
Australian Journal of Chemistry
Volume 66 Number 12 2013
RESEARCH FRONT: Fragment-Based Drug Discovery
A two-day workshop ‘Fragment-Based Drug Design Down Under’ was held at the Monash Institute of Pharmaceutical Sciences, Parkville, in November 2012. This Research Front captures the spirit of this workshop and highlights some of the leading work being undertaken in Australia in this rapidly developing area.
CH13280Design and Evaluation of the Performance of an NMR Screening Fragment Library
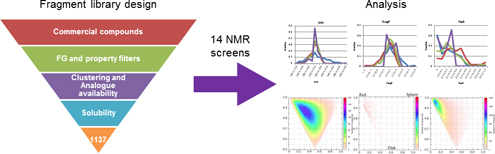
Fragment library design and evaluation is an essential part of fragment screening. We present the analysis of our fragment library that has been screened by NMR against 14 targets and discuss the results and implications for future fragment library designs.
CH13325Construction of the CSIRO Fragment Library
A proprietary fragment library was developed by identifying and extracting subsets of CSIRO’s Compound Collection using two complimentary approaches. Over time, the use of surface plasmon resonance as a front-line screening tool has enabled identification and exclusion of problematic compounds and led to a more reliable fragment screening library.
CH13551PAINS: Relevance to Tool Compound Discovery and Fragment-Based Screening
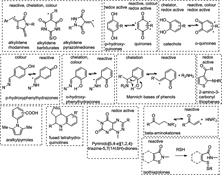
Pan assay interference compounds (PAINS) are readily discovered in any bioassay and can appear to be selective, optimisable, and real. However, the chances are very high that they will be found to be non-progressable, often after a significant waste of resources. Unfortunately, they frequently provide cellular readouts, leading to their selection ahead of more optimisable hits.
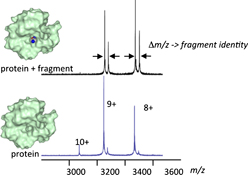
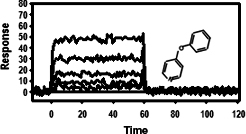
Fragment-based drug discovery (FBDD) is a recently validated approach that has confronted the challenge to identify more suitable small molecules as chemical starting points for drug discovery. In this review, the attributes of native state mass spectrometry for screening fragments to support continued FBDD successes is described.
CH13279Interrogating Fragments Using a Protein Thermal Shift Assay
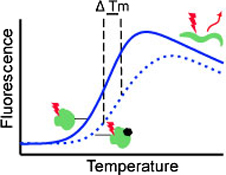
Protein thermal shift assays are a relatively rapid and inexpensive technique for the identification of low molecular weight compound interactions with protein targets. We review how the technique has been applied recently in fragment-based drug discovery programs for library screening and hit validation, and discuss some of the challenges of executing the approach.
CH13302Fragment Screening for the Modelling Community: SPR, ITC, and Crystallography
The SAMPL3 challenge aimed to predict how small fragments bind to bovine trypsin. It brought together experimentalists and modellers in an effort to improve our understanding of biomolecular interactions and, in so doing, develop better modelling tools. Described here are the biophysical methods used to select and confirm fragments that bound to trypsin.
CH13286Detection and Prevention of Aggregation-based False Positives in STD-NMR-based Fragment Screening
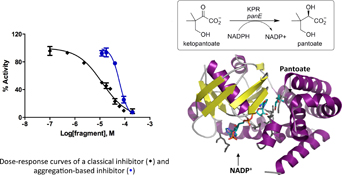
STD-NMR-based fragment screening against ketopantoate reductase identified inhibitors that were shown to be aggregators. Rescreening with detergent present in both STD-NMR and biochemical assays identified genuine inhibitors.
CH13290Blind Man’s Bluff – Elaboration of Fragment Hits in the Absence of Structure for the Development of Antitrypsin Deficiency Inhibitors
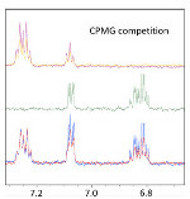
Using saturation transfer difference NMR, we screened for fragments binding to the Z mutant of α1-antitrypsin in an attempt to prevent its polymerization. In the absence of an atomic-resolution structure of Z-AAT, we used CPMG NMR competition experiments to achieve an optimized hit.
CH13266Development of Inhibitors of Plasmodium falciparum Apical Membrane Antigen 1 Based on Fragment Screening
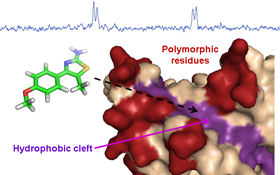
Apical membrane antigen 1 (AMA1) is an essential component of the moving junction complex used by Plasmodium falciparum to invade human red blood cells. We have identified fragments that bind to the AMA1 hydrophobic cleft and will serve as starting scaffolds for developing small molecule inhibitors with broad strain specificity.
CH13298Fragment Screening on Staphylococcus aureus HPPK – a Folate Pathway Target
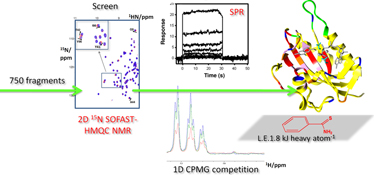
The first fragment screen of the folate pathway target, 6-hydroxymethyl-7,8-dihydropterin pyrophosphokinase (HPPK) from Staphylococcus aureus was performed using NMR and surface plasmon resonance. Chemical shift mapping revealed several pterin site binders with good ligand efficiency. They represent good starting points to develop into drug-like leads towards novel antimicrobials.

