton growth in nutrient rich seawater: importance of copper–manganese cellular interactions. Journal of Marine Research 39, 567–586.
Takahashi, M., Noonan, S. H. C., Fabricius, K. E., and Collier, C. J. (2016). The effects of long-term in situ CO2 enrichment on tropical seagrass communities at volcanic vents. ICES Journal of Marine Science 73, 876–886.
| The effects of long-term in situ CO2 enrichment on tropical seagrass communities at volcanic vents.Crossref | GoogleScholarGoogle Scholar |
Tarasov, V. G. (2006). Effects of shallow-water hydrothermal venting on biological communities of coastal marine ecosystems of the Western Pacific. Advances in Marine Biology 50, 267–421.
| Effects of shallow-water hydrothermal venting on biological communities of coastal marine ecosystems of the Western Pacific.Crossref | GoogleScholarGoogle Scholar |
Tarasov, V. G., Propp, M. V., Propp, L. N., Blinov, S. V., and Kamenev, G. M. (1986). Shallow-water hydrothermal vents and a unique ecosystem of Kraternaya Caldera (Kurile Islands). Biologiya Morya 2, 72–74.
Tarasov, V. G., Gebruk, A. V., Shulkin, V. M., Kamenev, G. M., Fadeev, V. I., Kosmynin, V. N., Malakhov, V. V., Starynin, D. A., and Obzhirov, A. I. (1999). Effect of shallow-water hydrothermal venting on the biota of Matupi Harbour (Rabaul Caldera, New Britain Island, Papua New Guinea). Continental Shelf Research 19, 79–116.
| Effect of shallow-water hydrothermal venting on the biota of Matupi Harbour (Rabaul Caldera, New Britain Island, Papua New Guinea).Crossref | GoogleScholarGoogle Scholar |
Tarasov, V. G., Gebruk, A. V., Mironov, A. N., and Moskalev, L. I. (2005). Deep-sea and shallow-water hydrothermal vent communities: two different phenomena? Chemical Geology 224, 5–39.
| Deep-sea and shallow-water hydrothermal vent communities: two different phenomena?Crossref | GoogleScholarGoogle Scholar |
Tarran, G. A., and Bruun, J. T. (2015). Nanoplankton and picoplankton in the western English Channel: abundance and seasonality from 2007–2013. Progress in Oceanography 137, 446–455.
| Nanoplankton and picoplankton in the western English Channel: abundance and seasonality from 2007–2013.Crossref | GoogleScholarGoogle Scholar |
Teixidó, N., Gambi, M. C., Parravacini, V., Kroeker, K., Micheli, F., Villéger, S., and Ballesteros, E. (2018). Functional biodiversity loss along natural CO2 gradients. Nature Communications 9, 5149.
| Functional biodiversity loss along natural CO2 gradients.Crossref | GoogleScholarGoogle Scholar |
Tercier-Waeber, M. L., Stoll, S., and Slaveykova, V. (2012). Trace metal behaviour in surface waters: emphasis on dynamic speciation, sorption processes and bioavailability. Archives des Sciences 65, 119–142.
Thomas, W. H., and Seibert, D. L. (1977). Effects of copper on the dominance and the diversity of algae: controlled ecosystem pollution experiment. Bulletin of Marine Science 27, 23–33.
Thomas, W. H., Hollibaugh, J. T., Seibert, D. L. R., and Wallace, G. T. (1980). Toxicity of a mixture of ten metals to phytoplankton. Marine Ecology Progress Series 2, 213–220.
| Toxicity of a mixture of ten metals to phytoplankton.Crossref | GoogleScholarGoogle Scholar |
Tortell, P. D., and Price, N. M. (1996). Cadmium toxicity and zinc limitation in centric diatoms of the genus Thalassiosira. Marine Ecology Progress Series 138, 245–254.
| Cadmium toxicity and zinc limitation in centric diatoms of the genus Thalassiosira.Crossref | GoogleScholarGoogle Scholar |
Trotter, J., Montagna, P., McCulloch, M., Silenzi, S., Reynaud, S., Mortimer, G., Martin, S., Ferrier-Pagès, C., Gattuso, J. P., and Rodolfo-Metalpa, R. (2011). Quantifying the pH ‘vital effect’ in the temperate zooxanthellate coral Cladocora caespitosa: validation of the boron seawater pH proxy. Earth and Planetary Science Letters 303, 163–173.
| Quantifying the pH ‘vital effect’ in the temperate zooxanthellate coral Cladocora caespitosa: validation of the boron seawater pH proxy.Crossref | GoogleScholarGoogle Scholar |
Van Briesen, J. M., Small, M., Weber, C., and Wilson, J. (2010). Modelling chemical speciation: thermodynamics, kinetics and uncertainty. In ‘Modelling of Pollutants in Complex Environmental Systems, Vol. 2’. (Ed. G. Hanrahan.) Chapt. 4, pp. 133–149. (ILM Publications.)
Vizzini, S., Di Leonardo, R., Costa, V., Tramati, C. D., Luzzu, F., and Mazzola, A. (2013). Trace element bias in the use of CO2 vents as analogues for low pH environments: implications for contamination levels in acidified oceans. Estuarine, Coastal and Shelf Science 134, 19–30.
| Trace element bias in the use of CO2 vents as analogues for low pH environments: implications for contamination levels in acidified oceans.Crossref | GoogleScholarGoogle Scholar |
Vraspir, J. M., and Butler, A. (2009). Chemistry of marine ligands and siderophores. Annual Review of Marine Science 1, 43–63.
| Chemistry of marine ligands and siderophores.Crossref | GoogleScholarGoogle Scholar |
Wernberg, T., Smale, D. A., and Thomsen, M. S. (2012). A decade of climate change experiments on marine organisms: procedures, patterns and problems. Global Change Biology 18, 1491–1498.
| A decade of climate change experiments on marine organisms: procedures, patterns and problems.Crossref | GoogleScholarGoogle Scholar |
Whitby, H., Posacka, A. M., Maldonado, M. T., and Van den Berg, C. M. (2018). Copper-binding ligands in the NE Pacific. Marine Chemistry 204, 36–48.
| Copper-binding ligands in the NE Pacific.Crossref | GoogleScholarGoogle Scholar |
Wisshak, M., Schönberg, C. H., Form, A., and Freiwald, A. (2013). Effects of ocean acidification and global warming on reef bioerosion lessons from a clionaid sponge. Aquatic Biology 19, 111–127.
| Effects of ocean acidification and global warming on reef bioerosion lessons from a clionaid sponge.Crossref | GoogleScholarGoogle Scholar |
Zeng, X., Chen, X., and Zhuang, J. (2015). The positive relationship between ocean acidification and pollution. Marine Pollution Bulletin 91, 14–21.
| The positive relationship between ocean acidification and pollution.Crossref | GoogleScholarGoogle Scholar |
Zitoun, R., Clearwater, S. J., Hassler, C., Thompson, K. J., Albert, A., and Sander, S. G. (2018). Copper toxicity to blue mussel embryos (Mytilus galloprovincialis) – the effect of natural dissolved organic matter on copper toxicity in estuarine waters. The Science of the Total Environment 65, 300–314.
Ziveri, P., Passaro, M., Incarbona, A., Milazzo, M., Rodolfo-Metalpa, R., and Hall-Spencer, J. M. (2014). Decline in coccolithophore diversity and impact on coccolith morphogenesis along a natural CO2 gradient. The Biological Bulletin 226, 282–290.
| Decline in coccolithophore diversity and impact on coccolith morphogenesis along a natural CO2 gradient.Crossref | GoogleScholarGoogle Scholar |



