
Australian Journal of Chemistry
Volume 66 Number 7 2013
RESEARCH FRONT: Proteomics and Mass Spectrometry
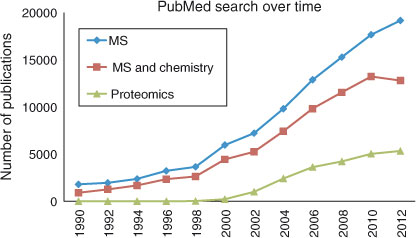
Deciphering the biological and clinical significance of the proteins is investigated by mass spectrometry in a relatively new field, named proteomics. Mass spectrometry is, however, also used in chemistry for many years. In this Research Front we try to show the potential use of mass spectrometry in chemical, environmental and biomedical research and also to illustrate the applications of mass spectrometry in proteomics.
CH13137Applications of Mass Spectrometry in Proteomics
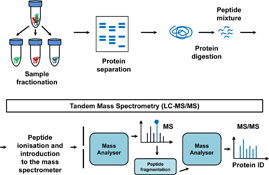
Mass spectrometry-based proteomics has been widely applied to the analysis of biological samples and has greatly advanced our understanding of cellular and physiological processes. These approaches allow accurate protein identification and characterisation, analysis of protein post-translational modifications (PTMs) and provide quantitative information as well as a basis for protein–protein interaction (PPI) investigations.
CH13144Identification of Post-Translational Modifications by Mass Spectrometry
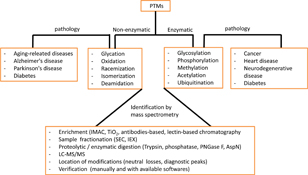
Regulation of protein activity, structure, stability, and turnover is in part controlled by their post-translational modifications (PTMs) such as phosphorylation, glycosylation, or acetylation. Mass spectrometry (MS) has become the method of choice for the analysis of PTMs. Here we describe how to use MS to identify natural and artificial (e.g. carbamidomethylation or oxidation) PTMs.
CH13164Chemical Cross-linking and Mass Spectrometry for the Structural Analysis of Protein Assemblies
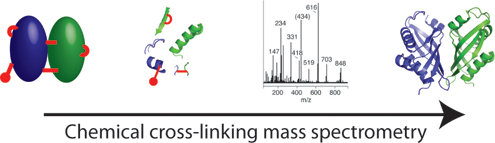
Protein–protein interactions are central to most biological processes, and represent a diverse target for modern structural biology efforts. Mass spectrometry is emerging as an invaluable tool for the analysis of intact protein complexes. This review describes the attributes of mass spectrometry for the structural investigation of multiprotein assemblies in the context of recent developments and highlights in the field.
CH13174Molecular Characterization of Collagen Hydroxylysine O-Glycosylation by Mass Spectrometry: Current Status
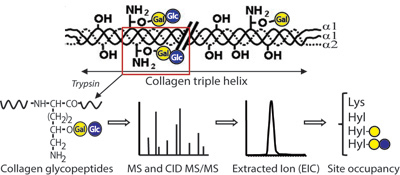
This review summarizes the recent literature on collagen glycosylation analysis by mass spectrometry. State-of-the art methods and approaches for data interpretation are presented.
CH13159Mass Spectrometry Investigation of Glycosylation Aberration via De-N-Glycopeptide Analysis
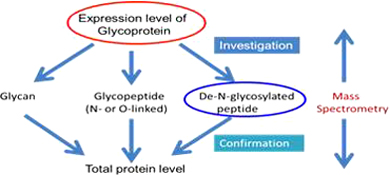
Mass spectrometry-based de-N-glycopeptide analysis is an evolving method for the investigation of glycosylation aberration in biomarker discovery research. Major advantages are: (a) increase in sensitivity and decrease in heterogeneity, (b) unambiguous identification of glycosylation site, (c) multi-labelling flexibility, (d) determination of site specific glycan changes, and (e) accurate quantitation. Glycopeptides identified in this manner may serve as early stage biomarkers.