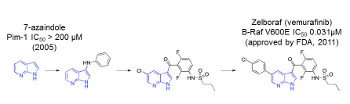
Fragment-Based Drug Discovery (FBDD) comes in a variety of ‘flavours’. Regardless of the chosen primary screen, robust validation and optimisation of fragment hits have delivered its first clinically used drug Zelboraf, as well as numerous clinical development candidates. For FBDD, the future is very bright.
CH13397Characterization Methods for Starch-Based Materials: State of the Art and Perspectives
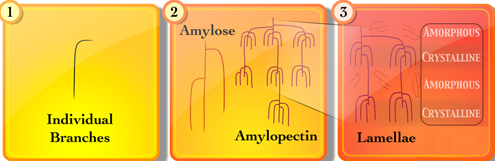
Starch supplies at least 50 % of our food energy, and is a renewable resource of significant current and increasing use as a biomaterial. It is a complex branched polymer of glucose, and functional properties depend on structural characterization. This review gives an overview of current characterization techniques and challenges.
CH13397 Abstract | CH13397 Full Text | CH13397PDF (1.1 MB) Open Access Article
CH13375RAFT Polymerization of Monomers with Highly Disparate Reactivities: Use of a Single RAFT Agent and the Synthesis of Poly(styrene-block-vinyl acetate)
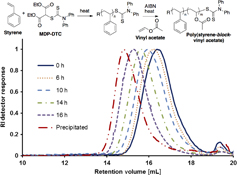
A single reversible addition–fragmentation chain transfer (RAFT) agent, malonate N,N-diphenyldithiocarbamate, is shown to successfully mediate the polymerization of several monomers with greatly differing reactivities, including both vinyl acetate and styrene, for which the synthesis of block copolymers is achieved. This RAFT agent may be useful to produce other block copolymers consisting of monomers with highly dissimilar reactivities.
CH13324A Mild and Convenient Synthesis of 1,2,3-Triiodoarenes via Consecutive Iodination/Diazotization/Iodination Strategy
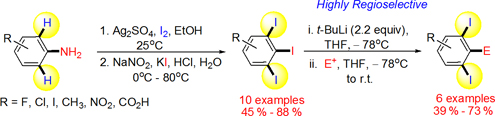
A concise and efficient method for the synthesis of 1,2,3-triiodoarenes and 1,2,3-trihaloarenes has been developed. This method is operationally simple, scalable, and is easy to workup and to purify. This report discloses a two-step synthesis of 1,2,3-triiodoarenes and 1,2,3-trihaloarenes that is rather general in scope and gives good yields (45–88 %). We also report the first regioselective metal-iodine exchange reactions of 1,2,3-triiodoarenes to provide ortho-diiodoaryl derivatives in good yields (up to 73 %), which are useful building blocks and indeed are hard to make by other means.
CH13297Methoxy-Poly(ethylene glycol)-block-Poly(ε-caprolactone) Bearing Pendant Aldehyde Groups as pH-Responsive Drug Delivery Carrier
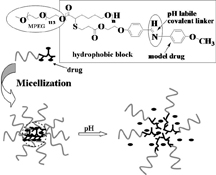
A biodegradable amphiphilic block copolymer bearing pendant aldehyde groups was synthesised by a combination of ring-opening polymerisation and thio-bromo ‘click’ chemistry. The free aldehyde groups were reacted with hydrophobic payloads by a pH responsive benzoic–imine linker. This new amphiphilic block copolymer is expected to have promising applications in biodegradable controlled drug delivery systems.
CH13273A Coumarin Fluorescent Probe for Ca2+ Containing Aza-Crown Ether Unit
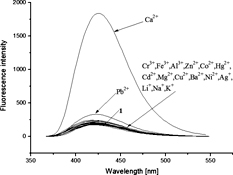
A novel fluorescent probe for Ca2+ based on coumarin containing an aza-crown ether unit was synthesised. The probe recognised Ca2+, and formed a 1 : 1 complex with Ca2+ in acetonitrile.
CH13420Synthesis, Characterisation, and X-Ray Crystal Structures of 8-Hydroxyquinoline Complexes of Group IV Metal Alkoxides
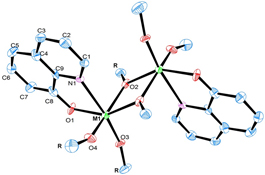
Interest in metal alkoxides revolves around their use as precursors for the preparation of metal oxides but they are easily hydrolysed and their chelating ligand is sometimes used to stabilise them. The present study details the synthesis and characterisation of three binuclear complexes from group IV metal alkoxides with 8-hydroxyquinoline which affords the compounds, [M2-μ-(OR)2(C9H6NO)2(OR)2] (1: M = Ti, R = Et, 2: M = Zr, R = iPr and 3: M = Hf, R = iPr).
CH13254Al(OTf)3 Catalysed Friedel-Crafts Michael Type Addition of Indoles to α,β-Unsaturated Ketones with PEG-200 as Recyclable Solvent
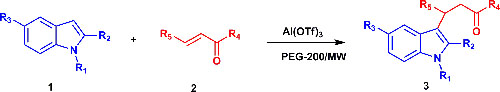
An Al(OTf)3 catalysed efficient Friedel–Crafts Michael type addition of indoles to α,β-unsaturated ketones using recyclable PEG-200 as an alternative reaction solvent is disclosed. The reaction under microwave irradiation is clean, leads to excellent yields in minutes and reduces the use of volatile organic compounds.