CH13173Mass Spectrometry of Gangliosides from Human Sensory and Motor Cortex
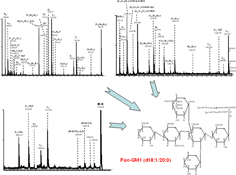
Gangliosides display a large variability related to the specialised function of brain regions. Here, we carried out the first comparative assessment of ganglioside profile in human motor and sensory cortex by high resolution mass spectrometry. Fragmentation analysis revealed in motor cortex a fucose-ganglioside Fuc-GM1 (d18 : 1/20 : 0) species with fucose and sialic acid attached to the inner galactose.
CH13179Formation of Ternary Complexes of Iron(III) Cations in Solution and Gas Phase
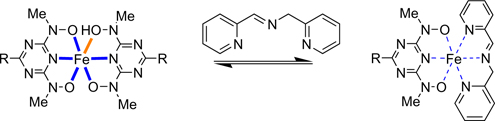
Qualitative determination of metal complexes by ESI MS spectroscopy was used to study of equilibrium between labile iron(iii) complexes of terdentate ligands. Obtained data on equilibrium constants show reasonably good correlation with equilibrium studied by conventional UV-Vis titrations.
CH13166Environmental Mass Spectrometry in the North American Great Lakes Fish Monitoring and Surveillance Program
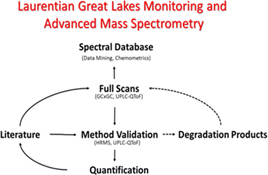
The Great Lakes Fish Monitoring and Surveillance Program has served to protect the Laurentian Great Lakes of North America for decades by screening lake trout and walleye for toxic chemicals. Using advanced mass spectrometry the program is creating a living chemical database for new toxins affecting the Great Lakes.
CH13094Formation of Dynamic Duolayer Systems at the Air/Water Interface by using Non-ionic Hydrophilic Polymers
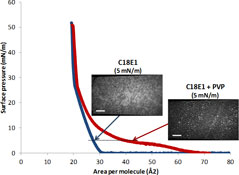
A water-soluble polymer was incorporated into a monolayer forming composition before application on the water surface. A different monolayer film, a duolayer, is demonstrated with changes in fundamental properties and formation mechanism.
CH13101A Much-Needed Mechanism and Reaction Rate for the Oxidation of Phenols with ClO2: A Joint Experimental and Computational Study
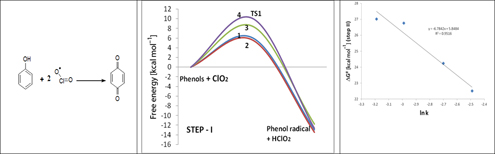
The kinetics of phenol oxidation with chlorine dioxide were analysed; 2,4,6-trichlorophenol exhibits a lower oxidation rate than the other phenols in agreement with the Hammett plot. The reaction is second order with respect to phenol and first order with respect to ClO2. A mechanism is proposed and is supported by DFT computations. The activation barriers predicted the observed reaction trend.
CH13083Electrochemical Characterisation of Poly(aniline-co-N-methylaniline) and Poly(aniline-co-N-ethylaniline) Films on Pencil Graphite Electrode for Supercapacitor Applications
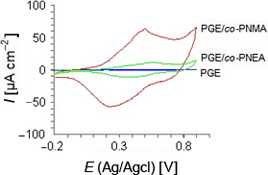
Poly(aniline-co-N-methylaniline) and poly(aniline-co-N-ethylaniline) coated pencil graphite electrodes have been electrochemically characterised by different methods and their specific capacitance values have been determined. They can be used as electrode active material for low voltage supercapacitor applications.
CH13121Stable Molecular Complex of Squaric Acid with 2-(Quinuclidinium)propionate
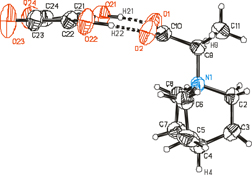
Squaric acid (H2SQ) forms a complex with 2-(quinuclidinium)propionate (QNPr). The molecules of H2SQ and zwitterions of QNPr are bridged by two strong non-equivalent O–H⋯O hydrogen bonds of 2.476(2) and 2.482(1) Å. The complex is investigated by X-ray diffraction, FTIR and NMR techniques, and the results are supported by DFT calculations.



